Abstract
Background
Bisphosphonate is used in osteoporosis treatment to repress osteoclast activity, which then decreases levels of osteocalcin (OC). OC, a protein secreted by osteoblasts and released from the bone matrix during osteoclastic bone resorption, has been found to control blood glucose levels by increasing insulin production and sensitivity. The question addressed in this study is whether decreasing OC through bisphosphonate treatment will provoke a change in glucose homeostasis.
Methods
Eighty-four patients with osteoporosis were treated with once-weekly risedronate 35 mg and cholecalciferol 5,600 IU. We measured fasting plasma glucose (FPG), insulin, and undercarboxylated (Glu) and carboxylated (Gla) OC levels at baseline and after 16 weeks. To estimate insulin resistance (IR) and β-cell function (B)%, homeostasis model assessment (HOMA)-IR and HOMA-B% were also calculated, respectively.
Results
The mean FPG level in total subjects increased significantly from 5.3 to 5.5 mmol/L, but no changes in blood glucose were noted in the 24 subjects with impaired fasting glucose. Glu and Gla OC levels declined significantly after treatment. No correlations were observed between changes in OC and changes in glucose, however.
Many researchers are becoming increasingly aware of the importance of the bone not only in the skeleton structure, but also in energy metabolism.[1-3] In particular, osteocalcin (OC), a protein secreted by osteoblasts and released from the bone matrix during osteoclastic bone resorption, has been proven to be involved in glucose metabolism. This process is induced by increasing insulin secretion and cell proliferation in the beta cells of the pancreas and by regulating insulin sensitivity through increased secretion of adiponectin.[3,4] Many cross sectional studies have shown that serum OC concentration are inversely proportional with plasma glucose levels, and fasting insulin, but directly proportional to lower insulin resistance (IR), insulin secretion, and serum adiponectin concentration.[5-7]
Bisphosphonates can reduce fracture risk through the reduction of osteoclastic activity and significantly decrease serum OC concentration.[8] We can speculate that significantly reduced levels of OC by long-term bisphosphate treatment may provoke IR, with subsequent decrease in insulin secretion eventually leading to changes in blood glucose.
We therefore conducted a prospective study to investigate the association between changes in glucose or insulin secretion and changes in OC for users of bisphosphonate.
Participants were selected from a previous randomized clinical trial that had tested for the effectiveness of a weekly bisphosphonate and cholecalciferol.[9] Study subjects were postmenopausal women over the age of 50 that were being treated with bisphosphonate for their osteoporosis at Kyung Hee University Hospital at Gangdong in Korea. The hospital's Institutional Review Board (IRB) approved this study. Patients were treated with once-weekly risedronate 35 mg and cholecalciferol 5,600 IU. None of the participants had previously been diagnosed with diabetes mellitus (DM).
We measured fasting plasma glucose (FPG), fasting plasma insulin (FPI), Undercarboxylated OC (Glu-OC) and carboxylated-type OC (Gla-OC) levels at baseline and after 16 weeks of treatment. Plasma glucose levels were determined by the hexokinase method using an auto-analyzer (Hitachi Koki, Tokyo, Japan). Plasma insulin levels (BioSource Europe S.A., Nivelles, Belgium) were determined using an immunoradiometric assay, which had intra- and inter-assay coefficients of variations (CVs) of 1.6-2.2 and 6.1-6.5%. The Glu-OC and Gla-OC levels in plasma were measured using a commercial enzyme immunoassay (EIA) kit (Takara Bio Inc., Shiga, Japan), which had intra- and inter-assay CVs of 4.4-6.6 and 5.6-9.8% (Glu-OC), and 3.0-4.8 and 0.7-2.4% (Gla-OC). Homeostasis model assessment (HOMA) of IR, as an indicator of IR, and HOMA of β-cell function (B)%, as a representative of pancreatic B, were estimated. HOMA-IR was defined as (FPI [µU/mL]×FPG [mmol/L])/22.5. HOMA-B% was calculated using (20×FPI)/(FPG-3.5).
All data are presented as mean±SD, unless otherwise indicated. All randomized patients were included in the baseline analyses. Homogeneity of treatment groups with respect to age was assessed using the Pearson Chi-square test. Two-way analysis of variance (ANOVA) was used to assess homogeneity with respect to weight, body mass index (BMI) and status of osteoporosis. Correlation coefficients were used to calculate the association between the change in glucose level and the change in OC. Analysis was performed using SPSS version 13.0 (SPSS Inc., Chicago, IL, USA). Null hypotheses of no difference were rejected if P values were less than 0.05.
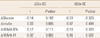
We enrolled 84 participants in this study and classified the subjects according to their fasting glucose levels into the normal group (n=60) and the impaired fasting glucose (IFG, n=24) group (Table 1). After 16 weeks of treatment in total subjects, fasting glucose levels increased significantly from 5.3±0.56 to 5.5±0.75 mmol/L (Table 2). In the subgroup analysis, fasting glucose in the normal group also increased from 5.0±0.35 to 5.4±0.62 mmol/L (P<0.0001) after treatment (Table 3), while no significant changes were seen in the IFG group (Table 4). There were no significant statistical changes in fasting insulin levels, HOMA-IR, and HOMA-B% in the total, and IFG group after 16 weeks, but HOMA-IR significantly increased in normal group only (Table 3). Significant decreases were also seen in Total OC, Glu-OC, and Gla-OC levels in each group. There were no statistical correlations between changes in osteoalcin and glucose parameters after the 16 week-treatment in the total subjects, and normal group (Tables 5, 6).
Knockout mice lacking insulin receptors in their osteoblast have low levels of Glu-OC in their plasma and decreased osteoblast differentiation. These effects, in turn, can impair insulin secretion.[4] It has been found that an endocrinological feedback loop between the bone and the pancreas. Both Glu-OC and total OC have been inversely associated with fasting glucose, fasting insulin, haemoglobin A1c (HbA1C), and HOMA-IR levels in men and women. [10,11] In their study of middle-aged Korean men, Hwang et al.[12] noted that Glu-OC was found to enhance B while Gla-OC was associated with improved insulin sensitivity. Iki et al.[13] found that the level of Glu-OC, but not intact OC, showed a negative correlation with the glycemic index and IR. Shea et al.[14] insisted that circulating OC in its uncarboxylated form was not associated with IR, but total OC and the protein in its Gla form were inversely associated with IR. From the clinical perspective, many cross-sectional studies have found an association between OC and glucose metabolism. However, the few longitudinal studies that have been conducted do not provide sufficient evidence. Furthermore, no study has examined the effects of bisphosphonate on glucose metabolism and insulin secretion or resistance. Therefore, our study was designed to examine the dynamic changes in glucose after bisphosphonate therapy and to show that changes in OC are associated with changes in glucose and insulin homeostasis. We investigated the correlation between the glycemic index and changes in OC after 16 weeks, as bisphosphonates sufficiently suppress bone turnover at 3 to 6 months.
The present study demonstrated that bisphosphonate decreased the levels of two forms of OC, but these changes were not correlated with insulin secretion or resistance. Glucose levels significantly increased after 16 weeks of treatment, and HOMA-IR also increased in normal subjects, but these changes were not observed in whole subjects or IFG group. Furthermore, these changes were within normal range and not associated with changes in OC levels. It is not clear why blood glucose levels and HOMR-IR increased in patients treated with bisphosphonate, but it may be coincidental finding because of small subjects. Although bisphosphonate may affect glucose homeostasis, its effect may be weak or attenuated by other factors. Many clinical trials estimating the efficacy for osteoporosis treatment have not reported that bisphosphonates cause DM as an adverse event.[15] In fact, among users of alendronate, the risk of developing type 2 (T2) DM was reduced in a nationwide cohort study in Denmark.[16] As an antiresorptive treatment, hormone replacement therapy (HRT) can not only decrease OC, but also decrease fasting glucose and insulin levels and lower the incidence of diabetes in randomized trials.[17-19] Furthermore, Hwang et al.[20] demonstrated in a retrospective cohort study that circulating OC levels were not associated with the risk for T2 diabetes in middle-aged male subjects. Even though OC may affect glucose metabolism as in animal system, we can speculate that OC may have weak effects on glucose metabolism for humans, and its impact may be affected by another controlling mechanism.
Our study has some limitations. The sample size is small, and we did not include enough subjects in the IFG group. We also excluded a diabetes group. Furthermore, we did not perform oral glucose tolerance tests to verify glucose status. A 16 week-trial may not be long enough to observe a causal relationship, and a longer-term study including a larger number of subjects is needed for more accurate data.
Despite some limitations, this is a prospectively designed trial. Our findings suggest that bisphosphonate treatment for osteoporosis reduces OC, but that this change is not correlated with glucose homeostasis.
Figures and Tables
Table 2
Changes in fasting glucose, insulin, osteocalcin, insulin secretion and sensitivity indices after a 16 week-bisphosphonate treatment in total subjects
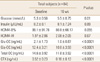
Table 3
Changes in fasting glucose, insulin, osteocalcin, insulin secretion and sensitivity indices after a 16 week-bisphosphonate treatment in subjects with normal fasting glucose
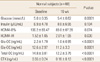
Table 4
Changes in fasting glucose, insulin, osteocalcin, insulin secretion and sensitivity indices after a 16 week-bisphosphonate treatment in subjects with impaired fasting glucose
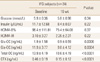
Table 5
Correlation between changes in osteocalcin and glucose parameters after a 16 week-bisphosphonate treatment in total subjects
References
1. Motyl KJ, McCabe LR, Schwartz AV. Bone and glucose meta bolism: a two-way street. Arch Biochem Biophys. 2010. 503:2–10.
2. Ferron M, Wei J, Yoshizawa T, et al. An ELISA-based method to quantify osteocalcin carboxylation in mice. Biochem Biophys Res Commun. 2010. 397:691–696.

3. Lee NK, Sowa H, Hinoi E, et al. Endocrine regulation of energy metabolism by the skeleton. Cell. 2007. 130:456–469.

4. Ferron M, Hinoi E, Karsenty G, et al. Osteocalcin differentially regulates beta cell and adipocyte gene expression and affects the development of metabolic diseases in wild-type mice. Proc Natl Acad Sci U S A. 2008. 105:5266–5270.

5. Fernández-Real JM, Izquierdo M, Ortega F, et al. The relationship of serum osteocalcin concentration to insulin secretion, sensitivity, and disposal with hypocaloric diet and resistance training. J Clin Endocrinol Metab. 2009. 94:237–245.

6. Im JA, Yu BP, Jeon JY, et al. Relationship between osteocalcin and glucose metabolism in postmenopausal women. Clin Chim Acta. 2008. 396:66–69.

7. Kindblom JM, Ohlsson C, Ljunggren O, et al. Plasma osteocalcin is inversely related to fat mass and plasma glucose in elderly Swedish men. J Bone Miner Res. 2009. 24:785–791.

8. Liberman UA, Weiss SR, Bröll J, et al. The Alendronate Phase III Osteoporosis Treatment Study Group. Effect of oral alendronate on bone mineral density and the incidence of fractures in postmenopausal osteoporosis. N Engl J Med. 1995. 333:1437–1443.

9. Chung HY, Chin SO, Kang MI, et al. Efficacy of risedronate with cholecalciferol on 25-hydroxyvitamin D level and bone turnover in Korean patients with osteoporosis. Clin Endocrinol (Oxf). 2011. 74:699–704.

10. Kanazawa I, Yamaguchi T, Yamamoto M, et al. Serum osteocalcin level is associated with glucose metabolism and atherosclerosis parameters in type 2 diabetes mellitus. J Clin Endocrinol Metab. 2009. 94:45–49.

11. Yeap BB, Chubb SA, Flicker L, et al. Reduced serum total osteocalcin is associated with metabolic syndrome in older men via waist circumference, hyperglycemia, and triglyceride levels. Eur J Endocrinol. 2010. 163:265–272.

12. Hwang YC, Jeong IK, Ahn KJ, et al. The uncarboxylated form of osteocalcin is associated with improved glucose tolerance and enhanced beta-cell function in middle-aged male subjects. Diabetes Metab Res Rev. 2009. 25:768–772.

13. Iki M, Tamaki J, Fujita Y, et al. Serum undercarboxylated osteocalcin levels are inversely associated with glycemic status and insulin resistance in an elderly Japanese male population: Fujiwara-kyo Osteoporosis Risk in Men (FORMEN) Study. Osteoporos Int. 2012. 23:761–770.

14. Shea MK, Gundberg CM, Meigs JB, et al. Gamma-carboxylation of osteocalcin and insulin resistance in older men and women. Am J Clin Nutr. 2009. 90:1230–1235.

15. Schwartz AV, Schafer AL, Grey A, et al. Effects of antiresorptive therapies on glucose metabolism: Results from the FIT, HORIZON-PFT and FREEDOM trials. J Bone Miner Res. 2013. doi: http://dx.doi.org/10.1002/jbmr.1865.

16. Vestergaard P. Risk of newly diagnosed type 2 diabetes is reduced in users of alendronate. Calcif Tissue Int. 2011. 89:265–270.

17. Dane C, Dane B, Cetin A, et al. Comparison of the effects of raloxifene and low-dose hormone replacement therapy on bone mineral density and bone turnover in the treatment of postmenopausal osteoporosis. Gynecol Endocrinol. 2007. 23:398–403.

18. Kanaya AM, Herrington D, Vittinghoff E, et al. Glycemic effects of postmenopausal hormone therapy: the Heart and Estrogen/progestin Replacement Study. A randomized, double-blind, placebo-controlled trial. Ann Intern Med. 2003. 138:1–9.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download