Abstract
Steroid cell tumors, not otherwise specified, are infrequently encountered ovarian neoplasms, which constitute <0.1% of all ovarian tumors. Most of these tumors are unilateral, and almost one-third of all cases are reportedly malignant. However, because most of these tumors are diagnosed in the early stage, and do not recur or metastasize, little is known about their response to therapies such as chemotherapy or radiation. Here, we present a rare case of recurrent steroid cell tumor, not otherwise specified that showed a complete response after debulking surgery, radiofrequency ablation, and adjuvant chemotherapy.
Ovarian steroid cell tumors are tumors derived from the stromal component of the ovary. Also known as sex-cord stromal tumors, these tumors constitute <0.1% of all ovarian tumors. The majority of these tumors are unilateral, benign, and characterized by the proliferation of steroid cells [1,2,3]. Steroid cell tumor, not otherwise specified (NOS) constitutes approximately 60% of all steroid cell tumors, with approximately one-third of NOS tumors reported as malignant [4].
Approximately half of all steroid cell tumors, NOS exhibit symptoms from excessive androgen secretion. Hirsutism and virilization are the most common primary symptoms, followed by anovulation, clitoromegaly, temporal hair loss, obesity, hypertension, impaired glucose tolerance, abdominal striae, and polycythemia. In approximately 8% of all cases, the increase in female hormones leads to symptoms such as abnormal uterine bleeding and sexual precocity among prepubescent females. Other symptoms exhibited due to this increase in female hormones include abdominal distension, abdominal pain, and on rare occasions, ascites [4,5].
When steroid cell tumors are accompanied by higher stage, large size, gross necrosis, and/or hemorrhage, the tumor generally exhibits higher malignant potential and worse prognosis. However, recurrence or metastasis rarely occurs, and anticancer treatment for these tumors is generally not required [4].
Here, we report on a rare case involving a patient who had undergone a total abdominal hysterectomy with bilateral salpingo-oophorectomy 5 years ago due to an ovarian steroid cell tumor, NOS. Five years later, the patient exhibited multiple peritoneal and hepatic recurrence, and debulking surgery and radiofrequency ablation of the liver metastases were performed to induce an optimal cytoreduction condition. Subsequently, adjuvant chemotherapy consisting of the bleomycin, etoposide, and cisplatin (BEP) regimen was administered to elicit a complete response.
A 51-year-old obese female patient with fatty liver underwent abdominal ultrasonography at her local clinic, and was admitted to our hospital due to liver masses, 25 and 15 mm in size. The patient had been on medication for diabetes and hypertension for 10 years. She had been admitted to the hospital 5 years ago with chief complaints of ascites and a 77-mm solid mass in the left ovary, which were confirmed by abdominopelvic computed tomography (CT). Under the clinical impression of ovarian cancer, total abdominal hysterectomy with bilateral salpingo-oophorectomy, omentectomy, and selective lymphadenectomy were performed. Postoperatively, the lesion was diagnosed as an ovarian steroid cell tumor, NOS.
Physical examinations did not yield any remarkable findings and the patient exhibited normal vital signs. Laboratory tests yielded normal findings of hemoglobin (13.3 g/dL), white blood cells (9,250/µL), CA-125 (3.73 IU/mL), and testosterone levels (0.074 ng/mL). Abdominopelvic CT revealed 24- and 10-mm arterial well-enhancing nodules at segment 5 and the left lateral segment of the liver, respectively. Moreover, approximately 10 to 30 mm multiple enlarged round masses were observed at the perigastric, gastrohepatic, and hepatoduodenal mesentery, and at the site of ligamentum teres fissure (Fig. 1A). In order to differentiate hepatocellular carcinoma and recurrent ovarian steroid cell tumor, NOS, we performed a sonography-guided liver biopsy. Immunohistochemically, the same findings were observed as in the specimen from 5 years ago, and accordingly, the specimen was diagnosed as a metastatic lesion.
We performed an exploratory laparotomy and noted approximately 30 metastatic masses, ranging from 5 to 30 mm in size, at the omentum, lesser sac, gastrosplenic ligament, and mesentery. In addition, numerous metastatic nodules, 1 to 2 mm in size, were observed. Subsequently, we performed multiple washing cytology and omentectomy and metastasectomy to induce an optimal cytoreduction state without residual lesions >5 mm in size. With respect to the liver metastasis, difficulties in resectability resulted in radiofrequency ablation being performed 5 days postsurgery. The patient recovered without any notable postoperative complications and was discharged from the hospital 9 days postoperation.
Postoperative histopathological analyses revealed tumor cells with abundant eosinophilic to clear cytoplasm. Moreover, immunohistochemical analyses revealed positive findings of inhibin-a and calretinin, confirming the diagnosis of recurrent ovarian steroid cell tumor, NOS (Fig. 2).
The patient was subjected to adjuvant chemotherapy 3 weeks postsurgery. The chemotherapy regimen administered to the patient consisted of bleomycin (20 units/m2 every 3 weeks×four cycles), etoposide (75 mg/m2 on days 1-5, every 3 weeks×four cycles), and cisplatin (20 mg/m2 on days 1-5, every 3 weeks×four cycles). The patient successfully completed the chemotherapy regimen without any notable complications, except grade III neutropenia.
The patient was examined every 3 months after the completion of chemotherapy. Upon abdominopelvic CT and positron emission tomography/CT performed at 9 months, the patient exhibited a complete response to the treatment, with no radiological findings of definitive noticeable nodules and discrete fluorodeoxyglucose uptake noted at the peritoneal cavity and liver (Fig. 1B, C). Currently , the patient is under follow-up without notable problems.
Although ovarian steroid cell tumors were formerly referred to as lipid cell tumors, Hayes and Scully [4] proposed that the tumors be termed steroid cell tumors, as 25% of the tumors do not contain intracellular lipids. Depending on the originating cell, the steroid cell tumors are categorized into three subtypes: stromal luteoma, Leydig cell tumor, and steroid cell tumor, NOS. Steroid cell tumors, NOS constitute approximately 60% of all steroid cell tumors. Steroid tumor cells typically occur in patients in their 40s and 50s, with the average age reported as 43 years. However, in rare occurrences, postmenopausal and prepubescent females are also diagnosed with steroid cell tumors. The sizes of the reported tumors range from 1 to 45 cm, with an average reported size of 8.4 cm [1,6,7,8]. In general, ovarian steroid cell tumors are benign and exhibit low differentiation. Interestingly, in cases where the tumors are pathologically benign but clinically malignant, 20% of patients present with intraperitoneal metastatic lesions, but rarely exhibit distant metastasis [9]. According to one report, 94% of all steroid cell tumors are unilateral and 28.6% of the tumors are malignant [4].
Steroid cell tumor, NOS typically exhibits clear boundaries with solid formation, and occasionally exhibits bands; they are morphologically polygonal and divided into eosinophilic and liquid cytoplasm. According to a report by Hayes and Scully [4], pathologic findings that suggest malignancy are observed in 92% of cases where ≥2 differentiations are observed under a high-powered microscope, 86% of cases where the tumor is accompanied by necrosis, 78% of cases where the size of the tumor is >7 cm, 77% of cases where the tumor is accompanied by hemorrhaging, and in 64% of cases where the tumor exhibits stage 2 to 3 dysplasia. However, most of the steroid cell tumors are screened in early stages and reports of recurrences or metastasis are rare.
In the diagnosis of steroid cell tumor, NOS, immunohistochemical staining of inhibin-a is generally useful. Inhibin-a not only exists in supporting and stromal cells, but also in granulosa cells and luteal cells of the ovary. Particularly, among the pituitary gonadotropins, it is inhibited by follicle stimulating hormone. Moreover, calretinin has recently been reported to be expressed by steroid cell tumors, NOS. Calretinin, a calcium binding protein, is originally found in nerve tissues, but also exists in the ovaries and testicles. According to a report by Deavers et al. [10], positive calretinin and inhibin-a expressions are observed in 60% to 90% and in 5% to 90% of all steroid cell tumors, NOS, respectively, and accordingly, in the present case, both inhibin-a and calretinin were positive upon immunohistochemistry.
Steroid cell tumors primarily secrete steroid hormones such as testosterone, estrogen, and cortisol. When patients are admitted with complaints of hirsutism and virilization, it is important to meticulously record their medical history, physical examination results, testosterone levels, and dehydroepiandrosterone sulfate levels. If the testosterone levels of a patient are >2.0 ng/mL, the dehydroepiandrosterone sulfate levels are normal, and there are no symptoms of 21-hydroxylase deficiency, the presence of ovarian androgen-secreting tumors should be considered [4,7]. In 6% to 23% of steroid cell tumors, estradiol is secreted, and in these patients, excessive menstruation and abnormal postmenopausal bleeding can be observed. In some cases, endometrioid adenocarcinoma has been reported to accompany steroid cell tumors [5].
No particular findings can lead to the diagnosis of steroid cell tumors, NOS with stand alone preoperative radiography, and transvaginal or abdominal ultrasound can only assist in the evaluation of ovarian tumors, while CT scans or magnetic resonance imaging (MRI) can evaluate the presence of both ovarian and adrenal tumors. According to a report by Wang et al. [11], MRI is the most appropriate choice for preoperative radiographical diagnosis. When gadolinium-diethylene-triamine-pentaacetic acid is injected and scanned, the presence of ovarian tumor and pelvic metastasis can be detected. As such, MRI was reported to aid in the preoperative diagnosis. Another diagnostic method for locating the primary site of the tumor consists of injecting gonadotropin-releasing hormones, and subsequently drawing blood samples from the ovarian and adrenal venous blood vessel area to measure the male hormone concentration [8,11].
As steroid cell tumors, NOS are very rare, the clinical outcomes of the tumors are not well understood. However, according to Hayes and Scully [4]'s report, among 63 diagnosed patients, only 6% cases were bilateral; and 88%, 6.8%, 12%, and 1.7% were stage 1, 2, 3, and 4 tumors, respectively. Among the 50 patients with complete follow-up information, 18 patients had malignant tumors. Despite the various chemotherapy and radiation treatments, 12 patients experienced tumor recurrence and 14 patients died, indicating that the prognosis of steroid cell tumors is poor if the tumor is late-stage, large, or recurring [4]. However, as steroid cell tumors, NOS are typically screened at an early stage, metastasis at the time of diagnosis is rare, and they are thus generally considered benign.
As the incidence of steroid cell tumor, NOS is very low, there are currently no established treatments, and the tumors are hence treated in a similar manner to stromal tumors, depending on a myriad of factors, including the surgical stage, histological classification, patient age, and history of childbirth. The primary treatment of steroid cell tumors is surgery. If there are no clinical findings of malignancy, surgery is sufficient for a complete response. In the case of advanced tumors, low differentiation, or dysplasia, the use of postoperative adjuvant chemotherapy should be considered [4,12]. However, as the tumors are typically screened at an early stage and as metastasis or recurrence rarely occurs, there are no established guidelines regarding the optimal postoperative adjuvant therapy.
The majority of steroid cell tumors, NOS are benign, and even in cases of malignancy, recurrences are rare. Thus, there are currently no established treatment methods for recurrent cases either. The patient described here in exhibited a complete response to optimal cytoreductive surgery and postoperative adjuvant chemotherapy using the BEP regimen for the treatment of a recurring steroid cell tumor, NOS that had been initially diagnosed 5 years earlier. But, since this study includes only one case with short term follow-up result, there is a limitation to propose a reference for the adjuvant chemotherapy regimen for steroid cell tumors. Nevertheless, we experienced a complete response after debulking surgery, radiofrequency ablation, and adjuvant chemotherapy of a recurrent steroid cell tumor, we report this case with a brief review of literature.
Figures and Tables
Fig. 1
(A) Preoperative abdominopelvic computed tomography shows 24-mm arterial well-enhancing mass at segment 5 of liver and 30-mm well-enhancing omental mass at paracolic gutter space. (B,C) Abdominopelvic computed tomography and positron emission tomography/computed tomography after the completion of treatment shows no definitive noticeable nodules and discrete fluorodeoxyglucose uptake noted at the peritoneal cavity and liver.
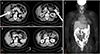
Fig. 2
(A) Steroid cell tumor, not otherwise specified composed of cells with abundant eosinophilic to clear cytoplasm. The cells have an appearance similar to adrenal cortical cells (H&E, ×200). (B) Diffuse nuclear and cytoplasmic staining for inhibin-a in steroid cell tumor (×200). (C) Diffuse nuclear and cytoplasmic staining for calretinin in steroid cell tumor (×200).

References
1. Young RH, Shully RE. Steroid cell tumors of the ovary. In : Fox H, Wells M, editors. Obstetrical and gynecological pathology. Edinburgh: Churchill Livingstone;2003. p. 845–856.
2. Reedy MB, Richards WE, Ueland F, Uy K, Lee EY, Bryant C, et al. Ovarian steroid cell tumors, not otherwise specified: a case report and literature review. Gynecol Oncol. 1999; 75:293–297.
3. Wang PH, Chao HT, Lee WL. Use of a long-acting gonadotropin-releasing hormone agonist for treatment of steroid cell tumors of the ovary. Fertil Steril. 1998; 69:353–355.
4. Hayes MC, Scully RE. Ovarian steroid cell tumors (not otherwise specified): a clinicopathological analysis of 63 cases. Am J Surg Pathol. 1987; 11:835–845.
5. Taylor HB, Norris HJ. Lipid cell tumors of the ovary. Cancer. 1967; 20:1953–1962.
6. Harris AC, Wakely PE Jr, Kaplowitz PB, Lovinger RD. Steroid cell tumor of the ovary in a child. Arch Pathol Lab Med. 1991; 115:150–154.
7. Berek JS, Rinehart RD, Adams Hillard PJ, Adashi EY. Endocrine disorders. In : Novak E, Berek JS, editors. Novak's gynecology. 13th ed. Philadelphia: Lippincott Williams & Wilkins;2004. p. 890–895.
8. Wang PH, Chao HT, Lee RC, Lai CR, Lee WL, Kwok CF, et al. Steroid cell tumors of the ovary: clinical, ultrasonic, and MRI diagnosis: a case report. Eur J Radiol. 1998; 26:269–273.
9. Clement PB, Young RH. Atlas of gynecologic surgical pathology. Philadelphia: WB Saunders;2000.
10. Deavers MT, Malpica A, Ordonez NG, Silva EG. Ovarian steroid cell tumors: an immunohistochemical study including a comparison of calretinin with inhibin. Int J Gynecol Pathol. 2003; 22:162–167.
11. Wang PH, Chao HT, Liu RS, Cho YH, Ng HT, Yuan CC. Diagnosis and localization of testosterone-producing ovarian tumors: imaging or biochemical evaluation. Gynecol Oncol. 2001; 83:596–598.
12. Gershenson DM, Copeland LJ, Kavanagh JJ, Stringer CA, Saul PB, Wharton JT. Treatment of metastatic stromal tumors of the ovary with cisplatin, doxorubicin, and cyclophosphamide. Obstet Gynecol. 1987; 70:765–769.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download