Abstract
Objective
The aim of this study was to investigate the anti-proliferative effect of the salinomycin in cell proliferation and apoptosis in primary cultured human uterine leiomyoma cells.
Methods
Cell viability was measured by MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay. Caspase-3 activity assay and DNA fragmentation assay were performed to determine the effect of apoptosis. The expression of apoptosis regulatory-related proteins was evaluated by western blot.
Results
The cell viability and proliferation of uterine leiomyoma cells were significantly reduced by salinomycin treatment in a dose-dependent manner. DNA fragmentation assay results showed apoptotic cell death after salinomycin incubation. Salinomycin activated caspase-3, -8, and -9, causing apoptosis in uterine leiomyoma cells. Down-regulation of Bcl-2, XIAP, and FLIP with a concomitant increase in Bax, Fas, and DR5 were observed.
A uterine myoma (myoma uteri) is a solid tumor made of fibrous tissue, often called a 'fibroid' tumor. Myomas vary in size and number, and are mostly slow-growing with no symptoms. Approximately 25% of myomas will cause symptoms and need medical treatment [1]. Myomas may grow as a single nodule or in clusters and range in size from 1 mm to over 20 cm in diameter. Myomas are the most frequently diagnosed tumor of the female pelvis and the most common reason for a hysterectomy. Although they are often referred to as tumors, they are not cancerous [2]. Treatment of leiomyoma has mainly been surgical resection, although some medical therapies are available for women who want to preserve uterus. Currently, management of leiomyoma relies on reducing the circulating levels of ovarian hormones by using gonadotropin-releasing hormone analogues, such as goserelin or leuprorelin acetate [3]. However, the use of these drugs is approved only for short-term therapy because they can result in loss of bone mass and cardiovascular changes [4,5]. Therefore, there is a need for safer and more effective non-surgical treatments for women suffering from uterine leiomyoma.
Salinomycin was originally used as anti-coccidial drug to kill bacteria, fungi, parasites and for efficient nutrient absorption in piggery [6,7,8,9]. It is a monocarboxylic polyether antibiotic isolated from Streptomyces albus strain and acts as an ionophore [10]. As an ionophore with strict selection for alkali ions and a strong preference for potassium, it interferes with transmembrane potassium potential. Recently, in an automated high-throughput screening test, salinomycin was found to be the most effective agent against cancer stem cells. Among the 16,000 small molecule chemicals studied, salinomycin was 100 times more effective than paclitaxel [11]. In addition, salinomycin induced apoptosis in many human cancer cells, but not in normal cells, by multiple mechanisms [12].
Apoptosis is a programmed cell death, a distinct process from necrosis. It involves a series of biochemical events that lead to a characteristic cell morphology which includes membrane blebbing, cell shrinkage, chromatin condensation, apoptotic body formation, DNA fragmentation, and eventual cell death [13]. There are two pathways that initiate apoptosis; the intrinsic signaling pathway associates non-receptor-mediated interactions that produce intracellular signals that directly affect targets within leiomyoma cells. The extrinsic signaling pathway involves transmembrane receptor-mediated interactions that involve death receptors in leiomyoma [14].
This study assessed the potential role of salinomycin as a novel chemotherapeutic agent in human uterine leiomyoma cells. To date, no study has been conducted in the evaluation of the growth inhibition effect of salinomycin on human uterine leiomyoma. The author therefore hypothesized that salinomycin would be useful in treating human uterine leiomyoma cells. In this study, the effects of salinomycin on apoptosis are investigated via intrinsic and extrinsic pathways on survival signaling pathways in human uterine leiomyoma cells.
Tissue was washed twice in cold phosphate buffered saline before being minced into 5 mm pieces in a sterile culture dish. The minced pieces were transferred into 50 mL conical tubes containing Hanks' balanced salts (Sigma-Aldrich, St Louis, MO, USA), supplemented with 25 mM HEPES, 100 units/mL antibiotics, 1.5 mg/mL collagenase IV (Sigma-Aldrich) and 0.2 mg/mL of DNase I (Roche Diagnostics, Mannheim, Germany). All the tubes were kept at 37℃ in a water bath with gentle agitation for 3 hours. Undigested tissue was filtered and the cells were centrifuged at 1,000 rpm for 5 minutes. The pellet was rinsed once with Hanks' balanced salts and dispersed in complete medium composed of DMEM/F12 with 10% fetal bovine serum, 1% penicillin/streptomycin. The culture medium was changed every two days.
The number of viable cells was evaluated by colorimetric 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Initially, cells were seeded at a density of 1×105 cells/mL in 24-well plate, and then cultured to allow their adhesion to the plate. After this pre-incubation, the culture medium was changed to experimental medium supplemented with salinomycin 1, 5, 10, and 20 µM or DMSO (control) for 24 hours. MTT reagent was added and incubated for an additional 3 hours at 37℃. The intensity of the purple color formed in this assay is proportional to the number of viable cells. Optical density (OD) was measured at 540 nm. Cell survival was calculated by subtracting the background OD of media alone and then normalized by dividing the OD of test wells by the OD of control (untreated) wells. The mean value and their standard deviation were calculated from triplicate experiments.
Salinomycin treated cells were harvested in 1×RIPA buffer (Thermo Scientific, Rockford, IL, USA) containing protease and phosphatase inhibitors with EDTA (Thermo Scientific). Protein concentrations were measured using Protein Assay Reagent (Bio-Rad, Hercules, CA, USA) following the manufacturer's protocol. Aliquots of protein were separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred to nitrocellulose membrane by Semi-Dry Transfer Cell (Bio-Rad). The membrane was blocked with Tris-buffered saline containing 5% skim milk. After washing, the membranes were incubated with primary antibodies to Akt, pAkt, cleaved caspase-3, cleaved caspase-8, cleaved caspase-9 and c-PARP (Cell Signaling, Beverly, MA, USA), Bcl-2, Fas, and DR4 (Santa Cruz Biotechnology, Santa Cruz, CA, USA), DR5 (Koma Biotech, Seoul, Korea), Bax and XIAP (BD Pharmingen, Franklin Lakes, NJ, USA), FLICE-like inhibitory protein (c-FLIP; Enzo Life Science International, Farmingdale, NY, USA) and β-actin (Santa Cruz Biotechnology). After reaction with horseradish peroxidase-conjugated secondary antibodies (Santa Cruz Biotechnology), bands on the membranes were visualized by an enhanced chemiluminescence (ECL) system (Thermo Scientific). The density of respective bands was analyzed by the Chemi-doc XRS imaging system (Bio-Rad).
After drug treatment, cells were centrifuged at 2,000 rpm for 5 minutes to harvest. Cells (5×106) were then lysed in 50 µL of lysis buffer on ice for 30 minutes and centrifuged at 14,000 rpm for 5 minutes at 4℃ and the supernatant was collected. Supernatant (50 µL) was added to an equal volume of 2×reaction/DTT Mix supplemented with DEVD-pNA (50 µM) and incubated at 37℃ for 2 hours. Caspase-3 activity was measured as the absorbance at 405 nm of the cleaved substrate pNA followed by ApoAlert Caspase Colorimetric Assay kits user manual (Clontech Laboratories, Palo Alto, CA, USA).
Fragmented nucleosomal DNA was quantified by cell death detection enzyme-linked immunosorbent assay (ELISA) plus kit (Roche Diagnostics) as described in the manufacturer's manual. Briefly, cells (4×105) were plated in 6 well plates in the presence of 10% fetal bovine serum with antibiotics (streptomycin and penicillin). The cells were exposed to 0, 5, 10 µM salinomycin for 24 hours. After careful removal of media, cells were pooled and lysed with 200 µL of lysis buffer for 30 minutes at room temperature. Lysates were transferred to 1.5 mL eppendorf tubes and centrifuged at 13,000 rpm for 10 minutes to obtain low molecular weight DNA from apoptotic cells. Supernatant (20 µL) was used to detect apoptosis with microplate reader (Tecan Sunrise, Durham, NC, USA) at 405 nm. Background values (incubation buffer alone) were subtracted and OD values representing nucleosomal DNA fragments in salinomycin treated samples were compared with those values obtained from control cells and expressed as percentage (%) of control.
Author used an MTT assay to analyze the effect of salinomycin on the growth of uterine leiomyoma cells. As shown in Fig. 1, in contrast with the control, cells treated with salinomycin showed decreased cell growth in a dose-dependent manner. The leiomyoma cell growth was decreased by about 95%, 73%, 62%, and 30% in the treatment with salinomycin 1, 5, 10, and 20 µM, respectively in contrast with the untreated control (Fig. 1).
The induction of apoptotic cell death could be in part due to inactivation of important survival genes and therefore the expression of molecules transcriptionally regulated by Akt was also evaluated. The expression of pAkt proteins were decreased upon salinomycin treatment in uterine leiomyoma cells. Next, we investigated whether apoptosis induced by salinomycin occurs via intrinsic mitochondrial apoptosis pathway in uterine leiomyoma cells. For that purpose, we measured the levels of Bcl-2 family proteins (Bcl-2 and Bax) and XIAP. There was no change in the expression of Bax but the antiapoptotic Bcl-2 protein and XIAP found decreased. The caspase-3 and PARP protein cleavage were increased after treatment (Fig. 2). These results suggest that salinomycin induced cell growth inhibition and induction of apoptotic cell death results in part from the inhibition of survival factors, and inactivation of Akt pathway.
In this step, we investigated whether salinomycin activated the extrinsic apoptosis pathway by measuring the expression of death receptor signaling-related proteins, such as DR4, DR5, Fas and caspase-8. The expression of DR5, Fas and cleaved caspase-8 proteins were significantly increased. In addition, we observed down-regulation of c-FLIP expression. There was no change in the expression of DR4 (Fig. 3). These results suggest that salinomycin-induced apoptotic cell death involve the activation of DR and Fas mediated pathway.
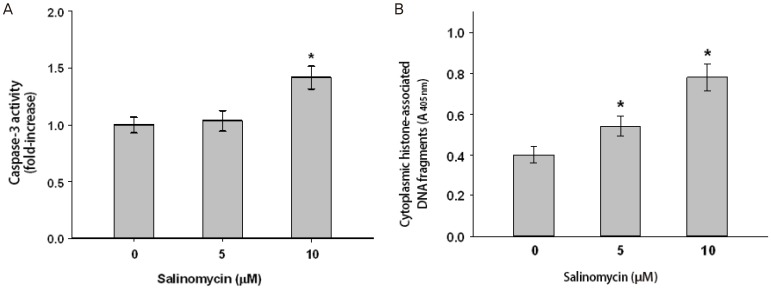
The apoptosis assay was performed to evaluate the mechanism of the inhibition of using uterine leiomyoma cell growth using csapase-3 activity test and cell death detection kit. Activity of caspase-3 was increased following salinomycin treatment (Fig. 4A). ELISA showed significant increase of histone associated DNA fragments after 24 hours exposure to salinomycin (Fig. 4B).
The present study demonstrated that salinomycin inhibited the growth of human uterine leiomyoma cells. The growth-inhibition effects of salinomycin and salinomycin-induced apoptosis in uterine leiomyoma cell was associated with Akt-mediated cell death pathway.
Anticancer effects of salinomycin have been established in variety of preclinical studies, using many different cancer cell lines, including breast cancer [11], colorectal cancer [15], prostate cancer [16], ovarian cancer [17], and chronic lymphocytic leukemia [18]. To date, this study successfully exhibited, the anti-proliferative effects of salinomycin on the uterine leiomyoma cells for the first time.
Akt regulates essential cellular functions such as migration, proliferation, differentiation, apoptosis, and metabolism. Activated Akt (p-Akt) regulates many downstream effectors such as c-FLIP, HIF-1α, and NF-κB. FLIP is a caspase-8 homologue that inhibits the death receptor pathway of apoptosis. The expression of c-FLIP has been shown to be regulated by Akt pathway in several solid tumors [19]. In this study, the Akt phosphorylation and expression of c-FLIP in human uterine leiomyoma cells were decreased after exposure to salinomycin.
This study demonstrated that salinomycin induced apoptosis through both intrinsic and extrinsic pathways in uterine leiomyoma cells. The intrinsic pathway is called the mitochondria-dependent pathway because it is induced by mitochondrial dysfunction and mitochondrial membrane potential dissipation [20]. In the present study, salinomycin reduced the expression of anti-apoptotic proteins (Bcl-2 and XIAP) and increased the pro-apptotic proteins (Bax) in the uterine leiomyoma cells. All of these results suggest that the salinomycin-induced apoptosis effect occurs via the intrinsic pathway and is regulated by Bcl-2 family proteins. This finding is consistent with a previous report which found that salinomycin downregulated the expression of Bcl-2 mRNA in endometrial cancer cells [21].
The extrinsic pathway is mediated by the death receptor located on the cell membrane [22]. DR4, DR5 and Fas are a death receptor involved in the extrinsic pathway. Although salinomycin treatment did not increase DR4 expression, DR5 and Fas expression levels were significantly enhanced, and caspase-8 was cleaved in salinomycin-treated uterine leiomyoma cells (Fig. 3), thereby suggesting that salinomycin also activates the extrinsic pathway. Salinomycin activated caspase-3, -8, and -9, causing apoptosis in uterine leiomyoma cells. Caspase-9 is activated through the intrinsic pathway, which can be initiated by DNA damage and several extracellular stresses [23]. In contrast, caspase-8 is activated through the extrinsic pathway triggered by DR4, DR5 and Fas receptor [24]. Both pathways activate caspase-3, finally causing PARP cleavage and apoptosis.
The investigation of salinomycin influence on apoptosis and apoptosis associated protein expression suggests that salinomycin could be useful in treatment of human uterine leiomyoma cells. This result provided the first evidence that salinomycin induces both intrinsic and extrinsic apoptosis. These data may provide a basis for salinomycin treatment of patients with uterine leiomyoma.
References
1. Buttram VC Jr, Reiter RC. Uterine leiomyomata: etiology, symptomatology, and management. Fertil Steril. 1981; 36:433–445. PMID: 7026295.
2. Velebil P, Wingo PA, Xia Z, Wilcox LS, Peterson HB. Rate of hospitalization for gynecologic disorders among reproductive-age women in the United States. Obstet Gynecol. 1995; 86:764–769. PMID: 7566845.

3. Corbin A, Beattie CW. Ihibition of the pre-ovulatory proestrous gonadotropin surge, ovulation and pregnancy with a peptide analogue of luteinizing hormone releasing hormone. Endocr Res Commun. 1975; 2:1–23. PMID: 1097236.
4. Johansen JS, Riis BJ, Hassager C, Moen M, Jacobson J, Christiansen C. The effect of a gonadotropin-releasing hormone agonist analog (nafarelin) on bone metabolism. J Clin Endocrinol Metab. 1988; 67:701–706. PMID: 2971080.

5. Friedman AJ, Harrison-Atlas D, Barbieri RL, Benacerraf B, Gleason R, Schiff I. A randomized, placebo-controlled, double-blind study evaluating the efficacy of leuprolide acetate depot in the treatment of uterine leiomyomata. Fertil Steril. 1989; 51:251–256. PMID: 2492232.
6. Miyazaki Y, Shibuya M, Sugawara H, Kawaguchi O, Hirsoe C. Salinomycin, a new polyether antibiotic. J Antibiot (Tokyo). 1974; 27:814–821. PMID: 4452657.

7. Mahmoudi N, de Julian-Ortiz JV, Ciceron L, Galvez J, Mazier D, Danis M, et al. Identification of new antimalarial drugs by linear discriminant analysis and topological virtual screening. J Antimicrob Chemother. 2006; 57:489–497. PMID: 16415127.

8. Danforth HD, Ruff MD, Reid WM, Miller RL. Anticoccidial activity of salinomycin in battery raised broiler chickens. Poult Sci. 1977; 56:926–932. PMID: 605065.

9. Naujokat C, Fuchs D, Opelz G. Salinomycin in cancer: a new mission for an old agent. Mol Med Rep. 2010; 3:555–559. PMID: 21472278.

10. Mitani M, Yamanishi T, Miyazaki Y. Salinomycin: a new monovalent cation ionophore. Biochem Biophys Res Commun. 1975; 66:1231–1236. PMID: 1191289.

11. Gupta PB, Onder TT, Jiang G, Tao K, Kuperwasser C, Weinberg RA, et al. Identification of selective inhibitors of cancer stem cells by high-throughput screening. Cell. 2009; 138:645–659. PMID: 19682730.

12. Fuchs D, Heinold A, Opelz G, Daniel V, Naujokat C. Salinomycin induces apoptosis and overcomes apoptosis resistance in human cancer cells. Biochem Biophys Res Commun. 2009; 390:743–749. PMID: 19835841.

14. Elmore S. Apoptosis: a review of programmed cell death. Toxicol Pathol. 2007; 35:495–516. PMID: 17562483.

15. Dong TT, Zhou HM, Wang LL, Feng B, Lv B, Zheng MH. Salinomycin selectively targets 'CD133+' cell subpopulations and decreases malignant traits in colorectal cancer lines. Ann Surg Oncol. 2011; 18:1797–1804. PMID: 21267784.

16. Kim KY, Yu SN, Lee SY, Chun SS, Choi YL, Park YM, et al. Salinomycin-induced apoptosis of human prostate cancer cells due to accumulated reactive oxygen species and mitochondrial membrane depolarization. Biochem Biophys Res Commun. 2011; 413:80–86. PMID: 21871443.

17. Parajuli B, Lee HG, Kwon SH, Cha SD, Shin SJ, Lee GH, et al. Salinomycin inhibits Akt/NF-κB and induces apoptosis in cisplatin resistant ovarian cancer cells. Cancer Epidemiol. 2013; 37:512–517. PMID: 23545383.

18. Lu D, Choi MY, Yu J, Castro JE, Kipps TJ, Carson DA. Salinomycin inhibits Wnt signaling and selectively induces apoptosis in chronic lymphocytic leukemia cells. Proc Natl Acad Sci U S A. 2011; 108:13253–13257. PMID: 21788521.

19. Panka DJ, Mano T, Suhara T, Walsh K, Mier JW. Phosphatidylinositol 3-kinase/Akt activity regulates c-FLIP expression in tumor cells. J Biol Chem. 2001; 276:6893–6896. PMID: 11145953.

20. Bucur O, Ray S, Bucur MC, Almasan A. APO2 ligand/tumor necrosis factor-related apoptosis-inducing ligand in prostate cancer therapy. Front Biosci. 2006; 11:1549–1568. PMID: 16368536.

21. Kusunoki S, Kato K, Tabu K, Inagaki T, Okabe H, Kaneda H, et al. The inhibitory effect of salinomycin on the proliferation, migration and invasion of human endometrial cancer stem-like cells. Gynecol Oncol. 2013; 129:598–605. PMID: 23500085.

22. Fulda S, Debatin KM. Resveratrol-mediated sensitisation to TRAIL-induced apoptosis depends on death receptor and mitochondrial signalling. Eur J Cancer. 2005; 41:786–798. PMID: 15763656.

23. Wallach D, Varfolomeev EE, Malinin NL, Goltsev YV, Kovalenko AV, Boldin MP. Tumor necrosis factor receptor and Fas signaling mechanisms. Annu Rev Immunol. 1999; 17:331–367. PMID: 10358762.

Fig. 1
Anti-proliferative effect of salinomycin in uterine leiomyoma cells. (A) Cell morphologic changes were observed by phase contrast microscopy. Cells were treated with DMSO (control) or salinomycin (1, 5, 10, and 20 µM) for 24 hours. (B) Cell viability was measured using MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay. Values are ±SD of three measurements *P<0.05.
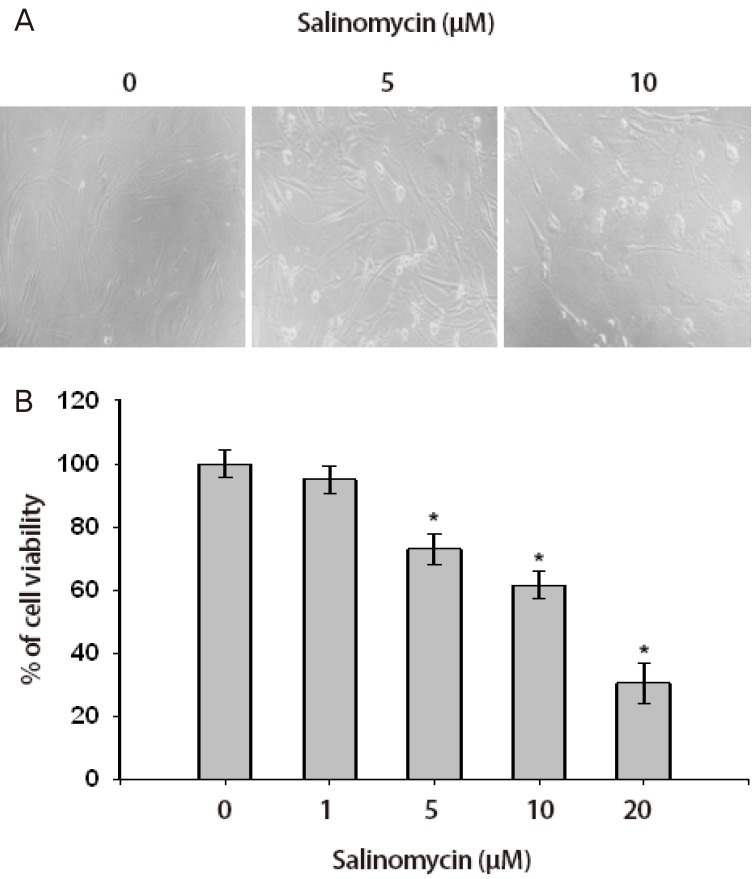
Fig. 2
Effect of salinomycin on intrinsic apoptotic pathway related protein expression in uterine leiomyoma cells. Twenty-four hours treated cell extracts were prepared and subjected for immunoblotting analysis. Beta-actin was used as an internal loading control.
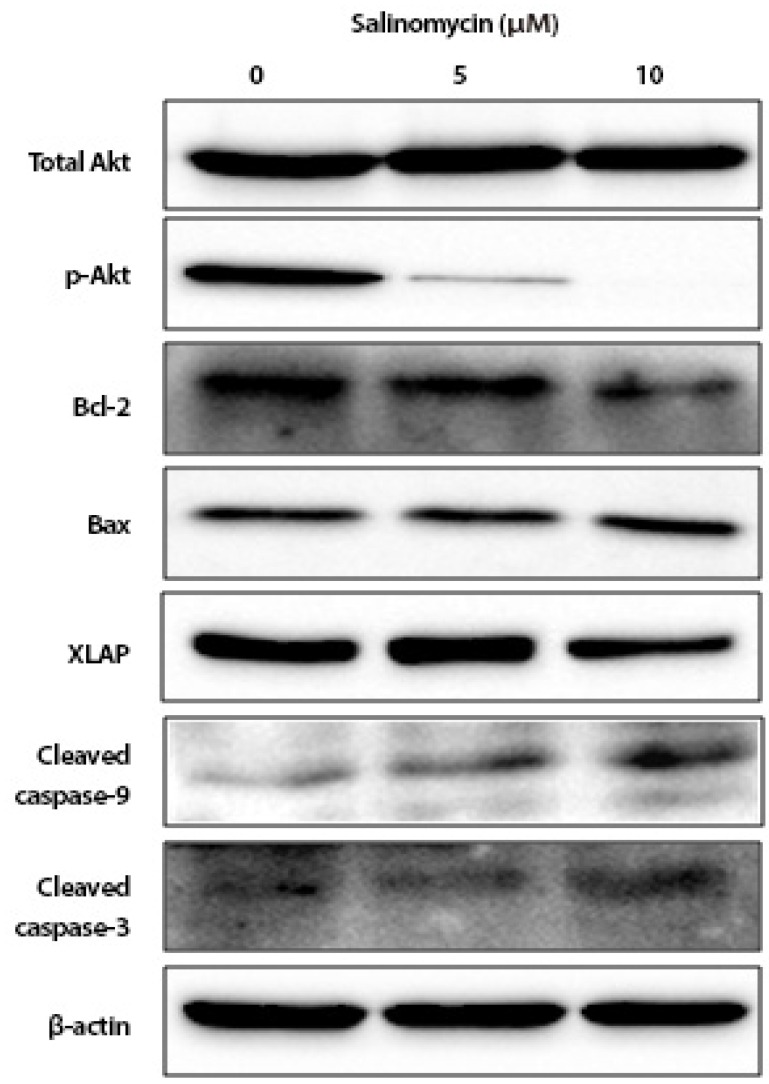
Fig. 3
Effect of salinomycin on extrinsic apoptotic pathway related protein expression in uterine leiomyoma cells. Twenty-four hours treated cell extracts were prepared and subjected for immunoblotting analysis. Beta-actin was used as an internal loading control.
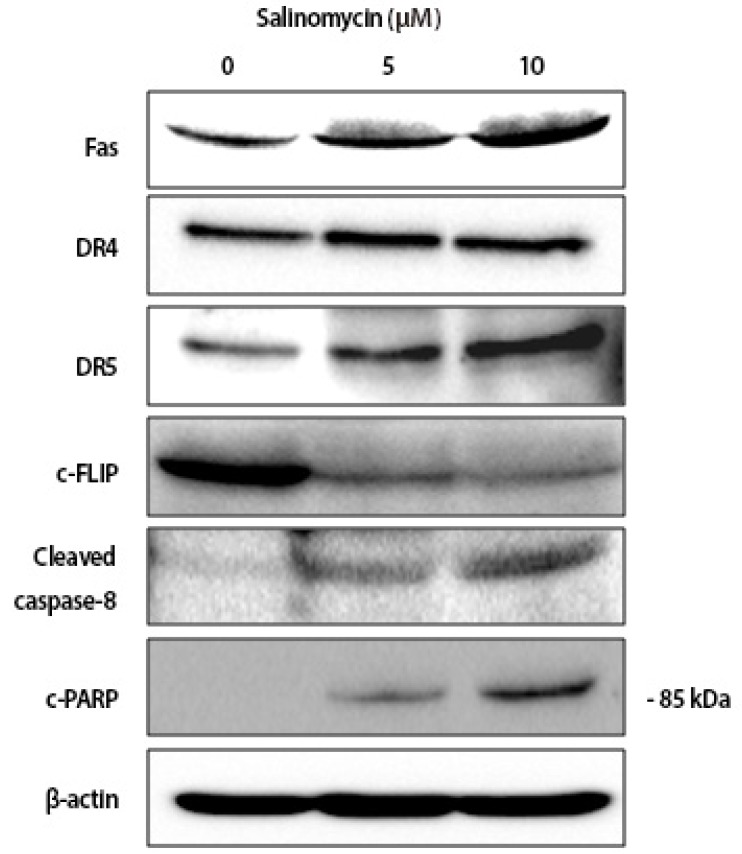
Fig. 4
Induction of apoptosis after salinomycin treatment in uterine leiomyoma cells. As describe in methodology section cells were exposed to DMSO (control) or salinomycin (5 and 10 µM) for 24 hours. Apoptosis was quantified by (A) caspase-3 activity and (B) enzyme-linked immunosorbent assay. Values are ±SD of three measurements *P<0.05.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download