Abstract
Purpose
We investigated the correlations of serum total testosterone (TT) levels with body composition and physical fitness parameters in patients with erectile dysfunction (ED) to know the best exercise for testosterone deficiency.
Materials and Methods
Eighty-seven ED patients underwent serum TT assessment as well as body composition and basic exercise testing. The bioelectrical impedance analysis was used to assess body composition. Seven types of basic exercise tests were used to determine physical fitness. Correlations between serum TT levels and body composition/physical function parameters were evaluated using partial correlation analyses. A serum TT cut-off value was obtained for the parameters significantly correlated with serum TT levels.
Results
The subjects had a mean serum TT level of 342.1 ng/dL. Among the body composition parameters, body and abdominal fat percentages showed statistically significant negative correlations with serum TT levels. Among the basic exercise test parameters, only the cycle ergometer test for cardiorespiratory fitness showed a statistically significant positive correlation with serum TT levels.
In a recent publication [1], we demonstrated that exercise improves the efficacy of testosterone treatment for patients with testosterone deficiency (TD), and prolongs the duration of the treatment effect after treatment cessation. In that study, we administered an exercise program that included 20 minutes of aerobic exercise followed by 10 minutes of whole-body stretching, 30 minutes of strength exercise, and 10 minutes of aerobic exercise. This training program was performed three times per week to maximize the effects of exercise on treatment. However, it is unknown what type of exercise would best improve symptoms and serum testosterone levels in patients with TD. Other previous studies have mainly focused on the effects of acute vigorous resistive strengthening exercises on serum testosterone levels, and few studies have examined the effects of long‑term chronic resistive strengthening exercise on serum testosterone levels [23456]. Better knowledge regarding which type of exercise is more effective in improving TD would aid in maximizing the treatment efficacy by better tailoring the existing treatment regimen. Moreover, a maximally effective treatment regimen could reduce the risk of adverse treatment effects by shortening the length of treatment.
To determine the best exercise program, it is important to first understand which body composition and physical fitness parameters are most closely related to serum testosterone levels, and then introduce exercise programs targeting these factors. Therefore, the present study aimed to investigate relationships between serum testosterone levels and body composition and physical fitness parameters in patients with erectile dysfunction (ED). Based on these results, we sought to derive a physical exercise that is more effective in treating TD.
Among patients who had their serum testosterone levels measured after visiting the hospital for ED between January 2014 and June 2015, data were analyzed for 87 patients who additionally underwent body composition and basic exercise testing. All men were included consecutively in this study, were at least 35 years of age, and complained of an inability to obtain or maintain a sufficiently firm erection for sexual intercourse for more than six months. The patients who answered ‘yes’ to one or more questions on the physical activity readiness questionnaire were excluded except for those patients whose blood pressure was well controlled by antihypertensive drugs. Patients who experienced difficulty in participating in exercise testing due to joint problems and had a history of treatment for hypogonadism were excluded from this study. Blood samples were obtained before 11 a.m., and serum testosterone levels were measured by radioimmunoassay. Body mass index (BMI) was calculated and bioelectrical impedance analysis (BIA) was used to assess body composition, including skeletal muscle mass, fat mass, lean body mass, body fat percentage, abdominal fat percentage, and visceral fat mass. The patients performed seven types of basic exercise testing: cardiorespiratory fitness (cycle ergometer test), flexibility (sit-and-reach test), muscular endurance (curl-up test), muscular strength (grip test, vertical jump test), agility (whole body reaction test), and balance (one-leg Stance test) (Fig. 1).
The correlations between serum testosterone levels and the results from the body composition and basic exercise tests were investigated. Partial correlation analyses with age as a confounding factor were performed. For parameters determined to be significantly correlated with serum testosterone levels, the serum testosterone cut-off value at which significant differences occurred in those parameters was calculated by multiple independent t-tests after dividing 87 patients into two groups based on each serum testosterone level.
The mean age of the subjects was 57 years (range, 36–73 years), and the mean serum total testosterone level was 342.1 ng/dL (range, 83.4–1,030.0 ng/dL). There were 55 patients with no underlying diseases (63.2%), while 10 patients had more than 1 underlying diseases (11.5%). For each disease, there were 17 patients with lower urinary tract symptoms suggestive of benign prostatic hyperplasia (19.5%), 8 patients with hypertension (9.2%), 8 patients with hyperlipidemia (9.2%), 7 patients with diabetes mellitus (8.0%), and 4 patients with other diseases (4.6%) (Table 1). The mean BMI was 25.50 kg/m2 (range, 18.50–34.20 kg/m2) and was negatively correlated with serum testosterone levels (Pearson correlation coefficient (r)=−0.200); however, the correlation was not statistically significant (p=0.082).
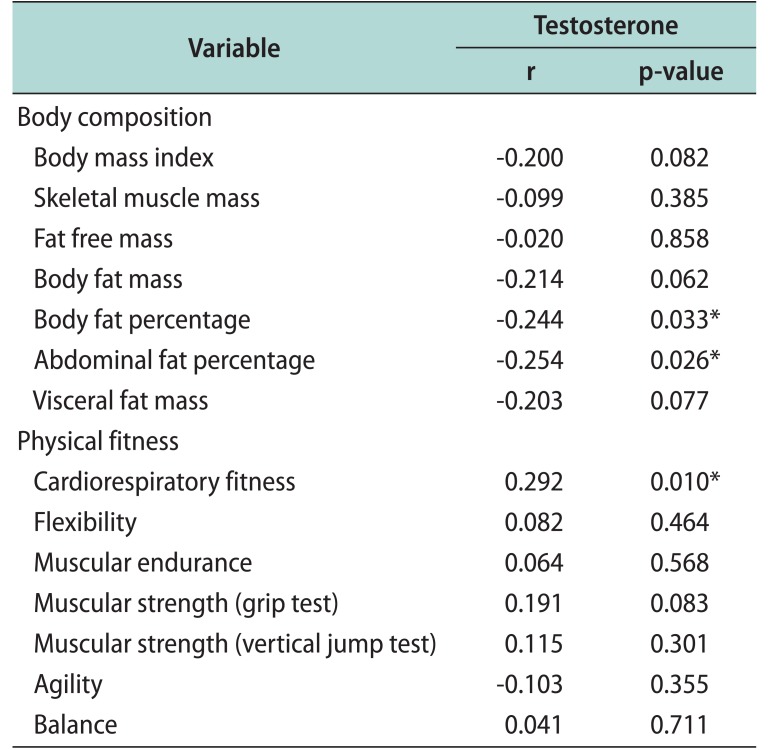
Among the body composition test parameters, body and abdominal fat percentages showed statistically significant negative correlations with serum total testosterone levels (r=−0.244, p=0.033; r=−0.254, p=0.026, respectively). In addition, body and visceral fat masses showed marginally significant negative correlations with serum total testosterone levels (r=−0.214, p=0.062; r=−0.203, p=0.077, respectively) (Table 2). Fat free mass and skeletal muscle mass were not significantly correlated with serum testosterone levels, and the correlations were smaller compared to that between fatrelated parameters and serum testosterone levels (r=−0.020, p=0.858; r=−0.099, p=0.385, respectively). Most of the basic exercise test parameters were positively correlated with serum total testosterone levels. However, only the cycle ergometer test for cardiorespiratory fitness showed a statistically significant positive correlation with serum total testosterone levels (r=0.292, p=0.010) (Table 2). The cut-off value for the serum total testosterone level at which significant differences in body fat percentage, abdominal fat percentage, and cardiorespiratory fitness occurred was determined to be 384.9 ng/dL.
Only limited data regarding the effects of regular exercise on serum testosterone—the most important factor in TD—are available, and existing studies have mainly focused on determining whether resistive strengthening exercise increases serum testosterone levels. Although acute vigorous resistive strengthening exercise is speculated to increase serum testosterone levels, few studies have examined the effects of long‑term chronic resistive strengthening exercise on serum testosterone levels [2345]. Smilios et al [6] demonstrated that an acute elevation in testosterone is not always observed after vigorous resistive strengthening exercise; the total amount of exercise appears to be more important in increasing testosterone levels. A few additional studies have combined other types of exercises with resistive strengthening exercise and examined their effects on serum testosterone. Heufelder et al [7] conducted a 52‑week, double‑blind clinical trial, examining the effects of supervised exercise, with or without testosterone treatment, on components of metabolic syndrome and newly diagnosed type 2 diabetes in hypogonadal men. Serum testosterone was significantly improved after 52 weeks of supervised exercise without testosterone treatment. In a recent study, we developed an exercise program consisting of aerobic exercise, whole-body stretching, and resistive strength exercises [1], and determined that the exercise group with testosterone treatment had significantly higher serum testosterone levels compared to those in the control group. Furthermore, the increased serum testosterone levels were relatively well maintained in the group that continued to perform the exercises after cessation of the testosterone treatment. Regular exercise that significantly reduces BMI, induces an elevation in serum testosterone levels via the following mechanism: the serum insulin level [8], oxidative stress [910], and conversion of testosterone to estradiol by aromatase in adipose tissue are reduced [11], and the muscular and plasma dihydrotestosterone concentrations are directly increased [11213].
These findings clearly imply that exercise is deeply associated with the serum testosterone level and thus with ED, a classic symptom of TD [14]. However, it is unclear which type of exercise would be better for improving symptoms and the endocrinologic status of patients with TD. Identifying the specific body composition or physical fitness parameters closely related to serum testosterone levels is beneficial for TD treatment development, as serum testosterone levels may be more effectively elevated through exercises targeting the related body composition and physical fitness parameters. Therefore, we administered seven simple exercise tests to measure physical fitness, as in Whaley et al [15], and conducted BIA testing to determine body composition. Body and abdominal fat percentages were found to have statistically strong, significant negative correlations with serum testosterone levels. Other fat-related parameters (body fat mass and visceral fat mass) also showed relatively high r-values and borderline statistical significance. On the other hand, muscle mass parameters, such as skeletal muscle mass and fat-free mass, lacked statistical significance and had near-zero r-values. These findings attest to the close association between serum testosterone levels and fat, suggesting that exercise targeting reductions in fat would effectively increase the serum testosterone level. With regard to physical fitness parameters, only cardiorespiratory fitness was found to have a significant positive correlation with serum testosterone levels, implying that exercise targeting cardiorespiratory fitness would be helpful in increasing serum testosterone levels. Thus, aerobic exercise, which reduces fat mass and improves cardiorespiratory fitness, is expected to be more beneficial than strengthening exercise in increasing serum testosterone levels.
In a study by Khoo et al [16] of 90 sedentary male patients with obesity (BMI>27.5 kg/m2, waist circumference> 90 cm), aerobic exercise of more than 200 minutes per week over 24 weeks significantly increased the International Index of ED-5 score and testosterone levels in serum. They also reported that the exercise reduced urinary tract symptoms and improved the quality of life. In addition, Hayes et al [17] confirmed that, through a 12-week high-intensity interval training using aerobic exercise, the testosterone levels in serum increased in 22 lifelong sedentary male patients (average age: 62 years). In these studies, not all participants were TD patients whose testosterone levels were below 350 ng/dL. Furthermore, the number of participants was small, with a limited period. Nevertheless, these studies demonstrate that there is a possibility of increasing serum testosterone levels through aerobic exercise.
It has already been reported from numerous studies that the testosterone level threshold can vary depending on the age or symptom. Zitzmann et al [18] showed that a decrease in libido or vitality can begin to manifest at 15 nmol/L, which is higher than the commonly used TD standard of 12.1 nmol/L, while depression or diabetes occurs at a significantly high frequency in men whose serum testosterone levels are below 10 nmol/L. In cases of ED adjusting for smoking or other metabolic syndrome-related factors, a significant influence is reported on testosterone levels that were decreased to below 8 nmol/L [18]. In this study, the threshold of serum testosterone level used for analysis was also confirmed with respect to body composition and physical fitness factors that showed a significant correlation with serum testosterone levels. The cut-off value for the serum testosterone level inducing significant differences in body fat percentage, abdominal fat percentage, and cardiorespiratory fitness, was determined to be 384.9 ng/dL. This value is close to the current clinical standard for TD treatment (350 ng/dL) [1920], strongly implying that TD is related to body fat percentage, abdominal fat percentage, and cardiorespiratory fitness.
The present study has a few limitations. First, the study was exploratory in nature and involved a relatively small sample size, which may have reduced the statistical power. Further, some patients with normal testosterone levels and only relatively healthy patients with ED who were able to undergo the basic exercise testing were included in the present study. Another limitation is that the BIA testing used in the present study has relatively lower accuracy in measuring body composition compared to dual-energy x-ray absorptiometry [21]. Lack of measuring other hormone levels, such as dehydroepiandrosterone, insulin-like growth factor, free testosterone, and sex hormone-binding globulin, which can give more accurate result was another limitation. Finally, erectile function scores and TD symptom scores were not included in the correlation analyses. Despite these limitations, the present study provides valuable information as the first explorative study identifying the specific body composition or physical fitness parameters closely related to serum testosterone levels and suggesting that aerobic exercise may be more beneficial than resistive strengthening exercises in enhancing testosterone treatment for TD patients with ED. In the future, large-scale prospective studies examining the impact of the type of exercise on changes in serum testosterone levels are needed to substantiate the findings of the present study.
In patients with ED, serum total testosterone levels were negatively correlated with body and abdominal fat percentages, and positively correlated with cardiorespiratory fitness. Thus, serum total testosterone levels, which are closely related to erectile function, may be increased in patients with ED by reducing fat percentage and improving cardiorespiratory fitness. These results suggest that aerobic exercise, which reduces fat and improves cardiorespiratory fitness, would be more beneficial for patients with TD and ED than strengthening exercises.
References
1. Cho DY, Yeo JK, Cho SI, Jung JE, Yang SJ, Kong DH, et al. Exercise improves the effects of testosterone replacement therapy and the durability of response after cessation of treatment: a pilot randomized controlled trial. Asian J Androl. 2017; 19:602–607. PMID: 27427553.

2. Häkkinen K, Pakarinen A, Newton RU, Kraemer WJ. Acute hormone responses to heavy resistance lower and upper extremity exercise in young versus old men. Eur J Appl Physiol Occup Physiol. 1998; 77:312–319. PMID: 9562359.
3. Linnamo V, Pakarinen A, Komi PV, Kraemer WJ, Häkkinen K. Acute hormonal responses to submaximal and maximal heavy resistance and explosive exercises in men and women. J Strength Cond Res. 2005; 19:566–571. PMID: 16095404.

4. Kraemer WJ, Marchitelli L, Gordon SE, Harman E, Dziados JE, Mello R, et al. Hormonal and growth factor responses to heavy resistance exercise protocols. J Appl Physiol (1985). 1990; 69:1442–1450. PMID: 2262468.

5. Häkkinen K, Pakarinen A, Kraemer WJ, Newton RU, Alen M. Basal concentrations and acute responses of serum hormones and strength development during heavy resistance training in middle-aged and elderly men and women. J Gerontol A Biol Sci Med Sci. 2000; 55:B95–B105. PMID: 10737684.
6. Smilios I, Pilianidis T, Karamouzis M, Tokmakidis SP. Hormonal responses after various resistance exercise protocols. Med Sci Sports Exerc. 2003; 35:644–654. PMID: 12673149.

7. Heufelder AE, Saad F, Bunck MC, Gooren L. Fifty-two-week treatment with diet and exercise plus transdermal testosterone reverses the metabolic syndrome and improves glycemic control in men with newly diagnosed type 2 diabetes and subnormal plasma testosterone. J Androl. 2009; 30:726–733. PMID: 19578132.

8. Kumagai H, Zempo-Miyaki A, Yoshikawa T, Tsujimoto T, Tanaka K, Maeda S. Lifestyle modification increases serum testosterone level and decrease central blood pressure in overweight and obese men. Endocr J. 2015; 62:423–430. PMID: 25753766.

9. Sajjadian F, Roshangar L, Hemmati A, Nori M, Soleimani-Rad S, Soleimani-Rad J. The effect of BSO-induced oxidative stress on histologic feature of testis: testosterone secretion and semen parameters in mice. Iran J Basic Med Sci. 2014; 17:606–612. PMID: 25422755.
10. Chigurupati S, Son TG, Hyun DH, Lathia JD, Mughal MR, Savell J, et al. Lifelong running reduces oxidative stress and degenerative changes in the testes of mice. J Endocrinol. 2008; 199:333–341. PMID: 18701639.

11. Lee HK, Lee JK, Cho B. The role of androgen in the adipose tissue of males. World J Mens Health. 2013; 31:136–140. PMID: 24044108.

12. Aizawa K, Iemitsu M, Maeda S, Mesaki N, Ushida T, Akimoto T. Endurance exercise training enhances local sex steroidogenesis in skeletal muscle. Med Sci Sports Exerc. 2011; 43:2072–2080. PMID: 21502890.

13. Sato K, Iemitsu M, Aizawa K, Mesaki N, Fujita S. Increased muscular dehydroepiandrosterone levels are associated with improved hyperglycemia in obese rats. Am J Physiol Endocrinol Metab. 2011; 301:E274–E280. PMID: 21285401.

14. Hehemann MC, Kashanian JA. Can lifestyle modification affect men’s erectile function. Transl Androl Urol. 2016; 5:187–194. PMID: 27141445.

15. Whaley MH, Brubaker PH, Otto RM, Armstrong LE. ACSM's Guidelines for exercise testing and prescription. 7th ed. Philadelphia: Lippincott Willians & Wilkins;2006.
16. Khoo J, Tian HH, Tan B, Chew K, Ng CS, Leong D, et al. Comparing effects of low- and high-volume moderate-intensity exercise on sexual function and testosterone in obese men. J Sex Med. 2013; 10:1823–1832. PMID: 23635309.

17. Hayes LD, Herbert P, Sculthorpe NF, Grace FM. Exercise training improves free testosterone in lifelong sedentary aging men. Endocr Connect. 2017; 6:306–310. PMID: 28515052.

18. Zitzmann M, Faber S, Nieschlag E. Association of specific symptoms and metabolic risks with serum testosterone in older men. J Clin Endocrinol Metab. 2006; 91:4335–4343. PMID: 16926258.

19. Corona G, Vignozzi L, Sforza A, Maggi M. Risks and benefits of late onset hypogonadism treatment: an expert opinion. World J Mens Health. 2013; 31:103–125. PMID: 24044106.

20. Saad F, Yassin A, Haider A, Doros G, Gooren L. Elderly men over 65 years of age with late-onset hypogonadism benefit as much from testosterone treatment as do younger men. Korean J Urol. 2015; 56:310–317. PMID: 25874045.

21. Hamada Y. Objective Data Assessment (ODA) methods as nutritional assessment tools. J Med Invest. 2015; 62:119–122. PMID: 26399333.

Fig. 1
Basic exercise testing to evaluate individual physical fitness: (A) cardiorespiratory fitness by cycle ergometer test; (B) flexibility by sit-and-reach test; (C) muscular endurance by curl-up test; (D) muscular strength by grip test; (E) agility by whole body reaction test; and (F) balance by one-leg stance test.
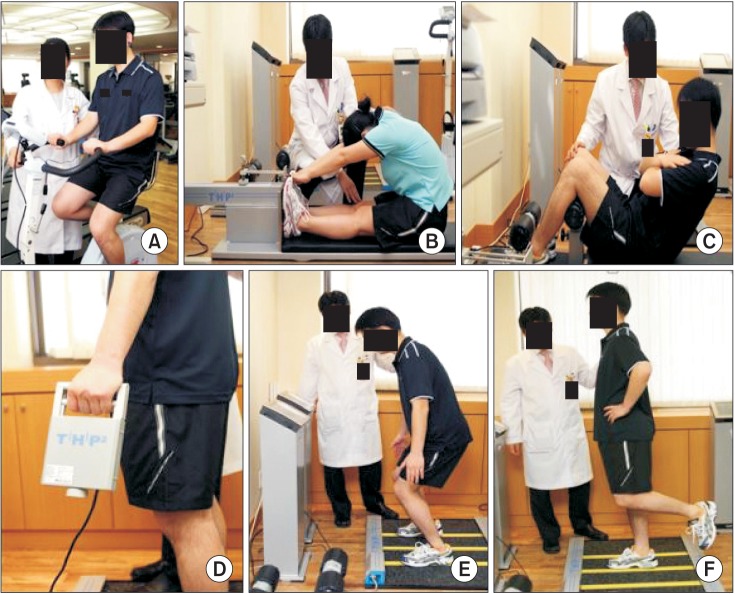
Table 1
Baseline characteristics (n=87)
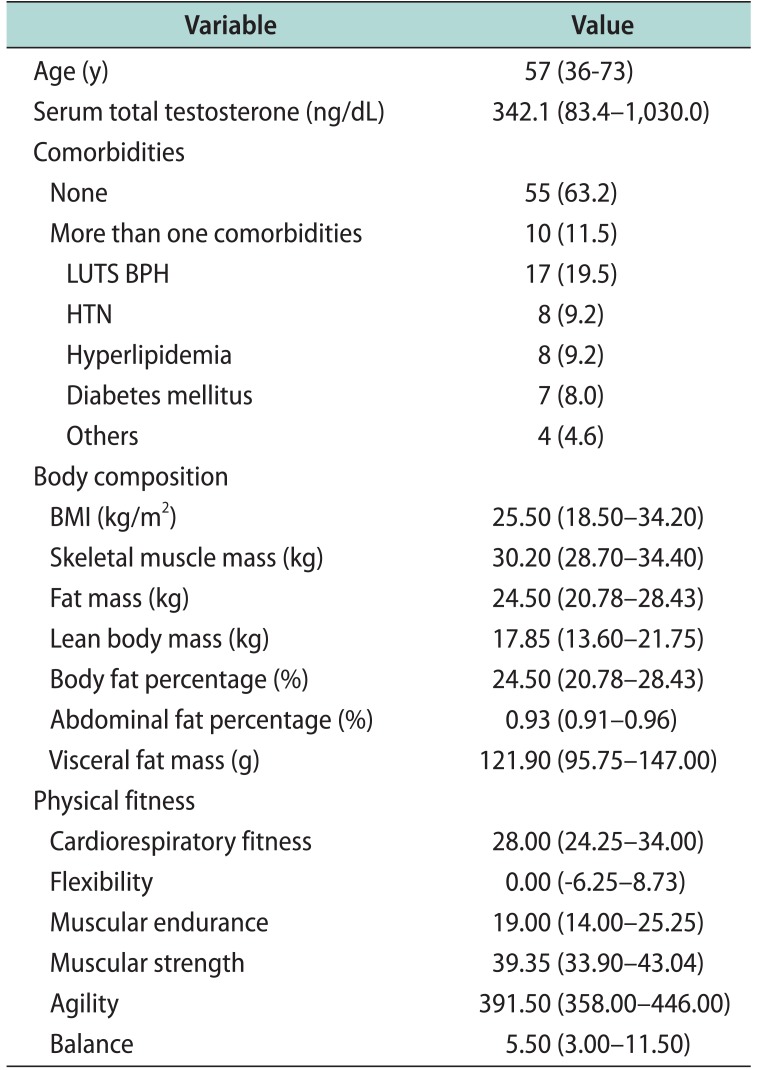
Table 2
Correlations between serum testosterone level and body composition and physical fitness parameters




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download