Abstract
Objective
The purpose of this study was to report our preliminary experience with endovascular treatment (EVT) for life-threatening bleeding from branches of the external carotid artery (ECA) in patients with traumatic maxillofacial fractures.
Materials and Methods
A total of 12 patients seen between March 2010 and December 2014 were included in this study. All subjects met the following criteria: 1) presence of maxillofacial fracture; 2) continuous blood loss from oronasal bleeding; and 3) EVT to stop bleeding. Various clinical factors were recorded for each patient and the correlations between those factors and clinical outcome (Glasgow Outcome Scale, GOS) were evaluated.
Results
Four patients were injured in traffic accidents, five in falls, and three by assaults. Mean initial Glasgow Coma Scale (GCS) was 6.9 ± 2.1 and the lowest hemoglobin measured was mean 6.3 ± 0.9 g/dL. GOS at discharge was 4 in five patients, 3 in three patients, and 1 (death) in four patients. GOS on follow-up (mean 13.7 months) was 5 in two patients, 4 in three patients, and 3 in three patients. Initial GCS (p = 0.016), lowest systolic blood pressure (p = 0.011), and lowest body temperature (p = 0.012) showed a significant positive correlation with good clinical outcomes. The number of units of red blood cells transfused (p = 0.030), the number of units of fresh frozen plasma transfused (p = 0.013), and the time from arrival to groin puncture (p < 0.001) showed significant negative correlation with good clinical outcomes.
Maxillofacial fractures are frequently seen in clinical practice; the most common cause is blunt facial trauma due to traffic accidents, followed by assault, falls, suicide attempts and industrial accidents.3) This type of fracture may lead to life-threatening bleeding from the branches of the external carotid artery (ECA), which is associated with airway obstruction and hemorrhagic shock. However, there are very few recommendations for treatment of ECA bleeding. Nasal packing or tamponade with balloon catheters is frequently ineffective because there is only soft tissue and no solid wall to stop massive bleeding in the oronasal cavity.2)6) Thus, endovascular treatment (EVT) was introduced in the 1980s as an alternative treatment option.2)5)6)7) The purpose of this study was to report our preliminary experience with using EVT for life-threatening bleeding from branches of the ECA in patients with traumatic maxillofacial fractures.
This retrospective study was approved by the institutional review board of our institution, and the requirement for informed consent was waived. Between March 2010 and December 2014, a total of 12 patients who met the following criteria were included in this study: 1) presence of maxillofacial fractures; 2) continuous blood loss from oronasal bleeding; and 3) EVT to stop bleeding due to uncontrolled hemostasis by other rescue strategies such as compression (packing), coagulation, tying-off, or nasal cavity tamponade (balloon catheter). Patients with other identified bleeding foci, such as splenic rupture, liver laceration, pelvic bone fractures, and mesentery tears, were excluded.
Clinical factors (age, sex, laterality of the fracture, main bleeder, cause of injury, initial Glasgow Coma Scale [GCS], lowest systolic blood pressure [SBP] before EVT, highest heart rate [HR], lowest body temperature [BT], acidosis, lowest hemoglobin [Hb] level, antiplatelet agents, anticoagulants, red blood cell [RBC] transfusion, fresh frozen plasma [FFP] transfusion, and time from arrival to groin puncture) and radiographic factors (combined skull fractures, pneumocephalus, intracranial hemorrhage, and severe brain injury) were reviewed and the correlation between these factors and clinical outcomes was evaluated. Clinical outcomes were evaluated using the Glasgow Outcome Scale (GOS) at their discharge and the latest clinical follow-up. Procedure-related complications were defined as new neurological deteriorations related to EVT or complications that needed additional procedures. Intracranial hemorrhage included traumatic epidural hematoma, subdural hematoma, parenchymal contusional hemorrhage, or subarachnoid hemorrhage. Severe brain injury was defined as diffuse cerebral edema with pseudo-subarachnoid hemorrhage on computed tomography (CT). Overall medical management and general care during the acute period were similar for all 12 patients.
Statistical analyses were performed using SPSS 22.0 (SPSS Inc., Chicago, IL, USA). Variables were expressed as mean ± SD or number of patients (%), as appropriate. Pearson's correlation coefficient was calculated to evaluate the significance of the linear relationship between the various factors and GOS at discharge. A p value < 0.05 was considered statistically significant.
Demographic and clinical characteristics of the patients are reported in Table 1. Four patients were injured in traffic accidents, five by falling, and three by assaults. Mean age of the 12 patients was 49.1 ± 18.2 years. Mean initial GCS was 6.9 ± 2.1 and the lowest hemoglobin level measured was mean 6.3 ± 0.9 g/dL. Main bleeding focus of all patients was from the third (pterygomaxillary) part of the internal maxillary artery (Fig. 1). Endovascular embolization was performed with polyvinyl alcohol (PVA) particles (particle size range 150-500 microns) in all patients. Technical success was achieved in 100% and there were no procedure-related complications. GOS at discharge was 4 in five patients, 3 in three patients, and 1 (death) in four patients. Mean clinical follow-up duration was 13.7 months. GOS at the follow-up was 5 in two patients, 4 in three patients, and 3 in three patients. Clinical data for each patient is provided in Table 2.
There were significant correlations between 6 clinical factors and GOS at discharge (Fig. 2). Initial GCS (Pearson correlation coefficient [r] = 0.677; p = 0.016), lowest systolic blood pressure (r = 0.702; p = 0.011), and lowest body temperature (r = 0.695; p = 0.012) showed significant positive correlations with GOS at discharge. The number of units of RBC transfused (r = -0.624; p = 0.030), the number of units of FFP transfused (r = -0.691; p = 0.013), and the time from arrival to groin puncture (r = -0.831; p < 0.001) showed significant negative correlations with GOS at discharge.
Six factors showed significant correlations with clinical outcomes. We argue that the most meaningful factor is the time from arrival to groin puncture because this suggests that a more rapid transition to EVT resulted in better clinical outcomes (higher GOS score). Poorer initial GCS, lower SBP, and lower BT are predictive of a poorer clinical outcome. In addition, it is not surprising that the greater the amount of RBC and FFP transfused, the poorer the patient's clinical condition and therefore the worse their clinical outcome. Based on our findings, rapid transition to EVT could achieve better clinical outcomes than struggling with other rescue strategies, such as compression (packing), coagulation, tying off, or nasal cavity tamponade (balloon catheter) to stop massive bleeding from branches of the ECA in patients with traumatic maxillofacial fractures.
The definition of life-threatening hemorrhage has not been well established in the literature. Life-threatening hemorrhage is reported as occurring secondary to blunt facial trauma in 0-24.4% of patients,4) reflecting the lack of a standardized definition due to large variance. In the present study, we defined life-threatening hemorrhage as oronasal bleeding uncontrollable by the general rescue strategies listed above. Controlling blood loss is an important part of trauma care. Superficial vessels may be compressed or tied off and fractures reduced or splinted to minimize blood loss. However, massive oronasal bleeding from maxillofacial fractures can be difficult to control. Bleeding typically results from multiple comminuted fracture fragments that lacerate the periosteum, mucosa and one or more major vessels. Traumatic facial bleeding originates from hard and soft tissue either individually or simultaneously, adding to the difficulty of targeting the exact source of bleeding.1) In addition, the exact source of bleeding is difficult to define as the vascular supply of the midface involves branches of the external and internal carotid arteries. The most commonly disrupted vessels include branches of the ECA, usually the internal maxillary artery, and particularly its intraosseous branches.3) In cases of severe bleeding from the oral cavity, tight oronasal packing is frequently ineffective because there is only soft tissue, and no solid wall to stop massive bleeding in the oronasal cavity.6)8)9)
While general rescue strategies such as compression (packing), coagulation, tying off, or nasal cavity tamponade (balloon catheter) are applied in our institution to treat profuse oronasal bleeding from maxillofacial fractures, physicians seem to feel that there are no other treatment options for patients struggling with hypovolemic shock due to profuse oronasal bleeding from their maxillofacial fractures other than active shock management. Open bony reduction usually cannot be performed because of unstable vital signs. There is risk associated with a critical patient undergoing general anesthesia, and it is difficult to perform reduction of the fracture bone in the presence of massive bleeding and severe tissue swelling.10) Neurointerventionists therefore suggested endovascular embolization of the branches of the ECA as an alternative treatment option. After applying ECA successfully in one patient,5) we were motivated to perform EVT in other patients with profuse oronasal bleeding from maxillofacial fractures. EVT has the advantages of indicating the bleeding points, allowing access near the bleeding points, controlling multiple bleeders, and not requiring general anesthesia.6) However, EVT may also result in soft tissue necrosis, blindness, facial nerve palsy, and migration of the embolus into the intracranial arteries. However, these disadvantages may not be considered immediately important in the context of patients with life-threatening bleeding.
One limitation of our study was its retrospective design with a very small number of included patients. Furthermore, there was not a control group for comparison of the effects of EVT with other currently-used treatment methods. Three patients did not receive EVT during the same period because their clinical condition, especially their vital signs, were not good enough to allow them to be moved to our angiosuite for EVT. All three of these patients died in the emergency department. In summary, EVT should be considered a treatment option in patients with massive oronasal bleeding from maxillofacial fractures, and should be performed as rapidly as possible.
References
1. Ardekian L, Rosen D, Klein Y, Peled M, Michaelson M, Laufer D. Life-threatening complications and irreversible damage following maxillofacial trauma. Injury. 1998; 5. 29(4):253–256. PMID: 9743742.

2. Bynoe RP, Kerwin AJ, Parker HH 3rd, Nottingham JM, Bell RM, Yost MJ, et al. Maxillofacial injuries and life-threatening hemorrhage: treatment with transcatheter arterial embolization. J Trauma. 2003; 7. 55(1):74–79. PMID: 12855884.
3. Chen CC, Jeng SF, Tsai HH, Liliang PC, Hsieh CH. Life-threatening bleeding of bilateral maxillary arteries in maxillofacial trauma: report of two cases.? J Trauma. 2007; 10. 63(4):933–937. PMID: 17110893.

4. Harris T, Rice S, Watts B, Davies G. The emergency control of traumatic maxillofacial haemorrhage. Eur J Emerg Med. 2010; 8. 17(4):230–233. PMID: 20603900.

5. Kim DY, Hyun DK, Park H, Chung J. Endovascular treatment of life-threatening bleeding of bilateral maxillary arteries in a patient with multiple facial bone fractures - a case report. J Korean Neurotraumatol Soc. 2011; 11. 7:108–111.
6. Komiyama M, Nishikawa M, Kan M, Shigemoto T, Kaji A. Endovascular treatment of intractable oronasal bleeding associated with severe craniofacial injury. J Trauma. 1998; 2. 44(2):330–334. PMID: 9498506.

7. Sakamoto T, Yagi K, Hiraide A, Takasu A, Kinoshita Y, Iwai A, et al. Transcatheter embolization in the treatment of massive bleeding due to maxillofacial injury. J Trauma. 1988; 6. 28(6):840–843. PMID: 3385830.

8. Schaitkin B, Strauss M, Houck JR. Epistaxis: medical versus surgical therapy: a comparison of efficacy, complications, and economic considerations. Laryngoscope. 1987; 12. 97(12):1392–1396. PMID: 3683049.
9. Wang L, Vogel DH. Posterior epistaxis: comparison of treatment. Otolaryngol Head Neck Surg. 1981; Nov-Dec. 89(6):1001–1006. PMID: 6801578.

10. Yang WG, Tsai TR, Hung CC, Tung TC. Life-threatening bleeding in a facial fracture. Ann Plast Surg. 2001; 2. 46(2):159–162. PMID: 11216612.

Fig. 1
Selective angiography of the external carotid artery demonstrated contrast extravasation (indicated [A] by arrow heads in a 77-year-old female and [B] by a circle in a 62-year-old male) in the third (pterygomaxillary) part of the internal maxillary artery indicated by a white arrow). IMA = internal maxillary artery.
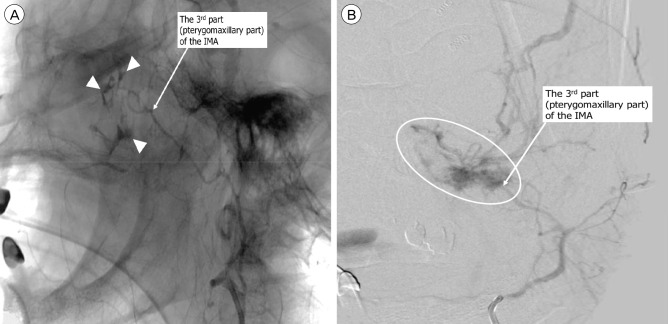
Fig. 2
Pearson's correlation coefficients were calculated and showed the significance of the linear relationship between Glasgow Outcome Scale at discharge and the various factors, such as (A) initial Glasgow Coma Scale, (B) lowest systolic blood pressure, (C) lowest body temperature, (D) red blood cells transfusion, (E) fresh frozen plasma transfusion, and (F) the time from arrival to groin puncture.
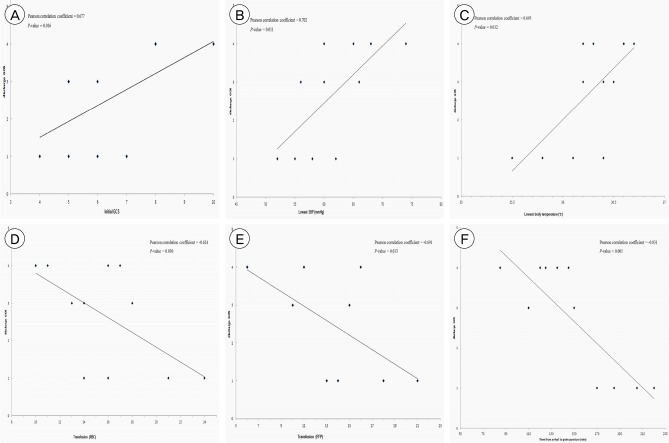
Table 1
Demographic and clinical characteristics of the patients
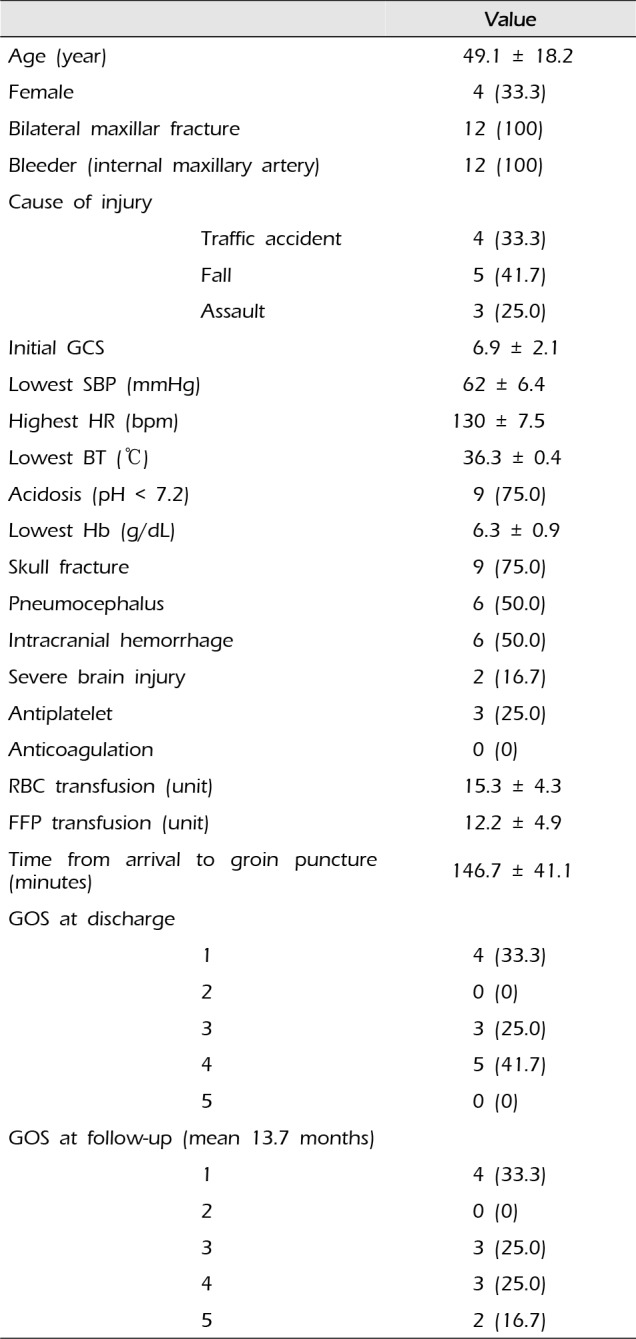
Table 2
Clinical data of all the patients
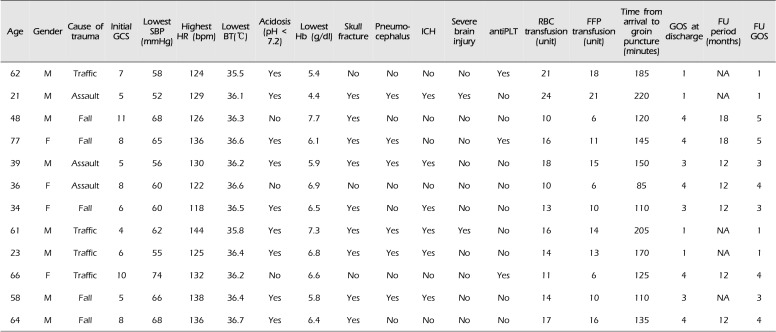
antiPLT = antiplatelet agents; BT = body temperature; F = female; FFP = fresh frozen plasma; FU = follow-up; GCS = Glasgow Coma Scale; GOS = Glasgow Outcome Scale; Hb = hemoglobin; HR = heart rate; ICH = intracranial hemorrhage; M = male; NA = not applicable; RBC = red blood cell; SBP = systolic blood pressure




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download