Abstract
Objective
This study was performed to compare the expression of CD44 in endometrial stromal cells (ESCs) of women with and without endometriosis and to evaluate the role of CD44 in the adherence of ESCs to peritoneal mesothelial cells (PMCs).
Methods
A PMC adherence assay was performed to evaluate the adherence of ESCs to PMCs in women with and without endometriosis. The expression of CD44 mRNA was measured by reverse transcription-polymerase chain reaction. CD44 protein was evaluated by Western blot analysis.
Results
There were no significant differences in the expression of CD44 mRNA and protein in ESCs or in the binding of ESCs to PMCs between patients with endometriosis and controls. Although the expression of CD44 protein was decreased in both women with endometriosis and controls after anti-CD44 antibody treatment, there was no effect on binding of ESCs to PMCs. Treatment of ESCs with peritoneal fluid from endometriosis patients resulted in a significant increase in binding of ESCs to PMCs compared to untreated ESCs in the endometriosis group.
Endometriosis is a chronic progressive disease inducing various symptoms such as infertility, pelvic pain, and menstrual pain due to the presence of endometrial tissues outside of the uterus. Even though endometriosis occurs in approximately 6% to 10% of women, the incidence increases to up to 35% to 50% for women with pelvic pain or infertility [1]. The causes of endometriosis are not clearly understood in spite of many research efforts. However, the transplantation theory by Sampson is generally accepted, suggesting that retrogradely menstruated endometrial cells implant in the pelvic peritoneum and thereby cause endometriosis. However, approximately 76% to 90% of women have shown the backflow of menstruation in laparoscopic examinations, while the incidence of endometriosis is much lower [2]. Thus, genetic factors, environmental factors, and changes in immunological as well as endocrinologic functions are thought to be involved in the occurrence of endometriosis. Additionally, some studies have demonstrated that the eutopic endometrial tissues in patients with endometriosis are considerably different from those in patients without endometriosis; therefore, such abnormal findings are also anticipated to play important roles in the occurrence of endometriosis [3]. The mechanism of the adherence of endometrial cells to the peritoneum has not yet been elucidated. Nevertheless, Lucidi et al. [4] found that endometrial stromal cells (ESCs) adhere to peritoneal mesothelial cells (PMCs) via experimental models, and they further suggested that the source of endometrium, rather than the source of PMCs, has the greatest effects on ESCs binding to PMCs.
Lately, several researchers have demonstrated that CD44, the main receptor of hyaluronic acid (HA), was associated with the peritoneal adherence of gastric and ovarian carcinoma cells [5,6]. CD44, a transmembrane glycoprotein, is involved in intercellular interactions as well as in interactions between cells and the extracellular matrix [7]. As the genomic locus for CD44 includes 20 exons, several isoforms are present. The smallest CD44 isoforms lack all variable regions and are called "standard" CD44 (CD44s), and the CD44 isoforms with variable regions consisting of 10 exons via alternative splicing are called v1-v10. To the best of our knowledge, this is the first systematic investigation of the expression of CD44 isoforms in the endometrium. Although the expression of CD44 was determined through reverse-transcription polymerase chain reaction (RT-PCR) and immunohistochemical staining in several studies, the results were not consistent [8-12].
Therefore, the objectives of this study were to compare the expression of CD44 in ESCs between patients with endometriosis and controls and to investigate the roles of CD44 in the adherence of ESCs to PMCs.
For the endometriosis group, we recruited 13 patients who were confirmed to have endometriosis based on pathologic examinations after conservative surgery in the department of obstetrics and gynecology at Pusan National University Yangsan Hospital. The recruited patients had no history of medications such as gonadotropin releasing hormone agonist or steroids within the previous six months. The patients were not pregnant and had normal menstrual cycles. For the control group, we recruited 13 females with normal menstrual cycles who underwent hysterectomy because of benign gynecologic diseases or carcinoma in situ of the cervix. There were no gross lesions to consider the presence of endometriosis during surgery of the control group.
This study was approved by the Institutional Review Board at Pusan National University. Sufficient explanation regarding the study was provided to all subjects, and informed consent was obtained.
Endometrial aspiration biopsy using a Novak curette was performed during conservative surgery in the endometriosis group, whereas the endometrial tissues were obtained from the resected uterus after hysterectomy in the control group. The entire process of collecting tissues was conducted under aseptic conditions. The collected tissues were placed in conical tubes containing Dulbecco's modified Eagle's media F12 (DMEM:F12, Gibco, Grand Island, NY, USA) with 1% antibiotics/antimycotics (Gibco), and then the samples were transferred from the operation room to laboratory. The rest of the blood was removed by washing several times with phosphate-buffered saline (PBS, Gibco).
The endometrial tissues were placed in culture dishes with DMEM:F12 and then minced to 1-2 mm in size using sterile scissors. Minced tissues in the solution were transferred to conical tubes and treated with 0.1% collagenase type 1 (Gibco) and 0.05% DNAse (Boehringer Mannheim, Mannheim, Germany) followed by incubation in a waterbath at 37℃ for 1 hour 30 minutes while shaking [13]. After that, the media containing 10% fetal bovine serum (FBS, Gibco, USA) was added to stop the enzyme reaction, the samples were filtered through a 100 µm nylon membrane (BD Falcon, Bedford, MA, USA) and the supernatant was removed through centrifugation.
The precipitated cells were reconstituted in media and filtered through a 40 µm nylon membrane (BD Falcon, Bedford, MA, USA) [14]. After centrifugation of the precipitates, the supernatant was removed and the precipitated cells were resuspended in DMEM:F12 (1:1) media with 10% FBS, 1% antibiotics/antimycotics and 5 µg/mL of insulin (Sigma, St. Louis, MO, USA). Then, the cells were aliquoted into 100 mm culture dishes (BD Falcon) and incubated at room temperature for 20 minutes. The cells that did not adhere to the culture dishes were discarded, and new media was added to the stromal cells adhering to the culture dishes. Then, the cells were placed in an incubator at 37℃ and 5% CO2 for 24 hour with replacement of the media once every two days [15]. Immunohistochemical staining against Vimentin and Cytokeratin (Novocastra Laboratories Ltd., Newcastle, UK) was performed on the cultured cells. Based upon the positive and negative results, only ESCs with greater than 97% purity were utilized in the study.
The LP9 human peritoneal cell line was purchased from NIA Aging Cell Culture Repository (USA). The cell line was maintained in MCDB131/M199 (1:1) media containing 20 ng/mL of epithelial growth factor (Sigma), 2 mM L-glutamin (Gibco), HEPES buffer (Sigma), 0.05 µg/mL of hydrocortisone (Sigma), 1% antibiotics/antimycotics, and 10% FBS [4].
SK-OV-3 human ovarian carcinoma cells were obtained from the Korean Cell Bank and utilized as the control group for adherence assays. SK-OV-3 cells were cultured in RPMI (Gibco) media with HEPES buffer, 10% FBS, and 1% antibiotics/antimycotics. Each cell line was cultured in an incubator at 37℃ and 5% CO2 with replacement of the media once every two to three days.
To perform an in vivo assay with adherence conditions similar to those of endometrial and peritoneal cells in the body, LP9 cells were attached to the bottom of the culture dishes, and adherence was determined by attaching fluorescent materials on the primarily cultured ESCs. The cultured LP9 cells (1×104 cells/well) were aliquoted in 96-well plates and then utilized in the study after culturing for three days. The cultured ESCs were detached from the culture dishes using non-enzymatic cell dissociation media (Sigma) and centrifuged. The precipitated cells were reconstituted in media and attached to Calcein AM, a fluorescent material. The precipitated fluorescent cells (100 µL/well, 2×104 cells/100 µL) were aliquoted in the 96-well plates in which LP9 cells had been placed. After 1 hour of incubation, absorbance was measured at 485 to 538 nm utilizing a fluorometer (FIUOstar OPTIMA, BMG Labtech, Offenburg, Germany). After the measurement, the substances in the 96-well plates were discarded and then cultured upside down in an incubator containing PBS with calcium and magnesium at 37℃ and 5% CO2 while shaking at 20 rpm for 20 minutes. The precipitated fluorescent ESCs that are not attached to LP9 cell layer will be removed, and then ESCs attached to LP9 cells are left in the 96-well plates. Then, the 96-well plates were subjected to absorbance measurements. The adherence was calculated using the equation (fluorescent values after washing/fluorescent values before washing ×100) in each well and then expressed as a percentage [4]. The assays were performed in triplicate, and the averages were used in the results.
The peritoneal fluids were centrifuged at 800 g for 10 minutes within 30 minutes after collection in the operating room, and then the supernatants were tightly sealed and stored in a deep freezer at -70℃. The mixture of the peritoneal fluids from the endometriosis group and the control group was used for the treatment of cells. The endometrial cells (1×106 cells) were aliquoted in 100 mm dishes. After incubation for 48 hours, the peritoneal fluids mixed with 10% of the culture media were added, and the cells were treated for 24 hours. The cells were then collected for further analysis.
The endometrial cells collected for the adherence assay with peritoneal cells (1×106 cells/mL) were treated with 10 µg/mL of anti-CD44 antibody (Lab vision corp., USA) at 37℃ for 30 minutes, and then the cells were collected for further analysis.
For RNA separation, the cells were dissolved by adding Trizol (Gibco, BRL) to the tissues, and chloroform (Sigma), 10% of total volume, was added prior to centrifugation to obtain supernatant. Then, the same amount of isopropanol (Sigma) was mixed to precipitate RNA. After washing the RNA precipitates with 75% ethanol, the supernatant was removed through centrifugation, and the precipitates were air-dried. The dried RNA was dissolved in diethylpyrocarbonate (DEPC, Sigma) and then subjected to absorbance and purity measurements at 260 and 280 nm, respectively. The dried RNA was stored in at -70℃ until needed for further analysis.
A total of 2 µg of RNA was added to the reaction solution containing 5×RNA PCR buffer, dNTP (2.5 mM), MgCl2 (25 mM), oligo dT primer, RNase inhibitor, and MMLV reverse transcriptase (Promega) and reacted at 42℃ for 60 minutes and at 95℃ for 5 minutes to synthesize cDNA. PCR was performed using the synthesized cDNA as templates. PCR was carried out utilizing CD44 primer (forward primer, 5' TCC CAG TAT GAC ACA TAT TGC 3'; reverse primer, 5' CAC CTT CTT CGA CTG TTG AC 3') [16]. The expression of mRNA was standardized by GAPDH (forward primer, 5' CGG AGT CAA CGG GAT ATG GA 3'; reverse primer, 5' AGC CTT CTC CAT GGT GGT GAA GAC 3') in each sample. The results of the PCR products were confirmed by gel electrophoresis with 2% agarose gel.
Proteins were extracted from the cultured cells using cell extraction buffer (Pierce, Rockford, IL, USA) and then quantified utilizing Bio-Rad protein quantification reagent (Bio-Rad, Hercules, CA, USA).
The protein extracts, 20 µg of each experimental group, were loaded on 10% SDS-polyacrylamide gel to separate the proteins via gel electrophoresis, and the acrylamide gel with separated proteins was transferred to polyvinylidene difluoride (PVDF) membrane (Bio-Rad, Hercules, CA, USA). The membrane was incubated in TBS with 0.1% TBS-T containing 5% non-fat dry milk for 1 hour in order to block non-specific binding. Then, the membrane was reacted with 1:1000 anti-CD44H (R&D Systems, Minneapolis, MN, USA) and β-actin (Sigma-Aldrich, St. Louis, MO, USA) at 4℃ overnight, followed by six ten-minute washes with TBS-T. Then, the membrane was reacted with 1:5,000 horseradish peroxidase (HRP)-conjugated anti-mouse goat Ig G antibody (Santa Cruz, San Diego, CA, USA) diluted by TBS-T at room temperature for 1 hour and washed six times for 5 minutes each. Lastly, the proteins were developed using an enhanced chemiluminescence (ECL) kit (Western blotting detection reagents, Amersham-Biosciences), and the X-ray film was exposed to light to determine protein expression.
All statistical analysis was performed using SPSS ver. 17.0 (SPSS Inc., Chicago, IL, USA). Mann-Whitney U-test was applied to compare the adherence of ESCs to the peritoneal cells between the two groups, while the Kruskall-Wallis test was utilized in the comparison of the three groups after the treatment with peritoneal fluids. A P-value <0.05 was considered to be statistically significant.
The adherence of ESCs to peritoneal cells is shown in Fig. 1. Slightly higher adherence was observed in the endometriosis group (42.8±7.4%) compared to the control group (38.9±6.4%), but no statistically significant differences were found (P=0.204).
RT-PCR was carried out using a primer from the study by Cannistra et al. [5] in order to identify expression of CD44 standard (CD44s) type and variant type in ESCs (Fig. 2). CD44s isoforms were expressed in both the endometriosis group and the control group, whereas the expression of variant CD44 isoforms was not observed in either group. Statistically significant differences were not found in the expression of CD44 types between the endometriosis group and the control group.
Fig. 3 demonstrates the expression of CD44s proteins via Western blot analysis. The CD44 protein expression in the endometriosis group and the control group showed differences between individuals, but expression was not consistent in each group. In addition, the CD44 protein expression in ESCs of each individual did not show a significant correlation with the adherence to peritoneal cells.
Fig. 4 describes the results of anti-CD44 antibody treatment of ESCs to evaluate the effects of CD44 expression on the adherence to the peritoneal cells. Anti-CD44 antibody induced remarkable reductions in CD44 protein expression in the endometriosis group as well as the control group, but the reductions were not significantly different between the two groups (Fig. 4A). However, despite the decrease of CD44 protein expression in ESCs, the adherence to peritoneal cells was not decreased in either the endometriosis group or the control group (Fig. 4B).
The adherence of the ESCs to peritoneal cells after treatment with peritoneal fluids was not found to be significantly different among the control group, non-peritoneal fluid-treated group, normal peritoneal fluid-treated group, and endometriosis peritoneal fluid-treated group (Fig. 5A). In the endometriosis group, on the other hand, the adherence tended to be the same or slightly increased in the normal peritoneal fluid-treated group compared to the non-peritoneal fluid-treated group. In particular, when treating with endometriosis peritoneal fluids, there was a statistically significant increase in the adherence of ESCs to the peritoneal cells (Fig. 5B).
The attachment of endometrial cells to the peritoneum is an essential step in the development of endometriosis. Lucidi et al. [4] demonstrated that ESCs cultured in experimental models adhere to PMCs. Recently, a study performed by Griffith et al. [17] compared endometriosis patients with normal women using menstrual endometrial cells. For the first time, they reported that the adherence to PMCs was increased in endometriosis patients and further suggested that the menstrual endometrium of endometriosis patients was different from that in normal women. In the present study, the ESCs of the endometriosis group and normal group were cultured, and the adherence to PMCs was tested. We found that the adherence tended to increase in the endometriosis group, but statistically significant differences were not exhibited. However, it seems to be very difficult to draw definitive conclusions because the endometrial tissues collected during surgery were not classified based upon menstrual cycles in this study, and the study by Griffith was focused on the endometrial tissues during menstruation assuming that the pathogenesis of endometriosis would be the backflow of menstruation.
In the analysis of the CD44 expression of ESCs in this study, the CD44 mRNA and protein were expressed, but the variant type of CD44 was not, without notable differences between the endometriosis group and the control group. As mentioned earlier, studies have investigated CD44 isoform expression in the endometrium, but the results were inconsistent. For normal endometrium, CD44 expression was increased in the secretory phase rather than proliferative phase [8,10], and CD44 was not always expressed in the proliferative phase [12,18]. Moreover, studies found that all CD44s were expressed in the secretory phase, while consistent patterns were not exhibited in the expression of variant type [8-12,18]. As shown in the present study, CD44 expression in endometria from endometriosis patients did not show any difference from normal endometrial tissues [9], or the expression of some variant types was increased in endometriosis patients [17]. Such differences may be due to differences in experimental methods. In particular, in the earlier studies, CD44 expression was mostly measured via immunohistochemical methods, while protein expression was determined through Western blot analysis after culture of the endometrial tissues. Furthermore, the small number of subjects, about 20 subjects in most studies, may have an effect on the results. In this study, the finding that variant type CD44 was not expressed could be due to the insufficient number of subjects as well as the different experimental methods. In addition, the timing of endometrial tissue collection seems to be important. According to the studies reported so far, CD44 expression is increased in the secretory phase and during menstruation and is more likely for the standard type than the variant type. In the present study, endometrial tissues were obtained during surgery. Considering that the surgeries were mostly performed within two weeks after menstruation, more endometrial tissues would be present in the proliferative phase than in the secretory phase, so that the variant type was not expressed, even though the obtained endometrial tissues were not classified by menstrual cycle.
Anti-CD44 antibody treatment of ESCs showed a notable decrease in the expression of CD44 proteins in endometriosis patients and the control group, while the adherence of peritoneal cells was not affected. In contrast, the adherence to peritoneal cells was remarkably increased when ESCs of the endometriosis patients were treated with endometriosis peritoneal fluids. CD44 adheres to extracellular matrix HA and induces cell-to-cell attachments and cell-to-matrix attachments. It is known that CD44 is particularly involved in the adherence of gastric carcinoma and ovarian carcinoma cells to peritoneal mesothelium [5,6]. Many studies have shown that the adherence to peritoneal cells was reduced when ovarian carcinoma cells were treated with anti-CD44 antibody or hyaluronidase [5,6,19]. However, CD44 expression in the present study was not significantly different between the endometriosis group and the control group, and the adherence to peritoneal cells was not influenced by the treatment with anti-CD44 antibody. Therefore, it seems that CD44 does not play an important role in peritoneal adherence of endometrial cells for endometriosis. The remarkable increase in adherence when ESCs of endometriosis patients were treated with endometriosis peritoneal fluids is in agreement with the results from previous studies indicating that cytokines and adhesion molecules in peritoneal fluids are associated with the peritoneal attachments of the endometrial tissues [20,21]. Additionally, considering that the adherence to peritoneal cells was not affected even with treatment with endometriosis peritoneal fluids in the ESCs of the control group, abnormalities in endometrial tissues of endometriosis patients are suggested to be considerably responsible for the occurrence of endometriosis [3].
In conclusion, CD44 expression in ESCs did not differ between the endometriosis group and the control group. Although a decrease of CD44 protein expression was observed in the endometriosis group and the control group after the CD44 antibody treatment, there were no differences in the attachment of ESCs to PMCs between the two groups even after the decrease in CD44 protein. Thus, CD44 expression does not seem to be directly associated with the attachment of ESCs to PMCs. However, since our study did not classify the ESCs based upon menstrual cycle, further studies are necessary to clarify if similar results are obtained when using endometrial cells in the secretory phase and during menstruation, where CD44 expression is known to be increased.
Figures and Tables
Fig. 1
Endometrial stromal cell adherence to peritoneal mesothelial cells in the normal group and the endometriosis group, as measured by peritoneal mesothelial cells adherence assay.
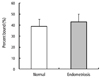
Fig. 2
Expression of CD44 mRNA in the control group (A) and the endometriosis group (B), as measured by reverse transcription-polymerase chain reaction.

Fig. 3
Expression of CD44 protein in the control group (A) and the endometriosis group (B), as measured by Western blot analysis.

Acknowledgments
This study was supported by the Medical Research Institute Grant (2005-21), Pusan National University Hospital.
References
1. Giudice LC, Kao LC. Endometriosis. Lancet. 2004. 364:1789–1799.
2. Berkkanoglu M, Arici A. Immunology and endometriosis. Am J Reprod Immunol. 2003. 50:48–59.
3. Sharpe-Timms KL. Endometrial anomalies in women with endometriosis. Ann N Y Acad Sci. 2001. 943:131–147.
4. Lucidi RS, Witz CA, Chrisco M, Binkley PA, Shain SA, Schenken RS. A novel in vitro model of the early endometriotic lesion demonstrates that attachment of endometrial cells to mesothelial cells is dependent on the source of endometrial cells. Fertil Steril. 2005. 84:16–21.
5. Cannistra SA, Kansas GS, Niloff J, DeFranzo B, Kim Y, Ottensmeier C. Binding of ovarian cancer cells to peritoneal mesothelium in vitro is partly mediated by CD44H. Cancer Res. 1993. 53:3830–3838.
6. Lessan K, Aguiar DJ, Oegema T, Siebenson L, Skubitz AP. CD44 and beta1 integrin mediate ovarian carcinoma cell adhesion to peritoneal mesothelial cells. Am J Pathol. 1999. 154:1525–1537.
7. Naor D, Sionov RV, Ish-Shalom D. CD44: structure, function, and association with the malignant process. Adv Cancer Res. 1997. 71:241–319.
8. Albers A, Thie M, Hohn HP, Denker HW. Differential expression and localization of integrins and CD44 in the membrane domains of human uterine epithelial cells during the menstrual cycle. Acta Anat (Basel). 1995. 153:12–19.
9. Prifti S, Sillem M, Arslic T, Monga B, Rehberger S, Runnebaum B. In vitro expression of soluble and cell surface-associated CD44 on endometrial cells from women with and without endometriosis. Eur J Clin Invest. 1998. 28:1055–1060.
10. Saegusa M, Hashimura M, Okayasu I. CD44 expression in normal, hyperplastic, and malignant endometrium. J Pathol. 1998. 184:297–306.
11. Behzad F, Seif MW, Campbell S, Aplin JD. Expression of two isoforms of CD44 in human endometrium. Biol Reprod. 1994. 51:739–747.
12. Yaegashi N, Fujita N, Yajima A, Nakamura M. Menstrual cycle dependent expression of CD44 in normal human endometrium. Hum Pathol. 1995. 26:862–865.
13. Kirk D, Irwin JC. Normal human endometrium in cell culture. Methods Cell Biol. 1980. 21B:51–77.
14. Osteen KG, Hill GA, Hargrove JT, Gorstein F. Development of a method to isolate and culture highly purified populations of stromal and epithelial cells from human endometrial biopsy specimens. Fertil Steril. 1989. 52:965–972.
15. Pierro E, Minici F, Alesiani O, Miceli F, Proto C, Screpanti I, et al. Stromal-epithelial interactions modulate estrogen responsiveness in normal human endometrium. Biol Reprod. 2001. 64:831–838.
16. Screaton GR, Bell MV, Bell JI, Jackson DG. The identification of a new alternative exon with highly restricted tissue expression in transcripts encoding the mouse Pgp-1 (CD44) homing receptor. Comparison of all 10 variable exons between mouse, human, and rat. J Biol Chem. 1993. 268:12235–12238.
17. Griffith JS, Liu YG, Tekmal RR, Binkley PA, Holden AE, Schenken RS. Menstrual endometrial cells from women with endometriosis demonstrate increased adherence to peritoneal cells and increased expression of CD44 splice variants. Fertil Steril. 2010. 93:1745–1749.
18. Afify AM, Craig S, Paulino AF. Temporal variation in the distribution of hyaluronic acid, CD44s, and CD44v6 in the human endometrium across the menstrual cycle. Appl Immunohistochem Mol Morphol. 2006. 14:328–333.
19. Gardner MJ, Catterall JB, Jones LM, Turner GA. Human ovarian tumour cells can bind hyaluronic acid via membrane CD44: a possible step in peritoneal metastasis. Clin Exp Metastasis. 1996. 14:325–334.
20. Wu MY, Ho HN. The role of cytokines in endometriosis. Am J Reprod Immunol. 2003. 49:285–296.
21. Harada T, Iwabe T, Terakawa N. Role of cytokines in endometriosis. Fertil Steril. 2001. 76:1–10.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download