Abstract
Objective
The study aimed to evaluate the feasibility and reproducibility of measuring phospholipase C zeta (PLCζ) using immunostaining in human sperm and to investigate the relationship between PLCζ immunoreactivity and DNA fragmentation and oxidation in human sperm.
Methods
Semen samples were obtained from participants (n=44) and processed by the conventional swim-up method. Sperm concentration, motility, normal form by strict morphology, DNA fragmentation index assessed by terminal deoxynucleotidyl transferase dUTP nick end labeling method and immunofluorescent expression for 8-hydroxy-2'-deoxyguanosine (8-OHdG) and PLCζ were assessed.
Results
When duplicate PLCζ tests were performed on two sperm samples from each of the 44 participants, similar results were obtained (74.1±9.4% vs. 75.4±9.7%). Two measurements of PLCζ were found to be highly correlated with each other (r=0.759, P<0.001). Immunoreactivity of PLCζ was not associated with donor's age, sperm concentration, motility, and the percentage of normal form as well as DNA fragmentation index. However, immunoreactivity of PLCζ showed a significant negative relationship with 8-OHdG immunoreactivity (r=-0.404, P=0.009).
The sperm-specific phospholipase C zeta (PLCζ) is a gamete-specific 70 kDa protein which is predominantly localized to the equatorial region of the human sperm, with relatively lower level in the acrosomal and post-acrosomal regions [1]. PLCζ exhibits the expected properties of a sperm-associated oocyte-activating factor in human [1,2]. Previous studies have demonstrated that PLCζ is the physiological agent responsible for inositol 1,4,5-trisphosphate pathway-mediated Ca2+ release in oocytes, a process known as oocyte activation [1,3]. A deficiency in the mechanism of oocyte activation is regarded as the principal cause of fertilization failure, or abnormally low fertilization after intracytoplasmic sperm injection (ICSI), and can occur in several cycle attempts [2]. Given the fundamental role of PLCζ in activating oocyte after gamete fusion, it was reported that reduced PLCζ protein level or mutated form of PLCζ are correlated with specific type of male infertility such as repetitive ICSI failure and globozoospermia [4]. Accordingly, numerous studies have reported assisted oocyte activation as a treatment for failed or low fertilization after ICSI [2,3,4].
In addition to its therapeutic role, PLCζ is thought to represent a prognostic biomarker of sperm quality. Several studies have demonstrated the differential expression of some key PLCζmRNAs in infertile males compare to fertile males [5,6]. In addition, a significant reduction in PLCζ mRNAs levels in individuals with low or failed fertilization with ICSI compared to fertile controls was reported [7].
DNA fragmentation is an important factor in the etiology of male infertility [8]. Men with high DNA fragmentation levels have significantly lower odds of conceiving, naturally or through procedures such as intrauterine insemination and in vitro fertilization [9]. The most common cause of DNA fragmentation in spermatozoa is reactive oxygen species and oxidative stress [10]. It is clinically important to investigate the sources of oxidative stress such as smoking, because, in most cases, they are modifiable.
Although PLCζ, DNA fragmentation and oxidation have been investigated a lot as a possible biomarker of sperm quality, there has been no study looking at the association among those three factors. In addition, most human data about PLCζ used PLCζ mRNAs and no studies have evaluated the clinical feasibility of sperm PLCζ level measured by immunostaining. Based on foregoing, we aim to investigate the feasibility and reproducibility of measuring PLCζ expression using immunostaining in human sperm. We also aimed to investigate the relationship between PLCζ immunoreactivity and DNA fragmentation or oxidation status in human sperm.
Semen samples were obtained from male participants between April 2013 and February 2014. Informed consent for enrollment into the study and for the use of semen in analysis was obtained from all participants. The institutional review board at Seoul National University Bundang Hospital reviewed and approved the study (B-1205-155-003). Subjects without a history of genital inflammation or genital surgery, subjective symptoms, self-reported medical risk factors and taking any prescription medications were included.
Semen samples were collected by masturbation after 3 days of sexual abstinence. After liquefaction for 30 minutes at room temperature, sperm quality was assessed using computer-assisted semen analysis (SAIS-PLUS 10.1, Medical Supply Co., Seoul, Korea) and classified according to World Health Organization guidelines published in 2010. Strict criteria for the definition of normal spermatozoa were used during morphological assessment. Baseline semen characteristics were as follows: volume, 2.9±1.4 mL (range, 1.0 to 6.0 mL), concentration, 99±82 million/mL (range, 18 to 460 million/mL), motility, 54.6±15.3% (range, 10.1% to 75.8%), total motile sperm, 177±238 million (range, 17 to 1,485 million); and normal forms by strict morphology, 11.3±5.6% (range, 2.3% to 25.0%). All semen samples contained motile sperm and no sample had significant numbers of round cells or leukocytospermia in accordance with World Health Organization guidelines (<1 million round cells/mL).
The semen was processed by the conventional swim-up method. After centrifuging the semen (300 xg for 5 minutes), a pellet was obtained via removal of seminal plasma. The pellet was suspended in fresh Ham's F10 (1.5 mL) supplemented with 10% serum substitute supplement (Irvine Scientific, Santa Ana, CA, USA). After centrifugation (300 xg for 5 minutes), the supernatant was discarded and Ham's F10 with 10% serum substitute supplement media (0.5 mL) was gently layered on the pellet and incubated at 37℃ in a 5% CO2 atmosphere for 1 hour. The supernatant (0.5 mL) was then transferred to a conical tube. Computer-assisted semen analysis and normal forms by strict morphology were assessed in the processed samples.
Nuclear DNA integrity was measured by the terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay as described previously [11]. The samples were smeared on a silane-coated slide (DAKO, Glostrup, Denmark) and air-dried. Sperm samples were fixed with 4% paraformaldehyde for 1 hour at 15℃ to 25℃, and then washed with phosphate-buffered saline (PBS). Sperm were permeabilized with 0.1% Triton X-100 in 0.1% sodium citrate (Sigma-Aldrich, St. Louis, MO, USA). A commercial apoptosis detection kit was used (In Situ Cell Death Detection Kit, Roche Diagnostics GmbH, Mannheim, Germany). The remaining procedures were performed as per manufacturer's instructions. Counterstaining was performed using a mounting medium with 4',6-diamidino-2-phenylindole (Vector Laboratories, Burlingame, CA, USA). Sperm with fragmented DNA had nuclei stained green, whereas the nuclei of other cells were blue. Sperm head with >50% of the area stained green was considered positive. At least 500 sperm were counted per experimental set and the percentage of sperm with fragmented DNA was determined as DNA fragmentation index (DFI).
This method was used for the detection of 8-hydroxy-2'-deoxyguanosine (8-OHdG), a known biomarker for oxidative stress. A specific antibody (Argutus Medical OxyDNA Test, BD Biosciences, Franklin Lakes, NJ, USA) conjugated to fluorescein isothiocyanate (FITC) was used. The intensity of FITC fluorescence was then assessed under fluorescent microscopic evaluation (Fig. 1). Briefly, sperm samples were fixed by 4% paraformaldehyde and permeabilized. The primary antibody was then added for 1 hour, according to the manufacturer's instructions. At least 500 sperm were counted in different areas of each slide. Sperm head with >50% of the area stained green was considered positive.
PLCζ was detected by immunofluorescent staining with a polyclonal anti-PLCζ antibody as described before [12]. Sperm samples were fixed with 4% paraformaldehyde/PBS, permeabilized with 0.5% (v/v) Triton X-100/PBS and stored at 4℃ until use. Sperm smears were created on pre-coated glass slides (Menzel-Gläser, Braunschweig, Germany), incubated in 3% bovine serum albumin-PBS, and labelled with a polyclonal anti-PLCζ antibody (25 µg/mL; Santa Cruz Biotechnology, Dallas, TX, USA) in 0.05% bovine serum albumin-PBS overnight at 4℃. After three washes with PBS, samples were labeled with Alexa Fluor 555 goat anti-rabbit IgG for 1 hour (1:400 dilution; Invitrogen, Merelbeke, Belgium), counterstained with 5 µg/mL Hoechst 33258 and 5 µg/mL FITC (Alexa 488, Invitrogen, Grand Island, NY, USA) for 15 minutes at 37℃ in the dark, washed with PBS and mounted in glycerol-DABCO. Sperm (n=400) were examined using a fluorescent microscopy (Axioskop40, Carl Zeiss Microscopy, Jena, Germany) (Fig. 2) [1]. Sperm head with >50% of the area stained green was considered positive regardless of the localization. PLCζ immunostaining was performed twice in each donor sample and the consistency was assessed by the Pearson's correlation test.
While several parameters were not normally distributed, the data are expressed as the mean±standard deviation. Nonparametric Spearman's correlation test was used to assess an association between different numerical parameters. Immunoreactivity of PLCζ was averaged from two measurements. A correlation between two measurements of PLCζ immunoreactivity was assessed by the Pearson's correlation test because this variable showed a normal distribution. Intergroup differences were assessed by nonparametric Wilcoxon test. A P-value of <0.05 (two-tailed) was considered statistically significant. Statistical analysis was performed using SPSS ver. 18.0 (SPSS Inc., Chicago, IL, USA).
Forty-four males were enrolled regardless of their fertility status. The mean age of the participants was 32.0±5.5 years (range, 23 to 49 years). Of those 44, 16 (36%) were smokers. While 47% (21/44) of the study participants were married, fertility was proven in only 5/21 married participants. The remaining 16 participants were not infertility patients.
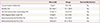
Table 1 shows the basic sperm characteristics of participant samples including DFI and immunoreactivity of 8-OHdG and PLCζ in swim-up samples. When duplicate PLCζ tests were performed on the same sperm samples from each of the 44 participants, similar results were obtained (74.1±9.4% vs. 75.4±9.7%). Two measurements of PLCζ immunoreactivity were found to be highly correlated with each other (r=0.759, P<0.001) (Fig. 3). Thus, the PLCζ test by immunostaining was highly consistent within the same sample. The mean intra-assay coefficient of the variation for PLCζ was estimated to be 3.4%.
In Table 2, the correlation coefficients among sperm parameters, immunoreactivity of PLCζ and 8-OHdG and DFI were presented. Immunoreactivity of PLCζ showed a negative relationship with 8-OHdG immunoreactivity (r=-0.404, P=0.009). Because 8-OHdG immunoreactivity was negatively correlated with sperm concentration, multivariate analysis was performed to eliminate the confounding effect of sperm concentration; as a result, immunoreactivity of PLCζ showed insignificantly negative correlation with 8-OHdG level (r=-0.317, P=0.056). Immunoreactivity of PLCζ was not associated with donor's age, sperm concentration, motility, and strict morphology as well as DFI.
When performed a subgroup analysis between smokers and non-smokers, no differences were found in the sperm parameters, immunoreactivity of PLCζ and 8-OHdG and DFI before or after swim-up between smokers and non-smokers (data not shown). In smokers, degree of cigarette exposure (pack-years) was not correlated with the sperm parameters including immunoreactivity of PLCζ and 8-OHdG and DFI before or after swim-up.
Semen analysis based on semen concentration, motility and morphology has been used for the diagnosis for male fertility for many years. However, a significant number of patients with normal sperm parameters still undergo difficulty in achieving successful pregnancy [13]. Several new diagnostic biomarkers for semen quality have been investigated recently including PLCζ, oxidative stress and DFI [14]. Here, we first demonstrated that sperm PLCζ immunoreactivity has a negative correlation with a oxidation marker, 8-OHdG.
While PLCζ has been investigated a lot as its role in oocyte activation was known, this study is the first to validate the clinical feasibility of measuring sperm PLCζ immunoreactivity in human sperm. Although the sample size was small, the high reproducibility of sperm PLCζ immunostaining in this study may suggest a possible clinical application of sperm PLCζ test in evaluating male infertility. The negative association between sperm PLCζimmunoreactivity and 8-OHdG immunoreactivity may be interpreted as a detrimental effect of oxidation on sperm-mediated oocyte activation which is consistent with previous studies. Morado et al. [14] reported that reactive oxygen species (ROS) levels differed between activated and non-activated human oocytes.
In our study a positive association was observed between 8-OHdG and DFI (r=0.216) but this was insignificant. Because oxidative stress is known to be a major causal factor of DNA fragmentation, it is now widely agreed that excess ROS contributes significantly to sperm DNA damage [15]. Previous studies reported that there is a significant positive relationship between sperm 8-OHdG levels and DFI [16,17]. The absence of the statistical significance might be attributed to small sample size in our study (i.e., weak power).
In our study, there was no difference in 8-OHdG immunoreactivity and DFI between smokers and non-smokers. This is consistent with a previous study reporting no association between smoking and DNA fragmentation in sperm of healthy men [18]. However, our result is discordant with two previous studies reporting a significantly higher amount of 8-OHdG in sperm in smokers than non-smokers [19,20]. In fact, 8-OHdG immunoreactivity in smokers (21.1±15.9) was higher than that of non-smokers (14.6±10.7), but this did not reach a statistical significance, thus further large-scaled study would be needed to clarify this topic.
The clinical strengths of this study can be summarized into two points. First, this is the first study to evaluate the laboratorial feasibility of measuring sperm PLCζ immunoreactivity which may propagate its use in the field of male infertility. Second, by looking at the possible association between sperm PLCζand 8-OHdG immunoreactivity, our result provides a significant pilot data for future lager studies to investigate further the efficacy of sperm PLCζ as a biomarker which can predict the semen quality and, in turn, assisted reproductive technique outcomes.
There are several limitations of this study. Because of the small number of participants, it was impossible to make population-based inferences based on our findings. Also, our study subjects were unselected population and subjects' fertility status could not be controlled. Finally, the effect of smoking was not evaluated thoroughly because of the small number of subjects.
In conclusion, sperm PLCζ can be measured using immunostaining and be possibly used as a semen quality marker in conjunction with other biomarkers such as semen analysis and 8-OHdG. Because of the relatively small number of subjects, however, a definitive judgment on the efficacy of sperm PLCζ measurement as a semen quality marker cannot yet be rendered. Further larger studies will be needed to determine whether sperm PLCζ has an impact on embryo development and ART pregnancy outcomes.
Figures and Tables
Fig. 1
A representative microphotograph showing sperm stained by immunofluorescent antibody for 8-hydroxy-2'-deoxyguanosine (×1,000); (A) 4,6-diamidino-2-phenylindole, (B) fluorescein isothiocyanate, and (C) merged capture.

Fig. 2
A representative microphotograph showing sperm stained by immunofluorescent antibody for anti-phospholipase C zeta (×400). (A) Hoechst 33258, (B) fluorescein isothiocyanate, and (C) merged capture. Four 'green' sperm were seen in (B). Non-specific binding in the tail has previously been reported by Grasa et al. [1].

Fig. 3
Two-times measurements of phospholipase C zeta immunoreactivity (round 1 and round 2) were highly correlated each other (r=0.759, P<0.001, by the Pearson correlation test).
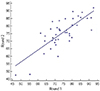
Table 1
Basic sperm parameters, DNA fragmentation index and immunoreactivity for 8-OHdG and PLCζ in human sperm after swim up
Acknowledgments
This work was supported by grant no. A120043 from the Korea Health Care Technology R&D Project, Ministry of Health and Welfare, Korea.
References
1. Grasa P, Coward K, Young C, Parrington J. The pattern of localization of the putative oocyte activation factor, phospholipase Czeta, in uncapacitated, capacitated, and ionophore-treated human spermatozoa. Hum Reprod. 2008; 23:2513–2522.
2. Heindryckx B, De Gheselle S, Gerris J, Dhont M, De Sutter P. Efficiency of assisted oocyte activation as a solution for failed intracytoplasmic sperm injection. Reprod Biomed Online. 2008; 17:662–668.
3. Nasr-Esfahani MH, Deemeh MR, Tavalaee M. Artificial oocyte activation and intracytoplasmic sperm injection. Fertil Steril. 2010; 94:520–526.
4. Heindryckx B, Van der Elst J, De Sutter P, Dhont M. Treatment option for sperm- or oocyte-related fertilization failure: assisted oocyte activation following diagnostic heterologous ICSI. Hum Reprod. 2005; 20:2237–2241.
5. Garrido N, Martinez-Conejero JA, Jauregui J, Horcajadas JA, Simon C, Remohi J, et al. Microarray analysis in sperm from fertile and infertile men without basic sperm analysis abnormalities reveals a significantly different transcriptome. Fertil Steril. 2009; 91:1307–1310.
6. Meseguer M, de los Santos MJ, Simon C, Pellicer A, Remohi J, Garrido N. Effect of sperm glutathione peroxidases 1 and 4 on embryo asymmetry and blastocyst quality in oocyte donation cycles. Fertil Steril. 2006; 86:1376–1385.
7. Aghajanpour S, Ghaedi K, Salamian A, Deemeh MR, Tavalaee M, Moshtaghian J, et al. Quantitative expression of phospholipase C zeta, as an index to assess fertilization potential of a semen sample. Hum Reprod. 2011; 26:2950–2956.
8. Venkatesh S, Singh A, Shamsi MB, Thilagavathi J, Kumar R, Mitra DK, et al. Clinical significance of sperm DNA damage threshold value in the assessment of male infertility. Reprod Sci. 2011; 18:1005–1013.
9. Zini A. Are sperm chromatin and DNA defects relevant in the clinic? Syst Biol Reprod Med. 2011; 57:78–85.
10. Aitken RJ, De Iuliis GN. On the possible origins of DNA damage in human spermatozoa. Mol Hum Reprod. 2010; 16:3–13.
11. Jee BC, Suh CS, Shin MS, Lee HJ, Lee JH, Kim SH. Sperm nuclear DNA fragmentation and chromatin structure in one-day-old ejaculated sperm. Clin Exp Reprod Med. 2011; 38:82–86.
12. Heytens E, Parrington J, Coward K, Young C, Lambrecht S, Yoon SY, et al. Reduced amounts and abnormal forms of phospholipase C zeta (PLCzeta) in spermatozoa from infertile men. Hum Reprod. 2009; 24:2417–2428.
13. Garrido N, Meseguer M, Alvarez J, Simon C, Pellicer A, Remohi J. Relationship among standard semen parameters, glutathione peroxidase/glutathione reductase activity, and mRNA expression and reduced glutathione content in ejaculated spermatozoa from fertile and infertile men. Fertil Steril. 2004; 82:Suppl 3. 1059–1066.
14. Morado S, Cetica P, Beconi M, Thompson JG, Dalvit G. Reactive oxygen species production and redox state in parthenogenetic and sperm-mediated bovine oocyte activation. Reproduction. 2013; 145:471–478.
15. Aitken RJ, De Iuliis GN, Finnie JM, Hedges A, McLachlan RI. Analysis of the relationships between oxidative stress, DNA damage and sperm vitality in a patient population: development of diagnostic criteria. Hum Reprod. 2010; 25:2415–2426.
16. Fraga CG, Motchnik PA, Shigenaga MK, Helbock HJ, Jacob RA, Ames BN. Ascorbic acid protects against endogenous oxidative DNA damage in human sperm. Proc Natl Acad Sci U S A. 1991; 88:11003–11006.
17. Kodama H, Yamaguchi R, Fukuda J, Kasai H, Tanaka T. Increased oxidative deoxyribonucleic acid damage in the spermatozoa of infertile male patients. Fertil Steril. 1997; 68:519–524.
18. Sergerie M, Ouhilal S, Bissonnette F, Brodeur J, Bleau G. Lack of association between smoking and DNA fragmentation in the spermatozoa of normal men. Hum Reprod. 2000; 15:1314–1321.
19. Shen HM, Chia SE, Ni ZY, New AL, Lee BL, Ong CN. Detection of oxidative DNA damage in human sperm and the association with cigarette smoking. Reprod Toxicol. 1997; 11:675–680.
20. Ji G, Yan L, Liu W, Qu J, Gu A. OGG1 Ser326Cys polymorphism interacts with cigarette smoking to increase oxidative DNA damage in human sperm and the risk of male infertility. Toxicol Lett. 2013; 218:144–149.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download