Abstract
Major hepatic resection, especially right hepatectomy, has been successfully performed by specialized hepatobiliary centers using the robotic platform with low morbidity, conversion rates and outcomes comparable to laparoscopic and open surgery. The authors report a case of robotic-assisted right hepatectomy done for intrahepatic cholangiocarcinoma using anterior approach, after right portal vein embolisation for future liver remnant volume enhancement.
The role of robotic surgery in a complex hepatectomy is still being evaluated as hepatobiliary surgeons become more adept in using this platform. We describe a case of a large-sized, intrahepatic cholangiocarcinoma (ICC) in the right lobe of the liver, resected by a robotic-assisted right hepatectomy via anterior approach. With the remnant left lobe volume being low, future liver remnant volume enhancement by portal vein embolisation (PVE) was done four weeks prior to surgery.
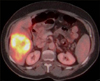
A 60-year-old female was found to have a liver lesion on imaging, during workup for right, upper abdominal pain 6 months prior to presentation at our institution. Core biopsy at an outside institution showed features of adenocarcinoma. Immunohistochemical staining was positive for CK7 and CK18. The patient received two cycles of chemotherapy consisting of gemcitabine and erlotinib. Tumor markers CA 19-9 (700 U/ml) and CA-125 (259 U/ml) were elevated. Imaging, including positron emission tomography-computed tomography (PET-CT; Fig. 1), showed a large, hypodense, FDG-avid, 10 cm, poorly defined and poorly enhanced liver lesion in segments V/VI, extending to segment VIII. Magnetic resonance imaging findings were suggestive of cholangiocarcinoma in segments V/VI with overlying capsular retraction and associated involvement of the distal aspect of the segment V branch of right hepatic vein, intrahepatic, right, posterior portal vein and right hepatic artery. Imaging also revealed Type B (trifurcation) portal venous anatomy and a replaced right hepatic artery arising from the superior mesenteric artery. Endoscopic ultrasound-guided, fine needle aspiration of aortocaval nodes was negative for malignant cytology.
Embolization of the right anterior and posterior portal veins (Type B portal vein), to enhance the FLRV (left lobe with middle hepatic vein), was done as part of preoperative chemotherapy status. The patient underwent robotic-assisted right hepatectomy four weeks after PVE, when the remnant liver volume had increased from 561 ml to 649 ml (future liver remnant volume proportion: 42 to 49.5%). Intraoperatively, the patient received two units of packed red blood cells. The postoperative course was uneventful and she was discharged five days after the procedure. Pathology showed moderately differentiated ICC with lympho-vascular invasion, with lymph nodes and resection margins being free of tumor. Postoperatively she received three more cycles of chemotherapy and is doing well, with no recurrence on imaging, two years following surgery.
Written informed consent was obtained from the patient, for publication of this case report and accompanying images, and no patient identifiers appear in the report.
The patient was placed in a supine position, general anesthesia was administered and pneumo-peritoneum was created using the Hassan technique via a supra-umbilical incision. A 12 mm-port was placed for the robotic camera. A 5-port technique was used. This included two 8 mm robotic ports that were placed in the left upper and right upper quadrant, a 12 mm assist port placed right lateral to the umbilical port and a 5 mm port placed left lateral to it. The da Vinci Si Surgical System (Intuitive Surgical Inc., Sunnyvale, CA, USA) robot was brought into position over the right shoulder of the patient and docked, after placement of the ports.
Porta dissection: Hilar dissection commenced with cholecystectomy and lowering of cystic plate, followed by sequential isolation, ligation and division of the right hepatic artery, and right anterior and posterior portal veins.
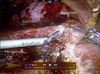
Parenchymal transection: The liver surface was marked for transection after vascular demarcation along the Cantlie's line. Parenchymal transection was then performed under low central venous pressure, using a harmonic scalpel (Ethicon Endo-surgery, Cincinnati, USA) and bipolar grasper with clipping of the vessels and pedicles (Fig. 2). Transection was performed, sparing the middle hepatic vein, and carried down to the edge of the right hepatic vein. The right hepatic duct was divided, using an Endo GIA stapler, after division of the caudate lobe.
Liver Mobilization: Liver was partially mobilized from the diaphragm by division of the coronary ligaments, inferiorly from the vena cava by division of the short hepatic veins between clips. All steps, up to this point, were performed with the robotic approach. The bulk of the tumor prevented complete mobilization.
Specimen extraction: A supra-umbilical, upper midline incision was made and liver mobilization was completed. Finally, the right hepatic vein was divided using an Endo GIA stapler with vascular load. The specimen, consisting of the right lobe with a large tumor, was then extracted from the midline incision.
There are few reported cases on the use of robotic surgery for large, right lobe, liver tumors.123 This is a case of large ICC, resected with anterior approach right hepatectomy, using the robotic platform, after a post portal vein embolization for future liver remnant volume enhancement. The advantages of the robotic platform in such difficult cases are highlighted, along with limitations of performing a right hepatectomy with bulky, right lobe tumors.
The robotic surgical platform (da Vinci Si Surgical System, Intuitive Surgical Inc., Sunnyvale, CA, USA), with its high quality three-dimensional images and a stable camera platform with scaling, tremor filtering, and coaxial alignment of the eyes, considerably helped with the porta dissection, along with the instrumentation, consisting of the EndoWrist, with a 360° range of motion, allowing for precise and meticulous dissection. Bulky right lobes and tumors are difficult to handle, making laparoscopic mobilization difficult, with risk of injury to the liver parenchyma and tumor. The robotic platform makes it easier, although caution is still advisable. Hence, we proceeded with an anterior approach, wherein, for large or capsule-broaching tumors of the right liver lobe, the right liver is not mobilized first. A Hilar dissection was performed and inflow control obtained. Liver parenchymal transection is then performed towards the inferior vena cava and the inferior and right hepatic veins are divided prior to en bloc right lobe removal. Manipulation of the tumor is minimal, with reduced risk of iatrogenic tumor rupture or hematogenous dissemination during surgery.4 Comparative data on the benefits of a minimally invasive approach for a right hepatectomy have been reported by Dagher et al.5 in a study which showed a similar operative time and morbidity rate, less blood loss and shorter length of stay in the laparoscopy group compared to the open hepatectomy group. These results can be replicated using the robotic platform with its use versatile and hepatobiliary surgeons increasing their expertise of it. Wu et al.6 showed that a robotic-assisted, minimally invasive liver resection was feasible, having comparable perioperative outcomes and conversion rates (5%) and morbidity (8%) similar to that of a laparoscopic liver resection. Robotic assistance has led to an increase in the percentage of major resections, from 15% to 39%. Systematic recent reviews by Boggi et al.7 and Ho et al.8 have shown that a major hepatectomy, including the right lobe, can be safely performed robotically with a low conversion rate and morbidity. Chen et al.9 suggested a learning curve for major robotic hepatectomies derived from their large series, comprising of three characteristic phases: initial (phase 1, 15 patients), intermediate (phase 2, 25 patients), and mature (phase 3, 52 patients). The learning effects were underlined by shorter operation time and hospital stay after phase 1 and less blood loss after phase 2.
Taking this technology to new frontiers, Giulianotti et al.10 reported the first case of a robotic-assisted, right lobe, donor hepatectomy in 2012. Thereafter, Chen et al.11 safely performed a donor right hepatectomy using the robotic system with similar blood loss, complication rates and recovery of donor liver function, compared to an open hepatectomy, with no compromise on graft function or recipient outcomes. The robotic group had a longer operative time, but less postoperative patient-controlled analgesia, and a shorter period before returning to work/school.11
This case is a reflection of the endeavor to use minimally invasive approaches for complex liver surgery and demonstrates the utility of the robotic platform, in a right hepatectomy using the anterior approach, for large right lobe tumors.
Figures and Tables
References
1. Giulianotti PC, Sbrana F, Coratti A, Bianco FM, Addeo P, Buchs NC, et al. Totally robotic right hepatectomy: surgical technique and outcomes. Arch Surg. 2011; 146:844–850.
2. Ji WB, Wang HG, Zhao ZM, Duan WD, Lu F, Dong JH. Robotic-assisted laparoscopic anatomic hepatectomy in China: initial experience. Ann Surg. 2011; 253:342–348.
3. Lai EC, Tang CN, Li MK. Robot-assisted laparoscopic hemi-hepatectomy: technique and surgical outcomes. Int J Surg. 2012; 10:11–15.
4. Lai EC, Fan ST, Lo CM, Chu KM, Liu CL. Anterior approach for difficult major right hepatectomy. World J Surg. 1996; 20:314–317. discussion 318.
5. Dagher I, Di Giuro G, Dubrez J, Lainas P, Smadja C, Franco D. Laparoscopic versus open right hepatectomy: a comparative study. Am J Surg. 2009; 198:173–177.
6. Wu YM, Hu RH, Lai HS, Lee PH. Robotic-assisted minimally invasive liver resection. Asian J Surg. 2014; 37:53–57.
7. Boggi U, Caniglia F, Amorese G. Laparoscopic robot-assisted major hepatectomy. J Hepatobiliary Pancreat Sci. 2014; 21:3–10.
8. Ho CM, Wakabayashi G, Nitta H, Ito N, Hasegawa Y, Takahara T. Systematic review of robotic liver resection. Surg Endosc. 2013; 27:732–739.
9. Chen PD, Wu CY, Hu RH, Chen CN, Yuan RH, Liang JT, et al. Robotic major hepatectomy: is there a learning curve? Surgery. 2017; 161:642–649.
10. Giulianotti PC, Tzvetanov I, Jeon H, Bianco F, Spaggiari M, Oberholzer J, et al. Robot-assisted right lobe donor hepatectomy. Transpl Int. 2012; 25:e5–e9.
11. Chen PD, Wu CY, Hu RH, Ho CM, Lee PH, Lai HS, et al. Robotic liver donor right hepatectomy: a pure, minimally invasive approach. Liver Transpl. 2016; 22:1509–1518.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download