Abstract
Background
Interfering RNA (iRNA) represents a recent breakthrough in effective blocking of the target genes in mammalian cells. Angiotensin-converting enzyme (ACE) has been shown to play an important role in the pathogenesis of hypertension. The purposes of this study were to investigate the effects on blood pressure, myocardial hypertrophy and gene expressions of iRNA targeting ACE.
Methods
Twelve week old male Wistar-Kyoto rats were grouped as follows: control group (C group), spontaneously hypertensive rat (SHR) group (H group), and ACE-iRNA group (A group) in which SHR was treated with recombinant lentiviral vectors carrying small hairpin RNA targeting ACE. Reverse transcription-polymerase chain reaction and western blot analysis of ACE, endothelin (ET)-1, angiotensin (AT) II receptor type 1A, neutrophil cytosolic factor, caspase 3, Bax, and Bcl-2 were performed in the heart tissues. Serum AT, ACE, and high sensitive-C reactive protein were estimated.
Results
Systolic blood pressure was significantly decreased in the A group compared with the H group in weeks 3 and 5. Serum AT level was significantly lower on day 1, weeks 3 and 5 after ACE-iRNA treatment. ACE protein contents were significantly lower after ACE-iRNA treatment in week 5. ET-1 and Bcl-2 protein contents were significantly lower after ACE-iRNA treatment in weeks 3 and 5. Bax protein contents were significantly lower after ACE-iRNA treatment in week 3.
Figures and Tables
 | Fig. 1
Construction of shRNA-expression lentiviral vector for targeting rat angiotensin-converting enzyme (ACE). The shRNA-expression lentiviral vector for targeting rat ACE was constructed by inserting synthetic double strand oligonucleotides. CMV, cytomegalovirus; cPPT, central polypurine tract; hCMV, human cytomegalovirus; GFP, green fluorescent protein; IRES, internal ribosome entry site. |
 | Fig. 2Systolic blood pressure after angiotensin-converting enzyme (ACE)-interfering RNA (iRNA) treatment. Systolic blood pressure was significantly decreased in the A group compared with the H group in weeks 3 and 5. C, control; H, hypertension; A, ACE-iRNA group. *p < 0.05 significantly different C group vs. H group. †p < 0.05 significantly different H group vs. A group. |
 | Fig. 3Change of collagen by Masson's Trichrome staining after angiotensin-converting enzyme (ACE)-interfering RNA (iRNA) treatment (×200). (A, B) Collagen penetration in the H group was significantly higher than that in the C group. (C) There was no statistically significant difference between the H and the A group. C, control; H, hypertension; A, ACE-iRNA group. |
 | Fig. 4Angiotensin-converting enzyme (ACE) and endothelin (ET)-1 protein content were significantly higher in the H group compared with that in the C group in weeks 3 and 5. (A) ACE protein contents were significantly lower in the A group compared with the H group in week 5. (B) ET-1 protein contents were significantly lower in the A group compared with the H group in weeks 3 and 5. C, control; H, hypertension; A, ACE-interfering RNA group. *p < 0.05 significantly different C group vs. H group. †p < 0.05 significantly different H group vs. A group. |
 | Fig. 5(A) Angiotensin II receptor (AT II R) type 1A and (B) neutrophil cytosolic factor (NCF) 1 protein levels were significantly higher in the H group compared with the C group in weeks 3 and 5 (p < 0.05) and not significantly different in the A group compared with the H group in weeks 3 and 5. C, control: H, hypertension: A, angiotensin-converting enzyme- interfering RNA group. *p < 0.05 significantly different C group vs. H group. |
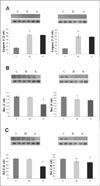 | Fig. 6Caspase-3, Bax, and Bcl-2 protein contents by westernblot analysis after angiotensin-converting enzyme (ACE)-interfering RNA (iRNA) treatment. (A) Caspase-3 protein contents were significantly higher in the H group compared with that in the C group in weeks 3 and 5 and not significantly different in the A group compared with the H group in weeks 3 and 5. (B) Bax protein contents were significantly lower in the A group compared with the C group in week 3. (C) Bcl-2 protein contents were significantly higher in the H group compared with that in the C group in weeks 3 and 5 and significantly lower in the A group compared with the A group in weeks 3 and 5. C, control: H, hypertension: A, ACE-iRNA group. *p < 0.05 significantly different C group vs. H group. †p < 0.05 significantly different H group vs. A group. |
Table 1
Total weight, left heart weight, left heart/total weight, kidney weight, kidney weight/total weight after ACE-iRNA treatment

References
4. Escors D, Breckpot K. Lentiviral vectors in gene therapy: their current status and future potential. Arch Immunol Ther Exp (Warsz). 2010. 58:107–119.

5. Trittibach P, Barker SE, Broderick CA, Natkunarajah M, Duran Y, Robbie SJ, et al. Lentiviral-vector-mediated expression of murine IL-1 receptor antagonist or IL-10 reduces the severity of endotoxin-induced uveitis. Gene Ther. 2008. 15:1478–1488.

6. Jazwa A, Jozkowicz A, Dulak J. New vectors and strategies for cardiovascular gene therapy. Curr Gene Ther. 2007. 7:7–23.

7. Navar LG, Kobori H, Prieto MC, Gonzalez-Villalobos RA. Intratubular renin-angiotensin system in hypertension. Hypertension. 2011. 57:355–362.

8. Ferreira AJ, Murca TM, Fraga-Silva RA, Castro CH, Raizada MK, Santos RA. New cardiovascular and pulmonary therapeutic strategies based on the angiotensin-converting enzyme 2/angiotensin-(1-7)/mas receptor axis. Int J Hypertens. 2012. 2012:147825.

9. Gunaruwan P, Maher A, Williams L, Sharman J, Schmitt M, Campbell R, et al. Effects of bradykinin on venous capacitance in health and treated chronic heart failure. Clin Sci (Lond). 2009. 116:443–450.

10. Sink KM, Leng X, Williamson J, Kritchevsky SB, Yaffe K, Kuller L, et al. Angiotensin-converting enzyme inhibitors and cognitive decline in older adults with hypertension: results from the Cardiovascular Health Study. Arch Intern Med. 2009. 169:1195–1202.
11. Martinez MV, Matulevicius S, Chin K. The association of right ventricular function and pulmonary arterial compliance in patients with idiopathic pulmonary arterial hypertension. J Am Coll Cardiol. 2012. 59:13 Suppl. E1605.
12. Arif H, Aijaz B, Islam M, Aftab U, Kumar S, Shafqat S. Drug compliance after stroke and myocardial infarction: a comparative study. Neurol India. 2007. 55:130–135.

13. Sijen T, Fleenor J, Simmer F, Thijssen KL, Parrish S, Timmons L, et al. On the role of RNA amplification in dsRNA-triggered gene silencing. Cell. 2001. 107:465–476.

14. Freeman EJ, Ferrario CM, Tallant EA. Angiotensins differentially activate phospholipase D in vascular smooth muscle cells from spontaneously hypertensive and Wistar-Kyoto rats. Am J Hypertens. 1995. 8:1105–1111.

15. Lopez B, Gonzalez A, Varo N, Laviades C, Querejeta R, Diez J. Biochemical assessment of myocardial fibrosis in hypertensive heart disease. Hypertension. 2001. 38:1222–1226.

16. An SJ, Boyd R, Wang Y, Qiu X, Wang HD. Endothelin-1 expression in vascular adventitial fibroblasts. Am J Physiol Heart Circ Physiol. 2006. 290:H700–H708.

17. Yokoyama H, Averill DB, Brosnihan KB, Smith RD, Schiffrin EL, Ferrario CM. Role of blood pressure reduction in prevention of cardiac and vascular hypertrophy. Am J Hypertens. 2005. 18:922–929.

18. Fortuno MA, Ravassa S, Etayo JC, Diez J. Overexpression of Bax protein and enhanced apoptosis in the left ventricle of spontaneously hypertensive rats: effects of AT1 blockade with losartan. Hypertension. 1998. 32:280–286.
19. Fortuno MA, Zalba G, Ravassa S, D'Elom E, Beaumont FJ, Fortuno A, et al. p53-mediated upregulation of BAX gene transcription is not involved in Bax-alpha protein overexpression in the left ventricle of spontaneously hypertensive rats. Hypertension. 1999. 33:1348–1352.
20. Cha JH, Lee HR, Kim KC, Cho MS, Hong YM. Changes of gene expressions in spontaneously hypertensive rat model after losartan treatment. Korean Circ J. In press 2012.

21. He J, Bian Y, Gao F, Li M, Qiu L, Wu W, et al. RNA interference targeting the ACE gene reduced blood pressure and improved myocardial remodelling in SHRs. Clin Sci (Lond). 2009. 116:249–255.

22. Vazquez J, Correa de Adjounian MF, Sumners C, Gonzalez A, Diez-Freire C, Raizada MK. Selective silencing of angiotensin receptor subtype 1a (AT1aR) by RNA interference. Hypertension. 2005. 45:115–119.
23. Phillips MI, Mohuczy-Dominiak D, Coffey M, Galli SM, Kimura B, Wu P, et al. Prolonged reduction of high blood pressure with an in vivo, nonpathogenic, adeno-associated viral vector delivery of AT1-R mRNA antisense. Hypertension. 1997. 29(1 Pt 2):374–380.
24. Puddu GM, Cravero E, Ferrari E, Muscari A, Puddu P. Gene-based therapy for hypertension: do preclinical data suggest a promising future? Cardiology. 2007. 108:40–47.
25. Gupta R, Tongers J, Losordo DW. Human studies of angiogenic gene therapy. Circ Res. 2009. 105:724–736.

26. Shim TJ, Choi EJ, Park KJ, Kim YJ, Kim SJ, Lee SY, et al. Establishment of a eNOS gene therapy model using PLGA nanosphere in spontaneous hypertensive rat. J Korean Soc Hypertens. 2010. 16:28–38.
27. Kim SJ, Kim MG, Choue CW, Kim KS, Song JS, Bae JH, et al. Association of angiotensin converting enzyme, angiotensinogen, and angiotensin II type 1 receptor gene polymorphisms with left ventricular hypertrophy in Korean hypertensive patients. J Korean Soc Hypertens. 2005. 11:44–52.




 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download