Abstract
Uterine arteriovenous malformation (AVM) is rare but potentially life-threatening from excessive vaginal bleeding. All uterine AVMs reported to date have been found in the endometrial or myometrial layers. Here we present a patient with a subserosal type AVM on the fundus of uterus, which spontaneously ruptured.
Uterine arteriovenous malformation (AVM) is a rare but potentially life-threatening disorder [1-3]. AVM, an abnormal connection between artery and vein, can result in profuse vaginal bleeding. Whenever there is unexpected, excessive vaginal bleeding after a first trimester pregnancy loss, dilatation and curettage, or Cesarean section, uterine AVM must be considered [4-6]. Currently, the gold standard for diagnosis of uterine AVM is investigation by angiography. However, less invasive modalities such as color and spectral flow Doppler ultrasound are increasingly used for diagnosis [7].
A 24-year-old woman, gravida 1 para 0, was referred to our hospital with vaginal bleeding for a week. She had a dilatation and curettage due to a missed abortion at a local hospital two months ago. After the procedure, she received an ultrasound, and heard that there were no specific findings in uterus. She had normal menstrual cycles, and no other medico-surgical history.
On admission, she had moderate vaginal bleeding. Transvaginal gray and color Doppler ultrasound examination demonstrated abnormal findings of increased high-velocity blood flows within the posterior myometrium (Fig. 1A, B), and anechoic cystic lesion with high velocity vascular flow on the fundus of the uterus (Fig. 1C, D). There was no communication between the subserosal AVM and endometrial cavity. An abdominal aortogram confirmed enlarged bilateral uterine arteries and subserosal AVM (Fig. 1E). The AVM was additionally supplied by left ovarian artery (Fig. 1F). Embolization of the bilateral uterine arteries and left ovarian artery was performed as treatment of choice to spare fertility. After embolization, there was no significant flow to the subserosal type AVM (Fig. 1G). The patient tolerated the procedure well, and was discharged without problem.
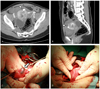
One month later, the patient presented to the emergency room with severe abdominal pain and distension without vaginal bleeding. On admission, her blood pressure was 95/60 mm Hg, and pulse rate was 115 beat per minute. Hemoglobin was 9.5 g/dL. Abdominopelvic computed tomography scan demonstrated that the fundal AVM of uterus was ruptured, and there was a large amount of hemoperitoneum in the abdominopelvic cavity (Fig. 2A, B). By emergency exploratory laparotomy, about 3×3 cm sized subserosal type AVM was noted on the fundus that had ruptured and was bleeding profusely (Fig. 2C, D). The ruptured AVM was removed, and successfully repaired. The hematoma in the abdominopelvic cavity was removed. After surgery, the patient recovered well, and was discharged at hospital day 6 without complications. Five weeks later the external surface and internal area of the uterus appeared normal on ultrasonic examination.
AVMs are abnormal communications between arteries and veins in which the usual intervening capillaries are absent. Though the true incidence of acquired uterine AVM is unknown due to its rarity, it can be a life-threatening cause of vaginal bleeding. Prompt diagnosis and intervention are required to prevent significant morbidity and mortality.
There are several published case reports and reviews for uterine AVM. Recently, Peitsidis et al. [10] reviewed about 91 studies including 85 case reports, and 6 observational studies for uterine AVM. The mean age of the women with uterine AVM was 30 ± 9.1 years, and 96% of the women were premenopausal. Most AVM cases were associated with pregnancy, vaginal bleeding, and dilatation and curettage. In all cases, uterine AVMs were found in the myometrium or endometrium. To our knowledge, this is the first case report of subserosal type uterine AVM, which has a potential risk of spontaneous rupture.
Ultrasound with color Doppler is a good choice for the initial diagnostic tool. In this rare case, ultrasound with color Doppler was helpful to diagnose. AVM and hemangioma have something in common. They are associated with pregnancy, and their main clinical symptom is vaginal bleeding. Localized abnormal vascular spaces in the myometrium and endometrium have been reported in AVM and hemangioma. However, unlike AVM, hemangioma is a kind of venous malformation, and usually show no blood flow by Doppler [11,12].
Because most uterine AVMs manifest in women between the age of 20 and 40 years (childbearing age), and are accompanied by excessive vaginal bleeding, it is difficult to decide on treatment [1]. Uterine artery embolization is now a commonly performed treatment, and considered first-line therapy in patients who wish to preserve fertility [13]. Although embolization for the AVM was performed appropriately in this case, the risk of spontaneous rupture seems to have remained. For successful removal of AVM, preoperative embolization can also be performed to minimize intraoperative blood loss in patients who desire fertility spared.
We do not know any of patients with subserosal type uterine AVM in the literature, as reported here. Because this type of uterine AVM has a risk of spontaneous rupture, closer observation is required. We recommend that subserosal type uterine AVM is best treated by surgical removal.
Figures and Tables
Fig. 1
Sagittal transvaginal gray-scale and color Doppler ultrasonographic images and pelvic angiography. (A) Multiple serpentine anechoic myometrial structures are showed by gray-scale image. (B) Multidirectional high-velocity vascular flow is demonstrated by color Doppler ultrasonographic scan. (C, D) About 1.7×1.7 cm sized anechoic round lesion with high velocity vascular flow (arrowhead) is noted on fundal area of uterus. (E) Abdominal aortogram shows enlarged bilateral uterine arteries and fundal arteriovenous malformation (AVM) (arrow). (F) Left ovarian angiography demonstrates additional supply to the AVM by enlarged, tortuous ovarian artery. (G) Postembolization aortogram for bilateral uterine arteries and left ovarian artery reveal no residual AVM flow.
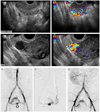
Fig. 2
Magnetic resonance image of ruptured arteriovenous malformation (AVM), and macroscopic findings by emergency exploratory laparotomy. (A) Axial and (B) sagittal computed tomography scan images demonstrate rupture of cystic lesion on left fundal portion of uterus (arrowhead). Large amount hemoperitoneum is noted in abdominopelvic cavity. (C) About 3×3 cm sized subserosal type AVM is noted on fundus (arrow). (D) The ruptured AVM is removed, and repaired.
References
1. Grivell RM, Reid KM, Mellor A. Uterine arteriovenous malformations: a review of the current literature. Obstet Gynecol Surv. 2005; 60:761–767.
2. Lin AC, Hung YC, Huang LC, Chiu TH, Ho M. Successful treatment of uterine arteriovenous malformation with percutaneous embolization. Taiwan J Obstet Gynecol. 2007; 46:60–63.
3. Brown JV 3rd, Asrat T, Epstein HD, Oglevie S, Goldstein BH. Contemporary diagnosis and management of a uterine arteriovenous malformation. Obstet Gynecol. 2008; 112:467–470.
4. Timmerman D, Wauters J, Van Calenbergh S, Van Schoubroeck D, Maleux G, Van Den Bosch T, et al. Color Doppler imaging is a valuable tool for the diagnosis and management of uterine vascular malformations. Ultrasound Obstet Gynecol. 2003; 21:570–577.
5. Ginsberg NA, Hammer R, Parihk S, Tamura R, Sabbagha RE. Arteriovenous malformation of the uterus associated with a missed abortion. Ultrasound Obstet Gynecol. 1994; 4:235–237.
6. Halperin R, Schneider D, Maymon R, Peer A, Pansky M, Herman A. Arteriovenous malformation after uterine curettage: a report of 3 cases. J Reprod Med. 2007; 52:445–449.
7. Nicholson AA, Turnbull LW, Coady AM, Guthrie K. Diagnosis and management of uterine arterio-venous malformations. Clin Radiol. 1999; 54:265–269.
8. Molvi SN, Dash K, Rastogi H, Khanna SB. Transcatheter embolization of uterine arteriovenous malformation: report of 2 cases and review of literature. J Minim Invasive Gynecol. 2011; 18:812–819.
9. Levy-Zaubermann Y, Capmas P, Legendre G, Fernandez H. Laparoscopic management of uterine arteriovenous malformation via occlusion of internal iliac arteries. J Minim Invasive Gynecol. 2012; 19:785–788.
10. Peitsidis P, Manolakos E, Tsekoura V, Kreienberg R, Schwentner L. Uterine arteriovenous malformations induced after diagnostic curettage: a systematic review. Arch Gynecol Obstet. 2011; 284:1137–1151.
11. Kobayashi T, Yamazaki T, Takahashi M. Characteristic radiologic findings for cavernous hemangioma of the uterus. AJR Am J Roentgenol. 1999; 172:1147–1148.
12. Huang MW, Muradali D, Thurston WA, Burns PN, Wilson SR. Uterine arteriovenous malformations: gray-scale and Doppler US features with MR imaging correlation. Radiology. 1998; 206:115–123.
13. Fleming H, Ostor AG, Pickel H, Fortune DW. Arteriovenous malformations of the uterus. Obstet Gynecol. 1989; 73:209–214.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download