Abstract
Background
To assess the cost-effectiveness of drug therapy to prevent osteoporotic fractures in postmenopausal women with osteopenia in Korea.
Methods
A Markov cohort simulation was conducted for lifetime with a hypothetical cohort of postmenopausal women with osteopenia and without prior fractures. They were assumed to receive calcium/vitamin D supplements only or drug therapy (i.e., raloxifene or risedronate) along with calcium/vitamin D for 5 years. The Markov model includes fracture-specific and non-fracture specific health states (i.e. breast cancer and venous thromboembolism), and all-cause death. Published literature was used to determine the model parameters. Local data were used to estimate the baseline incidence rates of fracture in those with osteopenia and the costs associated with each health state.
Results
From a societal perspective, the estimated incremental cost-effectiveness ratios (ICERs) for the base cases that had T-scores between -2.0 and -2.4 and began drug therapy at the age of 55, 60, or 65 years were $16,472, $6,741, and -$13,982 per quality-adjusted life year (QALY) gained, respectively. Sensitivity analyses for medication compliance, risk of death following vertebral fracture, and relaxing definition of osteopenia resulted in ICERs reached to $24,227 per QALY gained.
Conclusions
ICERs for the base case and sensitivity analyses remained within the World Health Organization's willingness-to-pay threshold, which is less than per-capita gross domestic product in Korea (about $25,700). Thus, we conclude that drug therapy for osteopenia would be a cost-effective intervention, and we recommend that the Korean National Health Insurance expand its coverage to include drug therapy for osteopenia.
Osteopenia is a precursor of osteoporosis and is defined as a bone mineral density (BMD) between 1.0 and 2.5 standard deviations below the mean of peak bone mass in healthy, young normal women (T-score between -1.0 and -2.5).[1] About 50% of the postmenopausal women in the U.S. have osteopenia.[2] According to T-scores measured through Korea National Health and Nutrition Examination (KNHANES) survey undertaken in 2009, about 48%, 50%, and 42% of women in their fifties, sixties, and seventies, respectively, were estimated to have osteopenia.[3]
Osteopenia can progress to osteoporosis if left untreated. Osteoporosis exhibits more severe decrements in BMD than osteopenia in which the BMD T-score is -2.5 or less. Compared with osteoporosis, the impact of osteopenia as a risk factor for fractures is under-recognized. However, recent evidence suggests that osteopenia is also a significant risk factor for fragility fractures in older women. A large community-based study undertaken in the U.S. reported that about half of all fragility fractures occur in women with T-score in the osteopenic range.[45] The findings from a prospective population-based study conducted in Rotterdam showed that, of the non-vertebral fractures that occurred in women aged 55 years and over, 44.09% occurred in those with osteoporosis and 43.29% occurred in those with osteopenia.[46] Similar proportions of patients with osteopenia (37.0%) and osteoporosis (37.2%) experienced lumbar spine fractures according to a nationwide cross-sectional study of postmenopausal women in Korea.[7]
The results from these studies demonstrate that if the current intervention threshold of a T-score of -2.5 is used, most individuals who experience fractures will not be offered treatment, and this could lead to missed opportunities to prevent future fractures. Since the prevalence of osteopenia is much higher than that of osteoporosis, the total number of osteoporotic fractures associated with osteopenia is greater than that associated with osteoporosis on a national scale.[689] This suggests that the socioeconomic burden of osteopenia may be as sizable as that associated with osteoporosis.
The active treatment of osteopenia with pharmaceutical interventions is effective at preventing progression to osteoporosis and subsequent fractures, and it helps to restore the BMD to normal levels. In the Multiple Outcomes of Raloxifene Evaluation (MORE) trial, the risks of vertebral or clinical vertebral fractures was significantly declined by 47% (relative risk [RR] 0.53; 95% confidence interval [CI] 0.32-0.88) or 75% (RR 0.25; 95% CI 0.04-0.63), respectively, in patients with osteopenia who were treated with raloxifene (60 mg/day) for 3 years compared with those treated with a placebo.[10] Jolly et al. [11] reported that raloxifene administered for 5 years to postmenopausal women with osteopenia significantly reduced the risk of developing osteoporosis (RR 0.13; 95% CI 0.00-0.37), and that it increased the probability of converting to a normal BMD status at the lumbar spine (RR 4.01; 95% CI 1.34-11.23). The risk reduction for fragility fracture was also observed in osteopenic women receiving 5mg risedronate daily for 1.5 to 3 years (RR of risedronate vs. placebo 0.33; 95% CI 0.01-0.44).[4]
In Korea, access to pharmaceutical intervention to prevent osteoporosis in patients with T-score in the osteopenic range is limited because the National Health Insurance (NHI) does not provide coverage for these patients. In a recent study, it was projected that if the NHI expanded its coverage to include drug therapy for osteopenic patients who are at high risks of fractures, the cumulative number of fractures that would have been prevented between 2011 and 2015 would have increased 2.3 times.[3] To assess the value of pharmaceutical interventions and to inform reimbursement decisions, it is important to evaluate both the clinical and economic implications. In the present study, we assessed the cost-effectiveness of drug therapy to prevent osteoporotic fractures in postmenopausal women with osteopenia to determine whether it would be appropriate to recommend that the NHI expands its coverage to include drug therapy for osteopenia.
Using a state transition Markov cohort model, we simulated the lifetime prognosis of a hypothetical cohort of postmenopausal women who were aged 55, 60, or 65 years, had BMD T-scores ranging from -1.0 to -2.4 at the femoral neck, and who did not have histories of osteoporotic fractures. We assumed that all of the cohort members received either calcium/vitamin D supplements only (the control arm), or drug therapy with raloxifene (60 mg/day) or risedronate (35 mg/week) in addition to calcium/vitamin D supplements for 5 years (the treatment arm). The 5-year treatment period was determined based on earlier studies that had evaluated the cost-effectiveness of raloxifene [1213] or risedronate.[14]
This study adopted a published model, which was developed
and validated in previous studies that assessed the cost-effectiveness of the prevention and treatment of osteoporosis.[1315] A literature search for a reference model revealed 2 types of models. For interventions with bisphosphonate products, a "fracture-specific model" has been used that includes only fracture events such as spine, hip, wrist, and other fractures.[15] For interventions with selective estrogen receptor modulators (SERMs) or hormone replacement therapy, a "non-fracture-specific or extra-skeletal model" has been utilized to account for fracture and non-fracture outcomes such as breast cancer (BC) and venous thromboembolism (VTE).[1316] Since our study included an SERM, namely, raloxifene, as a pharmaceutical intervention, we used the non-fracture-specific model. The structure of the model is presented in Figure 1.
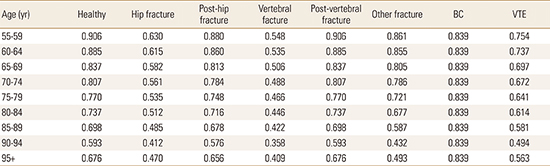
For each health intervention strategy (i.e., no drug therapy or drug therapy), all of the individuals began the simulation in the first health state, 'healthy (i.e., osteopenia without a fracture, BC, or VTE),' and they moved among the 6 fracture-specific health states (i.e., hip, vertebral, or other fracture, post-hip fracture, post-vertebral fracture, and healthy state), 2 non-fracture specific states (i.e., BC and VTE), or all-cause death in yearly cycles for any specified time horizon up to an age of 100 years. During the simulation, those who experienced other fractures can have all types of fractures in the future. However, we assumed that the patients who experienced hip fractures could only have hip fractures, and those experiencing vertebral fractures could have vertebral or hip fractures, because the clinical and economic consequences of vertebral and other fractures are negligible compared with those associated with hip fractures.[17] Once individuals have experienced a hip or a vertebral fracture, they will never return to a normal healthy state and remain in a "post-hip fracture" or a "post-vertebral fracture" state, unless they have a re-fracture, BC, or VTE, or dies. However, those experiencing other fractures can return to a "healthy state." We assumed that those who experienced BC remained in a "BC state" unless they died. However, those experiencing VTE could return to a "healthy state" unless they died. All of the health states could transition to all-cause death.
The published literature was used to determine the effectiveness of the drug therapy and the transition probabilities associated with each health state. Local epidemiologic data and Korean NHI claims data were used to estimate the baseline incidence of fractures among those with osteopenia and the costs associated with each health state. Cost-effectiveness was assessed as incremental cost-effectiveness ratios (ICERs), which are calculated as shown in equation (1); specifically, the incremental cost per fracture prevented and the incremental cost per quality-adjusted life year (QALY) gained. Since the objective of the present study is to determine the cost-effectiveness of drug therapy to prevent osteoporotic fractures in postmenopausal women with osteopenia, we presented the weighted average ICER of the two drugs (i.e., raloxifene and risedronate). To reflect the current usage of the two drugs in Korea, the market share for each drug (24% for raloxifene and 76% for risedronate), obtained from the Korean Information Management System, was used as weight to calculate the weighted average ICER of the two drugs.
Equation (1):
ICER=(Ct-Cc)/(Et-Ec)
here, Ct: cost of the treatment arm
Cc: cost of the control arm
Et: effectiveness of the treatment arm, measured as fracture prevented or QALY gained
Ec: effectiveness of the control arm, measured as fracture prevented or QALY gained
The key parameters and the data sources used in the model are summarized in Table 1, and are described in detail below. Additional information is available in the technical appendix.
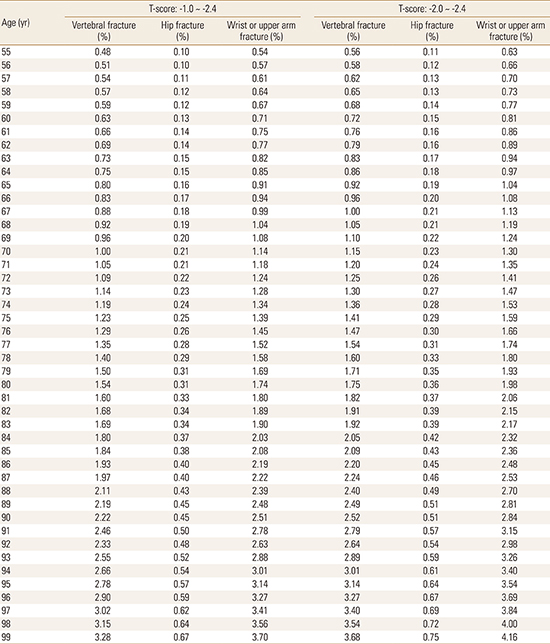
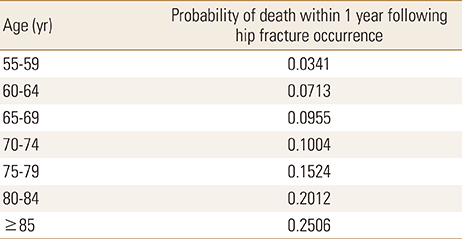
The baseline incidence rates of fragile fractures among Korean women with osteopenia were obtained from the literature, which calculated the age-, gender-, and T-score-dependent incidence rates of vertebral, hip, and other fractures using the KNHANES data (Appendix 1).[3] The increased risk of mortality associated with hip or vertebral fractures was incorporated into the model. We calculated the probability of fatal hip fractures for each age based on the age- and gender-specific post-hip fracture mortality rate within 1 year of the fracture occurring, which were obtained from a Korean population-based study (Appendix 2).[18] For those who experienced hip fractures, mortality risk multipliers of 2.5, 2.1, 1.8, 1.6, and 1.4 were applied to the other-cause mortality at years 1-5 after the hip fracture.[19] For those who experienced vertebral fractures, mortality risk multipliers were applied to the other-cause mortality risks at years 1 (2.5) and 2 (1.3) only,[19] following consultation with local clinicians to avoid overestimating the mortality associated with vertebral fractures.
Previous fractures increase the risk of future fractures [1320]; therefore, we used post-fracture risk multipliers to calculate the increased rates of subsequent fractures according to the locations of the previous fractures.[20] The age-specific annual risk of BC was based on the 2010 Korean National Cancer Surveillance data.[21] The average annual probabilities of BC mortality were estimated using the average annual mortality rates over 5 years that follow a BC diagnosis,[22] and they were based on the BC stage at diagnosis and distributions of the stages at diagnosis among Korean BC patients.[23] We assumed that 5% of the patients who experienced VTE died during the year of its occurrence.[19]
Raloxifene hydrochloride (60 mg/day) is a SERM that is indicated for the prevention and treatment of postmenopausal osteoporosis.[10] According to the MORE clinical trial, raloxifene significantly reduces the vertebral fracture risk in postmenopausal women with osteopenia compared with placebo (RR 0.53; 95% CI 0.32-0.88), but it does not have a significant effect on other sites, including hip and wrist (RR 0.9; 95% CI 0.8-1.1).[10] Raloxifene also reduces the risk of BC compared with placebo (RR 0.50; 95% CI 0.30-0.83),[121324] but it increases the risk of VTE (RR 6.0; 95% CI 1.4-25.5).[2425]
Risedronate (5 mg/day or 35 mg/week for the long-acting tablets) is a bisphosphonate product that is prescribed to prevent the loss of bone mass and to treat osteoporosis. Compared with placebo, it effectively reduces the risk of fractures at all sites, including spine, hip, wrist, and other locations (RR 0.33; 95% CI 0.01-0.44), and it does not have non-skeletal effects, for example, on BC and VTE.[4] For patients treated with raloxifene or risedronate, a patient's risk of fracture during the 5-year treatment period was assumed to be reduced by the RR associated with each drug compared with the baseline incidence of osteoporotic fractures in Korean women with osteopenia.[3] After treatment cessation, the residual fracture reduction benefits were phased out linearly over 5 years.[121317]
Since there are no data available regarding compliance with osteopenia medication for Korean patients, we assumed that the patients were 100% compliant with their therapy during the 5-year treatment period. However, this assumption was altered in the sensitivity analysis based on the findings from other countries.[26] In the sensitivity analysis, we assumed that the compliance with the medication in the third year was the same as that during the fourth and fifth years, and if compliance was less than 100%, the treatment effects, adverse events, and the costs of drug therapy associated with fractures, BC, and VTE were adjusted accordingly. We assumed that the patients would stop taking the medication if BC or VTE occurred during the 5-year medication period.[19] Once medication ceased, it was assumed that the non-fracture-specific effects of the medication, namely, BC or VTE, would cease immediately,[1217] but that the fracture-specific effects of the medication would linger in accordance with the duration of the administration of the medication, and that they would phase out linearly in proportion to the duration of the medication.[121317]
For those who were not administered drugs or who were receiving risedronate, the patients' risks of having BC were assumed to be equal to those of Korean women in the same age groups.[21] Age-specific annual incidence rates of VTE in the untreated cohort with osteopenia were derived from literature published overseas,[19] and they were used to provide the baseline risks for VTE in those who were not administered drugs or were receiving risedronate in this study. For those treated with raloxifene, the risk of developing BC was obtained from published randomized clinical trials. Compared with those who received placebo, the RRs for BC in women with osteoporosis who were younger than 80 years and were treated with raloxifene were 0.5 (95% CI 0.3-0.83) and 0.42 (95% CI 0.29-0.59) for the 4- and 8-year observational periods, respectively. Since our model assumed a 5-year medication period, we used the 4-year RR value for our analysis. We assumed that the effect of raloxifene on reducing the risk of BC started during the second year of medication.[19] Since the RR for the VTE incidence among those taking raloxifene compared with those taking a placebo is no longer statistically significant after the second year of medication, we restrictively accounted for the excess risk of VTE during the first year of raloxifene treatment.[25] The effects of raloxifene on BC and VTE stop immediately after the medication ceases.[12]
The utility values for each health state were derived from publications. Assuming that the utility value of the "healthy state", that is, osteopenia without fractures, BC, or VTE, is equivalent to that of the general population of the same age, we used the age-specific European Quality of Life-5 Dimensions (EQ-5D)-based utility values for Korean women who were older than 55 years from the KNHANES survey undertaken from 2008 to 2011. Literature-based health-related utility values determined from Korean patients with different types of fractures [2728] were utilized to estimate utility losses during the year after each type of fracture. The utility values for the post-fracture states in the second year after the fractures occurred were derived from overseas populations because of the lack of local data, and they were 0.813 for the post-hip fracture and 0.905 for the post-vertebral fracture states.[19] The utility value for the health state associated with BC was estimated as a weighted mean EQ-5D value that was calculated from the mean EQ-5D values for each BC stage [19] and the distributions of the stages at diagnosis among Korean BC patients.[23] By subtracting the utility decrement of 0.103 for those experiencing VTE from the median utility value of 0.8 for osteopenic patients without VTE,[29] we calculated 0.697 as the utility value of a VTE event.
Based on the utility values described above, we estimated the 5-year age-specific utility values for each health state. Yoon et al. [27] calculated an average EQ-5D value of 0.488 for 1,545 postmenopausal women who visited 17 hospitals in Korea from 2008 to 2009, were aged over 50 years, and had vertebral fractures. Since the mean age of the patients was 71.6 years, we assumed that the utility value for women aged 70 to 74 years who had experienced vertebral fracture events was 0.488. Using the multipliers of the utility values for Korean women, namely, 1, 0.977, 0.924, 0.891, 0.850, 0.813, 0.771, 0.654, and 0.746, for the 5-year age increments from 55 to 95 years and above, which were derived based on the EQ-5D-based utility values reported from the KNHANES, we calculated the utility values for vertebral fractures for the other age ranges. The age-specific utility values for each health state were estimated using the same method and are presented in Appendix 3.
The analysis was carried out from a societal perspective; hence, we included the medical costs, the transportation costs incurred as a consequence of visiting health care institutions for outpatient and inpatient care, and the caregivers' costs during hospitalization. Since the study subjects were women who were aged 55 years or older and most were out of the labor market, productivity loss costs as a consequence of illness were not considered. NHI claims data or statistical reports were utilized to calculate the medical costs for services covered by the NHI. For the services that were not covered by the NHI, the medical costs were estimated based on the overall or disease-specific cost ratios of covered to non-covered services published by NHI corporation. The transportation and caregivers' costs were estimated based on the average numbers of outpatient visits and hospital admissions, and on the numbers of days spent in hospital per year in association with each health state, which were determined from the NHI claims data or statistical reports. All of the costs are based on the 2014 value of U.S. dollars, when $1 was approximately equal to 1,100 Korean won. Both the costs and the effects were discounted at an annual rate of 5%.
For the "healthy", "post-hip fracture" (for the third year and subsequent years after its occurrence), and "post-vertebral fracture" (for the third year and subsequent years after its occurrence) health states, we assigned an annual treatment cost for osteopenia, which included medication costs for prescription drugs (i.e., raloxifene or risedronate), and/or calcium/vitamin D supplements, and costs for routine tests to monitor the BMD status and to manage any adverse effects associated with the medication. We assumed that all patients in the treatment and the control arms underwent radiography, central bone densitometry, and tests for vitamin D levels and the bone markers every 3 years, based on practice guidelines [30] and consultations with local clinicians. Those in the treatment arm who received raloxifene or risedronate therapy were assumed to have undergone annual laboratory tests to assess liver function, renal function, and serum calcium, phosphate, and fasting glucose levels to monitor any adverse effects of the drugs.
The average annual costs applied for treating a fracture were estimated from the literature,[3] and they differed according to the type of fracture, as follows: $11,909 for non-fatal hip fractures, $5,365 for vertebral fractures, and $2,839 for other fractures. Since the vertebral and hip fracture-related costs persisted for over 1 year,[1331] we applied annual post-fracture costs to the second year after these fractures, which were $960 for a hip fracture and $905 for a vertebral fracture.[3] For the BC health state, an average annual cost for outpatient and inpatient services provided to patients who were diagnosed with BC was estimated using the NHI claims data from 2011 to 2013, which was $4,933. For the VTE health state, the same data and method were used as those used for the BC health state to calculate the average annual cost of treating VTE, which was $1,711.
Mortality costs were defined as those costs that were incurred during the last stages of life, and they were estimated separately depending on the cause of death, namely, hip fracture, BC, VTE, and other causes. The cost of $17,963 for a BC death was derived from a report entitled "Cost estimation from diagnosis to death of cancer patients", in which the estimated medical costs incurred during the last 12 months before death were determined for each type of cancer based on the National Cancer Surveillance data gathered from 2001 to 2005.[32] Since no data nor literature were available that described the mortality costs associated with deaths caused by hip fractures or VTE, we assumed that the mortality costs for these 2 conditions were equal to the average costs per hospital admission for each condition (i.e., $8,878 for a fatal hip fracture and $6,111 for a VTE death), which were estimated based on a recent NHI statistical report. For the other causes of death, the mortality costs were derived from a publication that estimated the health care costs incurred during the last 12 months before deaths from all causes in elderly Korean people in 2001.[33]
To reflect the reality of routine clinical practice that may involve starting medication for osteopenia, we chose women with T-scores between -2.0 and -2.4, which is near the threshold of osteoporosis, as the base-case population. For 55-year-old women with T-scores in the upper osteopenic range of between -2.0 and -2.4 and without any previous fractures, 5 years of treatment with raloxifene or risedronate in combination with calcium/vitamin D supplements generated ICERs of $6,785 per fracture prevented and $16,472 per QALY gained over a lifetime (Table 2). As the fracture risk increased with age, so did the benefits of intervention to prevent fractures, which led to increases in both clinical gains and cost savings. Thus, treatment with raloxifene or risedronate was more cost-effective for women who began therapy at 60 years of age ($3,162 per fracture prevented and $6,741 per QALY gained). For women aged 65 years, 5-year medication is a dominant strategy, which reduces the fracture risk and is accompanied by cost savings, specifically, -$6,978 per fracture prevented and -$13,982 per QALY gained (Table 2). The expected net total declines in fractures in the treatment group over the remaining life years at an annual discount rate of 5% were estimated to be 62.3, 79.4, and 99.1 fewer fractures per 1,000 women for those starting treatment at 55, 60, and 65 years, respectively.
A series of univariate sensitivity analyses were performed to examine the impact of the uncertainty associated with selected parameters on the estimated ICERs (Fig. 2). The upper and lower limits of the 95% CIs for the effects of the treatments on fractures, BC, and VTE were used in the sensitivity analyses. Among the different parameters examined, the annual discount rate, the T-score intervention threshold, and the effectiveness of raloxifene and risedronate at fracture prevention had the strongest influences, generating ICERs that ranged from $20,318 to $24,227 per QALY gained. According to the World Health Organization's (WHO) guidelines,[34] these ICERs were within the accepted willingness-to-pay threshold, which was defined in this study as the per capita gross domestic product (GDP) in Korea ($25,700) (http://www.index.go.kr/potal/main/EachDtlPageDetail.do?idx_cd=2871).
If a patient with a T-score in full range of the osteopenic status (-1.0 <T-score <-2.5) receives pharmaceutical therapy for 5 years, the ICERs would increase to $9,313 and $5,177 per fracture prevented (or $22,813 and $11,116 per QALY gained) if therapy begins at 55 years and 60 years, respectively, and these values remain within the accepted willingness-to-pay threshold of the per capita GDP in Korea ($25,700). For older women who are aged 65 years, drug therapy remains a dominant option. When we anticipated a worst case scenario for treatment effectiveness with raloxifene (RR for vertebral fractures 0.88), the estimated ICERs were $20,627 and -$13,765 per QALY gained when therapy was initiated at 55 to 65 years of age. Similarly, a worst case scenario for risedronate therapy (RR for vertebral or non-vertebral fractures 0.44) showed that the estimated ICERs were $22,535 and -$13,075 per QALY gained when therapy was initiated at 55 to 65 years of age.
Another parameter that had a strong impact was compliance with medication. When we applied compliance with medication that was less than 100%, the ICERs decreased or became negative. This was mainly caused by the reduction in the cost of the intervention resulted from a low rate of medication intake. While the lower ICERs appeared to represent improvements in the cost-effectiveness of the medications, the numbers of fractures prevented decreased to 19.2, 24.9, and 31.5 fractures per 1,000 women who began treatment for osteopenia at 55, 60, and 65 years of age, respectively, which represents a 70% reduction in fracture prevention compared with the cases who were 100% compliant.
When we assumed a worst case scenario with respect to the risk reduction for BC (RR 0.83) that is associated with taking raloxifene, the ICER remained within the accepted willingness-to-pay threshold of the per capita GDP at all ages. Similarly, a worst case scenario in relation to the risk increase for VTE (RR 25.5) that is associated with raloxifene therapy did not change the cost-effectiveness status of our study's results.
Given the assumed variance, all of the parameters used in the sensitivity analysis had ICERs were less than the GDP per capita. This convinced us that the estimated cost-effectiveness of medical intervention to prevent osteoporosis and fractures in osteopenic patients is robust.
This study examined the cost-effectiveness of drug therapy with raloxifene or risedronate compared with no therapy in Korean postmenopausal women who had osteopenia. While evidence has accumulated that supports the economic outcomes of drug therapy administered to patients with osteoporosis, there is limited evidence that describes the cost-effectiveness of drug therapy for elderly women with osteopenia.[1935363738] Thus, the results from our study add to the empirical evidence that is guiding the selection of treatment strategies for individuals with osteopenia. The results from this study indicate that pharmaceutical interventions for osteopenia are cost-effective at preventing fragile fractures. Our base-case analysis showed that when treatment is initiated at 55, 60, and 65 years, the estimated ICERs are below $25,700 per QALY gained, which is the accepted willingness-to-pay threshold of the per capita GDP. In the sensitivity analysis, the ICERs remained fairly stable at below $25,000 per QALY gained. Hence, we consider that our conclusions are relatively robust.
For women with T-scores within the osteopenic range (i.e., -1.0 - -2.4), pharmaceutical treatment with raloxifene or risedronate in combination with calcium/vitamin D supplements for 5 years was a cost-effective treatment choice. For older women starting medication at 65 years of age, pharmaceutical intervention was a dominant strategy compared with calcium/vitamin D supplements administered alone. This result persisted in the sensitivity analysis, regardless of the parameters chosen, which implies that the T-score intervention threshold for postmenopausal women who are older than 65 years should be changed to -1.0 from the current T-score intervention threshold of -2.5.
Although the fracture risk is much higher for individuals with osteoporosis than for those with osteopenia at the single person level, a higher number of fractures in the population are attributable to osteopenia than to osteoporosis. For example, it was projected that the cumulative number of fractures prevented between 2011 and 2015 would increase by 2.3 times in Korea (77,392 vs. 33,462 fractures) if the NHI extended its coverage to include drug therapy for patients with osteopenia who are at a high risk of fractures.[3] Furthermore, the American Society for Bone and Mineral Research task force for the prevention of refracture strongly recommends early preventive treatment after the initial fragility fracture, which includes a large portion of osteopenic patients by default.[39] Thus, initiating treatment for osteopenia to reduce the fracture risk is a case of prevention paradox, whereby many people who are at a low risk of fractures may give rise to more fractures than the small number of people who are at a high risk of fractures.[640] This phenomenon advocates for a change in the T-score intervention threshold from -2.5 to -1.0 to reduce the social burden associated with fragile fractures in elderly women in our society.
In the sensitivity analysis, we observed that poor compliance with medication generated small or even negative ICERs, which misleadingly implied that poor compliance improves the cost-effectiveness of medical intervention. While poor compliance lowers the intervention costs and leads to better economic outcomes, it also lowers the effects of treatment on fracture prevention. Thus, an effective strategy to improve compliance with medication in clinical practice is necessary to achieve the clinical and economic outcomes expected with pharmaceutical interventions.
While the effect of risedronate is restricted to fracture prevention, raloxifene has non-fracture-specific effects: the beneficial effects of BC reduction and the adverse effects of increase in VTE. However, the sensitivity analyses, which were designed to achieve unfavorable ICERs by using the lowest risk reduction ratio for BC and the highest risk increase ratio for VTE from the 95% CIs, showed marginal changes in the ICERs relative to the results from the base-case analysis. This could be because the baseline risks of BC and VTE among elderly people in Korea are relatively low.
Although not all of the parameters were derived from Korea-specific data, our simulation reflected local scenarios as far as possible when the data were available. For example, our analysis was based on the baseline age-, gender-, and T-score-specific risks for fractures that were estimated from the Korean population. Since the impact of drug treatment on the fracture risk is affected by the baseline fracture risk, the availability of accurate data relating to the baseline risk of the target population is essential for valid simulations. Other epidemiologic data derived from the Korean population included the probability of fatal hip fractures, and the annual probability of the incidence and mortality of BC. All of the cost variables associated with the pharmaceutical interventions and the disease events were derived from NHI claims data and the Korean literature. Age-specific utility values associated with the fracture incidence rates were also derived from the Korean population.
In conclusion, our study results support the need for the initiation of osteopenia treatment to reduce the fracture risk. In relation to the WHO-recommended willingness-to-pay threshold for an intervention to be cost-effective, 5-year pharmaceutical interventions with raloxifene or risedronate for elderly women in Korea above 55 years old represent very cost-effective therapy options for patients with osteopenia. Indeed, drug therapy is a dominant strategy against no therapy for women with osteopenia who are older than 65 years. The fact that drug therapy with raloxifene or risedronate demonstrates cost-effectiveness among patients with osteopenia in Korea is a consequence of the reductions in costs associated with the medications' patent losses and generic products entering the market. When the costs of drugs start to decline, but medical and hospital costs remain the same or increase, then governments may need to rethink their intervention thresholds to manage healthcare costs.
Figures and Tables
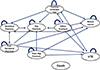 | Fig. 1Structure of the model. Note that it is possible to reach the dead state from all of the health states. VTE, venous thromboembolism. |
 | Fig. 2Tornado diagram of the one-way sensitivity analysis. For the sensitivity analysis, the incremental cost-effectiveness ratio in terms of the cost in United States dollars per quality-adjusted life year gained was computed for each parameter's value range, and they were compared with the willingness-to-pay threshold, which was defined as the per-capita gross domestic product in Korea in 2014 ($25,718). a)per-capita gross domestic product in Korea in 2014. b)upper bound, lower bound of 95% confidence interval for each type of fracture. c)base case (100%) vs. 54% (raloxifene) and 53% (risedronate) for year 1, 38% (raloxifene) and 34% (risedronate) for year 2, and 32% (raloxifene) and 20% (risedronate) for years 3 to 5. RR, relative risk; VF, vertebral fracture; NVF, non-vertebral fracture; BC, breast cancer; VTE, venous thromboembolism; CI, confidence interval; Fx, fracture; QALY, quality-adjusted life year. |
References
1. World Health Organization. Prevention and management of osteoporosis. 2003. cited by 2015 August 3. Available from: http://apps.who.int/iris/bitstream/10665/42841/1/WHO_TRS_921.pdf.
2. Looker AC, Orwoll ES, Johnston CC Jr, et al. Prevalence of low femoral bone density in older U.S. adults from NHANES III. J Bone Miner Res. 1997; 12:1761–1768.

3. National Evidence-based Healthcare Collaborating Agency. Developing Korea-specific assessment criteria for osteoporosis. Seoul: National Evidence-based Healthcare Collaborating Agency;2011.
4. Siris ES, Simon JA, Barton IP, et al. Effects of risedronate on fracture risk in postmenopausal women with osteopenia. Osteoporos Int. 2008; 19:681–686.

5. Siris ES, Miller PD, Barrett-Connor E, et al. Identification and fracture outcomes of undiagnosed low bone mineral density in postmenopausal women: results from the National Osteoporosis Risk Assessment. JAMA. 2001; 286:2815–2822.

6. Schuit SC, van der Klift M, Weel AE, et al. Fracture incidence and association with bone mineral density in elderly men and women: the Rotterdam study. Bone. 2004; 34:195–202.

7. Lee JH, Lee YH, Moon SH. Association between bone mineral density and clinical consequences: cross-sectional study of Korean postmenopausal women in an orthopaedic outpatient clinic. J Korean Med Sci. 2014; 29:1152–1160.

8. Pasco JA, Seeman E, Henry MJ, et al. The population burden of fractures originates in women with osteopenia, not osteoporosis. Osteoporos Int. 2006; 17:1404–1409.

9. Siris ES, Chen YT, Abbott TA, et al. Bone mineral density thresholds for pharmacological intervention to prevent fractures. Arch Intern Med. 2004; 164:1108–1112.

10. Kanis JA, Johnell O, Black DM, et al. Effect of raloxifene on the risk of new vertebral fracture in postmenopausal women with osteopenia or osteoporosis: a reanalysis of the Multiple Outcomes of Raloxifene Evaluation trial. Bone. 2003; 33:293–300.

11. Jolly EE, Bjarnason NH, Neven P, et al. Prevention of osteoporosis and uterine effects in postmenopausal women taking raloxifene for 5 years. Menopause. 2003; 10:337–344.

12. Ivergård M, Strom O, Borgström F, et al. Identifying cost-effective treatment with raloxifene in postmenopausal women using risk algorithms for fractures and invasive breast cancer. Bone. 2010; 47:966–974.

13. Kanis JA, Borgström F, Johnell O, et al. Cost-effectiveness of raloxifene in the UK: an economic evaluation based on the MORE study. Osteoporos Int. 2005; 16:15–25.

14. Wasserfallen JB, Krieg MA, Greiner RA, et al. Cost effectiveness and cost utility of risedronate for osteoporosis treatment and fracture prevention in women: a Swiss perspective. J Med Econ. 2008; 11:499–523.

15. Zethraeus N, Borgström F, Ström O, et al. Cost-effectiveness of the treatment and prevention of osteoporosis--a review of the literature and a reference model. Osteoporos Int. 2007; 18:9–23.

16. Kanis JA, Dawson A, Oden A, et al. Cost-effectiveness of preventing hip fracture in the general female population. Osteoporos Int. 2001; 12:356–361.

17. Borgström F, Johnell O, Kanis JA, et al. Cost effectiveness of raloxifene in the treatment of osteoporosis in Sweden: an economic evaluation based on the MORE study. Pharmacoeconomics. 2004; 22:1153–1165.

18. Kang HY, Yang KH, Kim YN, et al. Incidence and mortality of hip fracture among the elderly population in South Korea: a population-based study using the national health insurance claims data. BMC Public Health. 2010; 10:230.

19. Meadows ES, Klein R, Rousculp MD, et al. Cost-effectiveness of preventative therapies for postmenopausal women with osteopenia. BMC Womens Health. 2007; 7:6.

20. Klotzbuecher CM, Ross PD, Landsman PB, et al. Patients with prior fractures have an increased risk of future fractures: a summary of the literature and statistical synthesis. J Bone Miner Res. 2000; 15:721–739.

21. The Korea Central Cancer Registry, National Cancer Center. Annual report of cancer statistics in Korea in 2010. Sejong: Ministry of Health and Welfare;2012.
22. Korean Breast Cancer Society. 2006-2008 Breast cancer facts & figures. 2008. cited by 2015 August 3. Available from: http://www.kbcs.or.kr/journal/file/2006_2008_Breast_Cancer_Facts_and_Figures_updated.pdf.
23. Korean Breast Cancer Society. 2012 Breast cancer facts & figures. 2012. cited by 2015 August 3. Available from: http://www.kbcs.or.kr/journal/file/2012.pdf.
24. Martino S, Cauley JA, Barrett-Connor E, et al. Continuing outcomes relevant to Evista: breast cancer incidence in postmenopausal osteoporotic women in a randomized trial of raloxifene. J Natl Cancer Inst. 2004; 96:1751–1761.

25. Grady D, Ettinger B, Moscarelli E, et al. Safety and adverse effects associated with raloxifene: multiple outcomes of raloxifene evaluation. Obstet Gynecol. 2004; 104:837–844.

26. Weycker D, Macarios D, Edelsberg J, et al. Compliance with drug therapy for postmenopausal osteoporosis. Osteoporos Int. 2006; 17:1645–1652.

27. Yoon SP, Lee SH, Ki CH, et al. Quality of life in patients with osteoporotic vertebral fractures. Asian Spine J. 2014; 8:653–658.

28. Kim JK, Kang JS. Evaluation of the Korean version of the patient-rated wrist evaluation. J Hand Ther. 2013; 26:238–243. quiz 44.

29. Guillemin F, Martinez L, Calvert M, et al. Fear of falling, fracture history, and comorbidities are associated with health-related quality of life among European and US women with osteoporosis in a large international study. Osteoporos Int. 2013; 24:3001–3010.

30. Korean Society for Bone and Mineral Research. Physician's guide for diagnosis & treatment of osteoporosis. Seoul: Korean Society for Bone and Mineral Research;2013.
31. Kang HY, Park SE, Kang DR, et al. Estimating medical expenditure associated with osteoporotic hip fracture in elderly Korean women based on the National Health Insurance Claims Database 2002-2004. Value Health. 2009; 12:Suppl 3. S93–S96.

32. Health Insurance Review and Assessment Service. Cost estimation from diagnosis to death of cancer patients. Wonju: Health Insurance Review and Assessment Service;2010.
33. Ministry for Health WaFA. Medical service utilization and cost among Korean elderly in the period of last twelve months of life. Seoul: Ministry for Health, Welfare and Family Affairs;2001.
34. World Health Organization. Cost effectiveness and strategic planning (WHO-CHOICE). 2015. cited by 2015 August 3. Available from: http://www.who.int/choice/costs/CER_levels/en/.
35. Müller D, Pulm J, Gandjour A. Cost-effectiveness of different strategies for selecting and treating individuals at increased risk of osteoporosis or osteopenia: a systematic review. Value Health. 2012; 15:284–298.

36. Schwenkglenks M, Lippuner K. Simulation-based cost-utility analysis of population screening-based alendronate use in Switzerland. Osteoporos Int. 2007; 18:1481–1491.

37. Schousboe JT, Bauer DC, Nyman JA, et al. Potential for bone turnover markers to cost-effectively identify and select post-menopausal osteopenic women at high risk of fracture for bisphosphonate therapy. Osteoporos Int. 2007; 18:201–210.

38. Schousboe JT, Ensrud KE, Nyman JA, et al. Cost-effectiveness of vertebral fracture assessment to detect prevalent vertebral deformity and select postmenopausal women with a femoral neck T-score>-2.5 for alendronate therapy: a modeling study. J Clin Densitom. 2006; 9:133–143.





 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download