Abstract
The incidence of intracerebral hemorrhage in those aged 45–84 years is 0.3-0.5%. In people over 80 years of age, this incidence increases 25-fold compared with that of the total population. The most common causes of spontaneous intracerebral hemorrhage in the younger population are vascular malformation, aneurysm, and overuse of drugs. In contrast, common causes in the elderly include hypertension, tumors, and coagulation disorders. Here, we present a case involving a 72-year-old male patient who, without any of these predisposing conditions, was admitted to the hospital with spontaneous intracerebral hemorrhage and showed signs of multifocal intracerebral hemorrhage during his stay. We conclude that spontaneous intracerebral hemorrhage can occur without any predisposing factors, and can lead to a patient's death. Therefore, the possibility of recurrent spontaneous intracerebral hemorrhage must be considered in patients with primary spontaneous intracerebral hemorrhage.
Spontaneous intracerebral hemorrhage is a very dangerous disease as it is associated with severe mortality and morbidity.11) The degree of hemorrhage is an important factor when assessing prognosis. There have been few reports of enlargement of hematomas following hemorrhage in hospitalized patients, which would worsen their prognosis.7) Several risk factors and underlying conditions are associated with spontaneous brain hemorrhage. In younger populations, these include vascular malformation, aneurysm, and overuse of drugs. In contrast, in the elderly, spontaneous intracerebral hemorrhage is associated with hypertension, tumors, vasculopathy, and coagulation defects. Cerebral amyloid angiopathy is a common risk factor that applies to everyone, regardless of age.16) Observation of these risk factors and predispositions signify a need for greater precautions in patients with brain hemorrhage. Here, we describe a 72-year-old man without any specific risk factors who nonetheless developed recurrent cerebral hemorrhages on three occasions in a single hospitalization.
A 72-year-old right-handed man with a history of severe headaches was admitted to our hospital emergency room. He was a non-smoker, not an alcoholic, and had no history of vascular disease. In 1995, he underwent an appendectomy and was diagnosed with gastritis. When his headaches were investigated in another hospital, the remnant of a transitory minimal subarachnoid hemorrhage was observed. A few years later, follow-up brain computed tomography (CT) to investigate the causes of recurrent headaches also revealed a right frontal chronic subdural hemorrhage. The time of diagnosis of these conditions is unknown.
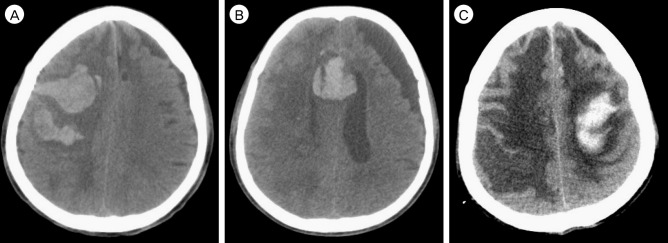
On the morning of his visit to the emergency room, the patient had stuttering speech and left-sided hemiparesis. On arrival, his blood pressure was 137/87 and his pulse rate was 104. He was drowsy and the motor grade of his left side was zero. Subsequent brain CT showed a right frontal lobe intracerebral hemorrhage (Fig. 1A), and intracerebral hemorrhage catheter insertion was performed on the same day. Brain CT conducted two days after the procedure showed resolution of the intracerebral hemorrhage. The patient's vital signs were stable, but he was still drowsy. Immediately after the operation, he suddenly became stuporous. However, brain CT performed to assess the cause of this mental change did not show signs of intracerebral hematoma. However, he was placed under observation. Approximately three hours after the operation, the patient returned to his previous state of drowsiness. He could not follow instructions but had some verbal output.
Seven days after the operation, the patient suffered respiratory difficulty and was intubated. No specific reasons could be identified that had aggravated his condition. Thus, another brain CT was performed and a new intracerebral hemorrhage was observed at the septum pellucidum (Fig. 1B). On the 20th postoperative day, hyponatremia was observed in a routine laboratory test. On the 24th postoperative day, the patient's serum sodium level fell further. A brain CT scan conducted to identify the cause of this revealed a third intracerebral hemorrhage at the left frontal subcortical area (Fig. 1C). The patient finally passed away due to pneumonia and severe cerebral edema approximately five weeks after his first hemorrhage.
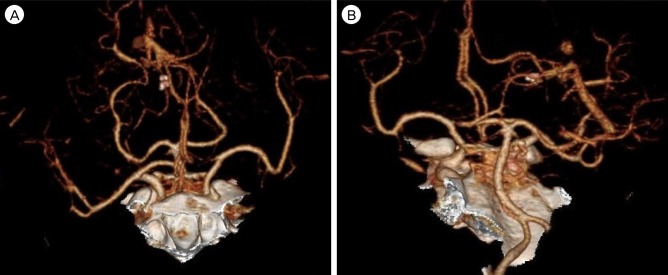
To determine the causes of these recurrent cerebral hemorrhages, we conducted laboratory examinations to exclude conditions such as autoimmune etiology and blood coagulopathy. A multidisciplinary test in cooperation with the Rheumatology Department showed positive anti-cardiolipin antibody (IgG), which suggests the possibility of antiphospholipid syndrome. Subsequent tests were performed for DNA, RNP, SM, RO, RA, lupus anticoagulant, anti-beta2GP1 IgM/IgG, ANA IFA and centromere auto-antibodies, but all yielded negative findings. This ruled out possible autoimmunity. Laboratory tests for blood coagulopathy included assessments of fibrinogen, D-Dimer, thrombin time, antithrombin III, and FDP. The D-dimer and FDP levels were 662 ng/mL (range 0-243) and 7.87 µg/mL(range 0-5), respectively. It was thought that these results were because of the poor general condition, rather than blood coagulopathy as results of other tests were normal. No definite abnormality was found in CT angiography (Fig. 2). Due to the patient's condition, we could not perform further evaluation of other vascular anomalies, such as venous sinus thrombosis or dural arteriovenous fistula. The patient's lung condition was very poor due to pneumonia and pleural effusion. We therefore discussed the possibility of other diseases with the Radiology Department, who advised that the possibility of other disease was very low.
We also performed a pathologic examination on brain tissues acquired from the intracerebral hemorrhage catheter. Nothing of interest was found except for fragmented brain tissue with hemorrhage (Fig. 3). The Pathology Department explained that tissue staining was not necessary since it was could be reasonably deemed that the tissues of the brain were normal.
Currently known risk factors for intracerebral hemorrhage include arterial hypertension, male sex, antiplatelet or anticoagulant agents, alcoholism, blood disorders, old age, liver problems, tumor bleeding, and trauma.1) A history of previous cerebrovascular accident (CVA) is associated with hemorrhagic transformation during the first month in up to 43% of cases.9) Furthermore, carotid endarterectomy is known to cause intracerebral hemorrhage.4) Hemorrhage related to carotid-cavernous fistula occurs in 3% of patients.21) In addition, a case of spontaneous intracerebral hematoma with cortical venous drainage after spontaneous relief of initial symptoms caused by traumatic carotid-cavernous fistula has been reported.6)
Metabolic disturbances, such as diabetic ketoacidosis, are not known causes of intracerebral hematomas. However, bilateral posterior temporal hematomas in a patient with juvenile diabetic ketoacidosis (DM type I, 11-year-old girl) have been reported.2) Indeed, metabolic syndrome may be another possible risk factor for intracerebral hemorrhage.17) The pathophysiology underlying this risk is hypertension induced by insulin resistance, leading to sympathetic nervous system activation and elevated inflammatory status signified by CRP elevation.17)
Several other conditions have been related to intracerebral hematoma. Idiopathic thrombocytopenic purpura (ITP) is found in patients with intracerebral hemorrhage. There is great variation in the latency between the onset of ITP and the occurrence of intracerebral hemorrhage, but it is generally accepted that intracerebral hemorrhage occurs commonly within the first month after ITP onset.14) Pheochromocytoma is observed in less than 0.2% of patients who have sustained high blood pressure.8)18) Cerebral hemorrhage occurs rarely in Churg-Strauss syndrome (CSS), but fatal intraventricular and subarachnoid hemorrhages (SAH) have been reported in patients with pathologically proven CSS.5) Moreover, Liou et al.15) reported a case involving a CSS patient who exhibited bilateral hemispheric involvement with multiple cerebral hemorrhage.
However, in our case, the patient had only a minimal subarachnoid hemorrhage and chronic subdural hemorrhage history. He had no hypertension or diabetes mellitus, no known vascular disease, and he was not on any medication. At admission, his blood pressure was controlled well using a calcium channel blocker. Results of all other tests were uneventful.
Hypertensive intracerebral hemorrhage usually occurs once and rarely recurs.12) Moreover, hemorrhages in hospitalized patients are rarely extended.7) Multiple intracerebral hemorrhage has been reported in just 2% of intracerebral hemorrhages.19) In Korea, the intracerebral hemorrhage recurrence rate is 5.4%.3) Other studies have reported rates that vary between 2.7 and 11.7%. The interval between hemorrhages ranges from 13.1 to 22.3 months, while 0.9% of cases had re-bleeding.13) Further reports suggest that re-bleeding occurs in 2.8% of hemorrhagic stroke patients and 3.4% of intracerebral hemorrhage patients.16) Several case reports suggest that cerebral amyloid angiopathy is the cause hemorrhage.10)
In this regard, diagnosis and treatment were especially difficult in our case because bleeding occurred three times without any preceding risk factors. As mentioned above, a pathology test was performed to eliminate the possibility of cerebral amyloid angiopathy, but it came back negative.
Single spontaneous intracerebral hemorrhages primarily occur deep in the brain, most commonly in the putamen, thalamus, or cerebellum, followed next often in subcortical white matter.22) In contrast, multiple simultaneous intracerebral hemorrhages mostly appear in the bilateral thalami and putamen.20) However, our patient had three consecutive intracerebral hemorrhages in uncommon locations: the cortex and septum pellucidum. Usually, these sites bear a lower possibility of bleeding compared with the basal ganglia, pons, and thalamus.
As discussed, hypertension is the most common cause of intracerebral hemorrhage. Other causes include primary or metastatic brain cancer, cerebral amyloid angiopathy, old age, anticoagulant or antiplatelet medication history, and trauma. However, as shown by the case we describe here, intracerebral hemorrhage can occur without any specific underlying conditions. Importantly, this case indicates that the possibility of re-bleeding should not be overlooked. Observing deterioration of the patient in physical and neurologic examinations, laboratory results, and chest x-ray, we attempted to identify reasons other than re-bleeding, as there was only a slight chance that bleeding had occurred again. Such procedures to elucidate the causes of aggravation of the patient's condition delayed application of appropriate treatment. Therefore, the possibility of re-bleeding should not be neglected even during hospitalization.
References
1. Ariesen MJ, Claus SP, Rinkel GJ, Algra A. Risk factors for intracerebral hemorrhage in the general population: a systematic review. Stroke. 2003; 8. 34(8):2060–2065. PMID: 12843354.
2. Atluru VL. Spontaneous intracerebral hematomas in juvenile diabetic ketoacidosis. Pediatr Neurol. 1986; May-Jun. 2(3):167–169. PMID: 3150280.

3. Bae H, Jeong D, Doh J, Lee K, Yun I, Byun B. Recurrence of bleeding in patients with hypertensive intracerebral hemorrhage. Cerebrovasc Dis. 1999; Mar-Apr. 9(2):102–108. PMID: 9973653.

4. Bernstein M, Fleming JF, Deck JH. Cerebral hyperperfusion after carotid endarterectomy: a cause of cerebral hemorrhage. Neurosurgery. 1984; 7. 15(1):50–56. PMID: 6472594.

5. Chang Y, Kargas SA, Goates JJ, Horoupian DS. Intraventricular and subarachnoid hemorrhage resulting from necrotizing vasculitis of the choroid plexus in a patient with Churg-Strauss syndrome. Clin Neuropathol. 1993; Mar-Apr. 12(2):84–87. PMID: 8097441.
6. d'Angelo VA, Monte V, Scialfa G, Fiumara E, Scotti G. Intracerebral venous hemorrhage in "high-risk" carotid-cavernous fistula. Surg Neurol. 1988; 11. 30(5):387–390. PMID: 3055384.
7. Fujii Y, Takeuchi S, Sasaki O, Minakawa T, Tanaka R. Multivariate analysis of predictors of hematoma enlargement in spontaneous intracerebral hemorrhage. Stroke. 1998; 6. 29(6):1160–1166. PMID: 9626289.

8. Guerrero MA, Schreinemakers JM, Vriens MR, Suh I, Hwang J, Shen WT. Clinical spectrum of pheochromocytoma. J Am Coll Surg. 2009; 12. 209(6):727–732. PMID: 19959041.

9. Hornig CR, Dorndorf W, Agnoli AL. Hemorrhagic cerebral infarction--a prospective study. Stroke. 1986; Mar-Apr. 17(2):179–185. PMID: 3515635.

10. Ishii N, Nishihara Y, Horie A. Amyloid angiopathy and lobar cerebral haemorrhage. J Neurol Neurosurg Psychiatry. 1984; 11. 47(11):1203–1210. PMID: 6502178.

11. Juvela S. Risk factors for impaired outcome after spontaneous intracerebral hemorrhage. Arch Neurol. 1995; 12. 52(12):1193–1200. PMID: 7492294.

12. Kumar NS, Neeraja V, Raju CG, Padala RK, Kumar TA. Multiple spontaneous hypertensive intracerebral hemorrhages. J Stroke Cerebrovasc Dis. 2015; 1. 24(1):e25–e27. PMID: 25541521.
13. Lee KS, Bae HG, Yun IG. Recurrent intracerebral hemorrhage due to hypertension. Neurosurgery. 1990; 4. 26(4):586–590. PMID: 2330079.

14. Lee MS, Kim WC. Intracranial hemorrhage associated with idiopathic thrombocytopenic purpura: report of seven patients and a meta-analysis. Neurology. 1998; 4. 50(4):1160–1163. PMID: 9566417.

15. Liou HH, Liu HM, Chiang IP, Yeh TS, Chen RC. Churg-Strauss syndrome presented as multiple intracerebral hemorrhage. Lupus. 1997; 6(3):279–282. PMID: 9104737.

16. Mauriño J, Saposnik G, Lepera S, Rey RC, Sica RE. Multiple simultaneous intracerebral hemorrhages: clinical features and outcome. Arch Neurol. 2001; 4. 58(4):629–632. PMID: 11295994.
17. Park YK, Yi HJ, Lee YJ, Cho H, Chun HJ, Oh SJ. The relationship between metabolic syndrome (MetS) and spontaneous intracerebral hemorrhage (ICH). Neurol Sci. 2013; 9. 34(9):1523–1528. PMID: 23263735.

18. Petri BJ, van Eijck CH, de Herder WW, Wagner A, de Krijger RR. Phaeochromocytomas and sympathetic paragangliomas. Br J Surg. 2009; 12. 96(12):1381–1392. PMID: 19918850.

19. Seijo M, Ucles A, Gil-Nagel A, Balseiro J, Calandre L. [Multiple cerebral hematomas: review of 7 cases]. Rev Neurol. 1996; 5. 24(129):549–553. PMID: 8681171.
20. Seo JS, Nam TK, Kwon JT, Park YS. Multiple spontaneous simultaneous intracerebral hemorrhages. J Cerebrovasc Endovasc Neurosurg. 2014; 6. 16(2):104–111. PMID: 25045650.

21. Turner DM, Vangilder JC, Mojtahedi S, Pierson EW. Spontaneous intracerebral hematoma in carotid-cavernous fistula. Report of three cases. J Neurosurg. 1983; 10. 59(4):680–686. PMID: 6886790.
22. Yen CP, Lin CL, Kwan AL, Lieu AS, Hwang SL, Lin CN. Simultaneous multiple hypertensive intracerebral haemorrhages. Acta Neurochir (Wien). 2005; 4. 147(4):393–399. discussion 399. PMID: 15605198.

Fig. 1
CT scans of the three hemorrhages. (A) the first hemorrhage in the right frontal cortex. (B) the second hemorrhage in the septum pellucidum, which occurred 7 days after the first hemorrhage. (C) the third hemorrhage in the left fronto-parietal cortex, which occurred 17 days after the second hemorrhage. CT = computed tomography.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download