Abstract
Purpose
The purpose of this study was to investigate the effects of the hydro-alcoholic extract of Rhus coriaria seeds on the reproductive system of nicotinamide-streptozotocin-induced type-2 diabetic mice.
Materials and Methods
In this experimental study, 56 male Naval Medical Research Institute mice were randomly divided into seven groups (n=8): control; diabetic mice; diabetic mice administered glibenclamide (0.25 mg/kg); diabetic mice who received the hydro-alcoholic extract of R. coriaria seeds (200 and 400 mg/kg groups); and normal mice who received this extract (200 and 400 mg/kg groups). Diabetes was induced by intraperitoneal administration of streptozotocin (65 mg/kg) 15 minutes after an injection of nicotinamide (120 mg/kg). Then, glibenclamide and the above mentioned extract were administered orally for 28 consecutive days. Twenty-four hours after the last treatment, serum samples, the testes, and the cauda epididymis were removed immediately for hormonal, testis morphology, and sperm parameter assessments.
Results
Body and testicular weight, sperm count and viability, and serum luteinizing hormone, follicle-stimulating hormone and testosterone levels were significantly lower in the diabetic mice (p<0.05). The diabetic mice treated with 400 mg/kg of the hydro-alcoholic extract of R. coriaria seeds recovered from these reductions (p<0.05). Further, glibenclamide alleviated hormonal and sperm count depletion in diabetes-induced mice (p<0.05).
Diabetes mellitus is an important metabolic disorder in human society; according to recent estimations, 280 million people across the world will be affected by this disorder by the year 2030. Two main types of diabetes have been identified and classified as type-1 and type-2 diabetes. About 90% to 95% of all diabetic patients suffer from type-2 diabetes [1]. Further, male reproductive dysfunction is an important fertility disorder in human society. Couple infertility is estimated to be about one in seven couples worldwide, and approximately 50% of these cases have been attributed to the male [2]. Sexual and sperm quality dysfunction is an established complication of diabetes [3]. It has been reported that diabetes mellitus can induce infertility in 90% of male diabetic patients [4]. Further, the prevalence of secondary hypogonadism in men with type-2 diabetes is at least 25%, and 4% of them have primary hypogonadism [5]. Moreover, investigators have reported that a remarkable reduction of testicular weight and abnormal spermatogenesis have been observed in diabetic rats [4]. Hypothalamo-pituitary-testicular axis damage and decreased pituitary response to gonadotropin releasing hormone (GnRH) have been reported in several investigations of diabetic animal models. Further, diabetic human studies have reported a blunted response of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) secretion to GnRH [6]. In fact, many of the sexual organ disorders in diabetic animals result from oxidative stress and an increase in reactive oxygen species (ROS) generation in mitochondria [7]. Hence, many natural and industrial antioxidant agents have been recommended to inhibit oxidative-stress-induced reproductive hormonal imbalance and infertility [8]. As medicinal plants have several effective compounds and relatively low adverse effects as compared to synthetic drugs, traditional medicinal plants have been used for the treatment of different health disorders [9]. One of these medicinal plants is Rhus coriaria (sumac), which belongs to the Anacardiaceae family. This plant has small fruits with a sepia color and grows especially in the Northern mountainous areas of Iran [10]. This herb is traditionally use in the treatment of diarrhea, hemorrhoids, and animal bite pain [11]. According to the available literature, R. coriaria is a rich source of flavonoids and tannins. Flavonoids are polyphenols that have antitumor, hypoglycemic, and free radical scavenging as well as antioxidant properties [12]. In previous studies, it has been demonstrated that the hydro-alcoholic extract of R. coriaria seeds increases the activity of antioxidant enzymes such as superoxide dismutase and catalase, which leads to improved type-2 diabetes induced in rats [13,14]. Thus, taking in to account the high prevalence of type-2 diabetes and its negative effects on the reproductive system and the presence of antioxidant components in R. coriaria to improve this complication, the present study was conducted to evaluate the effects of the hydro-alcoholic extract of R. coriaria seeds extract on serum LH, FSH, and testosterone levels and sperm count in nicotinamide-streptozotocin (NA-STZ)-induced type-2 diabetic mice.
In this experimental study, fresh seeds of R. coriaria (sumac) were obtained from a credible green grocery of Ahvaz city and authenticated scientifically by the Botany Department of Ahvaz Jundishapur University, Ahvaz, Iran. Voucher specimens of the plant are deposited in the Herbarium of University of Tehran (Tehran, Iran. No. PRL/JH 22/03). After desiccation, the seeds under shade, were crushed and powdered using an electric grinder.
Three hundreds gram of acquired R. coriaria seeds' powder was soaked in 1,200 mL of a distilled water-ethanol mixture (60 : 40) and maintained for 72 hours at room temperature. After filtering the combination with What-man No. 1 filter paper, it was centrifuged at 3,500 rpm for 20 minutes. Eventually, the supernatant was taken away and dried at 37℃, and the acquired somewhat solid mass was refrigeratored until use [15].
Tifty-six adult male Naval Medical Research Institute mice (30~35 g) were obtained from the animal house of Ahvaz Jundishapur University of Medical Sciences and were kept in cages under standard laboratory conditions (temperature: 20℃±4℃ with a 12 hours-12 hours light/dark cycle); they were permitted access to normal commercial chow and tap water. The protocols for all animals were prepared in accordance with the standards for laboratory animal care accepted by the National Research Council of the National Academic.
Type-2 diabetes was induced by an intraperitoneally injection of STZ (65 mg/kg) (dissolved in citrate buffer, pH 4.5) (Sigma-Aldrich Co., St Louis, MO, USA) 15 minutes after a single-dose administration of NA (120 mg/kg) (dissolved in normal saline) (Sigma-Aldrich Co.). Progression of diabetes was confirmed by blood glucose level assaying at 72 hours after the NA-STZ injection. Ultimately, mice with fasting blood glucose levels of more than 250 mg/dL were considered diabetic, and the other animals with lower serum glucose levels were removed from the following experiments [16].
Animals were randomly divided into seven groups (n=8 in each group): Group I: control; Group II: diabetic mice; Group III: diabetic mice administered glibenclamide (0.25 mg/kg) [17] as standard drug for diabetes; Groups IV and V: diabetic mice that received the hydro-alcoholic extract of R. coriaria seeds (200 and 400 mg/kg, respectively); Groups VI and VII: normal mice that received the same extract (200 and 400 mg/kg, respectively) [13]. Glibenclamide and the R. coriaria seed extract were administered orally for 28 consecutive days [18].
Twenty-four hours after the last treatment, the animals were anesthetized by ether; blood samples were collected by cardiac puncture and centrifuged at 3,500 rpm for 20 minutes. Then, serum samples were kept at -80℃ until hormonal measurements were performed. The testes and cauda epididymis of all animals were removed instantly. Then, testicular weight, width, length, and volume in each group were assessed [19]. Further, the testis volume was analyzed using the following formula: volume=length×width×height×0.52 [20].
For determination the sperm count, the cauda epididymis of all animals was separated and transferred a into Petri dish containing 1.5 mL 0.9% normal saline, and minced. After squeezing these slices, spermatozoa were vented to the surrounding fluid. In order to estimate the sperm count, a drop of the Petri dish solution was transferred onto Neubauer hemocytometer lam (HBG Company, Giessen, Germany) (Tiefe Depth Profondeur 0.100 mm; area: 0.0025 mm2). Then, the exact sperm count was manually assayed in white blood cell chambers by light microscopy (Olympus Light Microscope, Tokyo, Japan). Finally, data were expressed as the count of sperm per milliliter [21].
Sperm viability was assessed by the staining of eosin 1% (Merck Chemical Co., Darmstadt, Germany) used for the evaluation of live (unstained) and dead (red stained) sperm. After leaving this stain on each blood cell chamber of the Neubauer hemocytometer for 30 seconds, we manually counted the total number of live spermatozoa within two minutes and expressed it as the sperm viability percentage [21].
Hormonal assessment was performed for calculating the serum levels of FSH, LH, and testosterone by using the enzyme-linked immunosorbent assay (ELISA) method with commercial assay kits (DRG Instruments GmbH, Marburg, Germany); the hormone detection sensitivity per assay tube of each kit was 1.27, 0.856 mIU/mL, and 0.287 nmol/L, respectively.
As shown in Table 1, after the NA-STZ diabetes induction, body weight decreased in these diabetic mice when compared to the control group (p<0.05). However, treatment with hydro alcoholic extract and glibenclamide could recover the body weight after reduction induced by diabetes in mice (p<0.05).
Testis volume did not show significant changes in all animals of the control and diabetes groups. However, induction of type-2 diabetes reduced the testicular weight remarkably (p<0.05), the administration of the extract (200 and 400 mg/kg), and glibenclamide increased the testicular weight when compared to the diabetes-only group (p<0.05) (Table 1).
The results showed that epididymal sperm count and viability were reduced in diabetic animals significantly (p<0.05). Further, administration of the R. coriaria seed extract (400 mg/kg) improved these factors in diabetes-treated mice (p<0.05), and glibenclamide (0.25 mg/kg) increased the sperm count as compared to the diabetes-only group (p<0.05) (Table 2, Fig. 1, 2).
As presented in Table 2 and Fig. 3, 4, 5, diabetes induction resulted in a significant decrease in serum LH, FSH, and testosterone levels as compared to the control group (p<0.05). Administration of the R. coriaria seed extract (400 mg/kg) improved the serum levels of gonadal hormones and testosterone in diabetic animals (p<0.05). Further, serum levels of LH, FSH, and testosterone increased significantly in glibenclamide-treated diabetic mice (p<0.05).
The results obtained in the present study showed that, diabetes induced by NA-STZ decreased the animals' body and testis weight. Further, administration of the hydro-alcoholic extract of R. coriaria seeds improved these changes in diabetic mice. Hence, our results were consistent with many studies that reported a testicular and body weight decline in diabetic cases [22]. This might be a consequence of protein wasting as a result of the unavailability of carbohydrates for producing energy [23]. Further, this study demonstrated that oral administration of the hydro-alcoholic extract of R. coriaria seeds were ameliorated body and reproductive organ weight in diabetic mice.
Previous research has revealed low sperm count and viability in rodents with diabetes [22]. Sperm analysis in the present study showed a significant decrease in sperm count and viability in diabetic animals. Further, administration of the R. coriaria seed extract restored the epididymal sperm count and sperm viability to control levels. Simultaneous administration of NA and STZ produces a chronic diabetic situation like type-2 diabetes. NAcan protect Langerhans islets from STZ-induced cytotoxic damages. There is some evidence that suggests that, oxidative stress plays a key role in the pathogenesis of diabetes-induced male reproductive defects [24]. Further, some studies indicate that STZ injection cause sperm depletion and abnormalities due to several mechanisms such as ROS overproduction. Excessive generation of ROS leads to testicular oxidative damage, germ cell apoptosis, and sperm count and viability depletion. The spermatozoa membrane is highly sensitive to oxidative stress since it contains rich polyunsaturated fatty acid [25]. Moreover, the ROS, particularly H2O2, might diffuse across the membrane into the cells and inhibit some antioxidant enzyme activities, which might in turn lead to a decrease in the formation of adenosine three phosphate and sperm motility. Moreover, it has been reported that antioxidant agent treatments have a protective effect on sperm viability [26]. Some investigators have also reported that R. coriaria has an antioxidant effect due to its components such as flavonoids [27]. Therefore, on the basis of the results of the present study, we conclude that the R. coriaria seed extract can increase sperm count and viability via improved ROS generation and antioxidant defense.
In this study, we found that serum testosterone, LH, and FSH levels, diminished in NA-STZ-induced diabetic mice; these finding are in agreement with previous reports [28]. Further, it has been established that regulation of testicular function is mainly done FSH and LH. FSH plays the main role in the development and function of Sertoli cells; therefore, any decrease in this hormone activity leads to Sertoli cell dysfunction, which in turn results in decreased sperm production [29]. Testosterone is produced by Leydig cells in response to LH under the control of the hypothalamic-pituitary-testis axis. Furthermore, a decrease in the number and function of Leydig cells in diabetic reduces the serum testosterone level. It is known that changes in glucose and insulin metabolism are associated with the production of LH and FSH. Further, insulin plays a positive role in Leydig cell function and testosterone production. It has also been reported that, a reduced insulin level leads to a remarkable decrease in the number and function of Leydig cells as well as the serum testosterone level [30]. Therefore, in this research, administration of the R. coriaria seed extract recovered the serum LH, FSH, and testosterone levels in diabetic mice. Gonadal hormone and testosterone play a prominent role in spermatogenesis [31]. Hence, it could be suggested that the applied extract in the present study improved sperm parameters via enhanced serum levels of gonadotropins and testosterone. Although the exact functional mechanism of the R. coriaria seed extract with respect to the increase in the serum levels of these hormones is unclear, some researchers have reported that flavonoids and saponins have positive effects on androgen bioavailability [27,32]. Saponins can increase testosterone production after the effect of pituitary LH secretion on Leydig cells [33]. Further, it has been reported that the R. coriaria seed is rich in saponins and flavonoids; [32] therefore, it can be inferred that the increasing effect of its extract on serum gonadotropin and testosterone levels is related to the presence of these agents in this plant.
Glibenclamide belongs to the category of sulfonylurea antidiabetic drugs that promote pancreatic islet insulin secretion in type-2 diabetes mellitus. It has been demonstrated that this drug can increase the serum levels of gonadotropin and, testosterone, and the spermatozoa generation in diabetic animals through improved antioxidant defense and increased antioxidant enzyme activities [24]. Therefore, the present results of glibenclamide administration are in accordance with the finding of previous studies. Hence, it can be concluded that this drug ameliorates reproductive system dysfunction in diabetic mice via its antioxidant properties.
In conclusion, this study revealed that the hydro-alcoholic extract R. coriaria seeds improved the reproductive dysfunction induced by type-2 diabetes, by increasing the serum LH, FSH, and testosterone levels; sperm count; and sperm viability. Further, administration of glibenclamide as a standard antidiabetic drug had very impressive effects on the improvement of the sperm count and the serum levels of LH, FSH, and testosterone in NA-STZ-induced diabetic mice. Moreover, on the basis of the present results, it can be suggested that the extract applied in this research acted via its antioxidant agents, but further investigations are required to clarify the precise mechanism of these agents.
Figures and Tables
 | Fig. 1Effect of hydro-alcoholic extract of Rhus coriaria (S) seeds on sperm count in normal and nicotinamide-streptozotocin-induced diabetic mice. Values are presented as mean±standard error; n=8. GLI: glibenclamide, S: Sumac. ap<0.05 compared with the diabetic group, bp<0.05 compared with the control group. |
 | Fig. 2Effect of hydro-alcoholic extract of Rhus coriaria (S) seeds on sperm viability in normal and nicotinamide-streptozotocin-induced diabetic mice. Values are presented as mean±standard error; n=8. GLI: glibenclamide, S: Sumac. ap<0.05 compared with the diabetic group, bp<0.05 compared with the control group. |
 | Fig. 3Effect of hydro-alcoholic extract of Rhus coriaria (S) seeds on serum luteinizing hormone (LH) levels in normal and nicotinamide-streptozotocin-induced diabetic mice. Values are presented as mean±standard error; n=8. GLI: glibenclamide, S: Sumac. ap<0.05 compared with the diabetic group, bp<0.05 compared with the control group. |
 | Fig. 4Effect of hydro-alcoholic extract of Rhus coriaria (S) seeds on serum follicle-stimulating hormone (FSH) levels in normal and nicotinamide-streptozotocin-induced diabetic mice. Values are presented as mean±standard error; n=8. GLI: glibenclamide, S: Sumac. ap<0.05 compared with the diabetic group, bp<0.05 compared with the control group. |
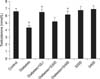 | Fig. 5Effect of hydro-alcoholic extract of Rhus coriaria (S) seeds on serum testosterone level in normal and nicotinamide-streptozotocin-induced diabetic mice. Values are presented as mean±standard error; n=8. GLI: glibenclamide, S: Sumac. ap <0.05 compared with the diabetic group, bp<0.05 compared with the control group. |
Table 1
Effect of hydro-alcoholic extract of Rhus coriaria (Sumac) seeds on body weight and testis morphology of normal and nicotinamide-streptozotocin-induced diabetic mice

ACKNOWLEDGEMENTS
This paper is labeled Student Research Project No. 92S23 and was financially supported by the Student Research Committee of Ahvaz Jundishapur Medical Sciences University, Ahvaz, Iran.
References
1. Tiwari AK, Madhusdana JR. Diabetes mellitus and multiple therapeutic approaches of phytochemicals: present status and future prospects. Curr Sci. 2002; 83:30–38.
2. Brown DB, Merryman DC, Rivnay B, Houserman VL, Long CA, Honea KL. Evaluating a novel panel of sperm function tests for utility in predicting intracytoplasmic sperm injection (ICSI) outcome. J Assist Reprod Genet. 2013; 30:461–477.

3. Bose R, Adiga SK, D'Souza F, Salian SR, Uppangala S, Kalthur G, et al. Germ cell abnormalities in streptozotocin induced diabetic mice do not correlate with blood glucose level. J Assist Reprod Genet. 2012; 29:1405–1413.

4. Amaral S, Moreno AJ, Santos MS, Seiça R, Ramalho-Santos J. Effects of hyperglycemia on sperm and testicular cells of Goto-Kakizaki and streptozotocin-treated rat models for diabetes. Theriogenology. 2006; 66:2056–2067.

5. Dandona P, Dhindsa S. Update: hypogonadotropic hypogonadism in type 2 diabetes and obesity. J Clin Endocrinol Metab. 2011; 96:2643–2651.

6. Baccetti B, La Marca A, Piomboni P, Capitani S, Bruni E, Petraglia F, et al. Insulin-dependent diabetes in men is associated with hypothalamo-pituitary derangement and with impairment in semen quality. Hum Reprod. 2002; 17:2673–2677.

7. Kianifard D, Sadrkhanlou R, Hasanzadeh S. The histological, histomorphometrical and histochemical changes of testicular tissue in the metformin treated and untreated streptozotocin-induced adult diabetic rats. Vet Res Forum. 2011; 2:13–24.
8. Sahreen S, Khan MR, Khan RA, Shah NA. Effect of Carissa opaca leaves extract on lipid peroxidation, antioxidant activity and reproductive hormones in male rats. Lipids Health Dis. 2013; 12:90.

9. Kumar S, Rashmi Kumar D. Evaluation of antidiabetic activity of Euphorbia hirta Linn In streptozotocin induced diabetic mice. Indian J Nat Prod Resour. 2010; 1:200–203.
10. Gharaei A, Khajeh M, Ghaffari M, Choopani A. Iranian Rhus coriaria (sumac) Essential Oils Extraction. TEOP. 2013; 16:270–273.
11. Shabbir A. Rhus coriaria linn, a plant of medicinal, nutritional and industrial importance: a review. J Anim Plant Sci. 2012; 22:505–512.
12. González R, Ballester I, López-Posadas R, Suárez MD, Zarzuelo A, Martínez-Augustin O, et al. Effects of flavonoids and other polyphenols on inflammation. Crit Rev Food Sci Nutr. 2011; 51:331–362.

13. Anwer T, Sharma M, Khan G, Iqbal M, Ali MS, Alam MS, et al. Rhus coriaria ameliorates insulin resistance in non-insulin-dependent diabetes mellitus (NIDDM) rats. Acta Pol Pharm. 2013; 70:861–867.
14. Mohammadi S, Montasser Kouhsari S, Monavar Feshani A. Antidiabetic properties of the ethanolic extract of Rhus coriaria fruits in rats. Daru. 2010; 18:270–275.
15. Ahangarpour A, Mohammadian M, Dianat M. Antidiabetic effect of hydroalcholic urticadioica leaf extract in male rats with fructose-induced insulin resistance. Iran J Med Sci. 2012; 37:181–186.
16. Perez-Gutierrez RM, Damian-Guzman M. Meliacinolin: a potent α-glucosidase and α-amylase inhibitor isolated from Azadirachta indica leaves and in vivo antidiabetic property in streptozotocin-nicotinamide-induced type 2 diabetes in mice. Biol Pharm Bull. 2012; 35:1516–1524.
17. Padee P, Nualkaew S, Talubmook C, Sakuljaitrong S. Hypoglycemic effect of a leaf extract of Pseuderanthemum palatiferum (Nees) Radlk. in normal and streptozotocin-induced diabetic rats. J Ethnopharmacol. 2010; 132:491–496.

18. Singh A, Singh SK. Evaluation of antifertility potential of Brahmi in male mouse. Contraception. 2009; 79:71–79.

19. Ahangarpour A, Oroojan AA, Heydari H. Effect of hydro-alcoholic extract of dorema aucheri on serum levels of testosterone, FSH and sperm count in nicotinamide-STZ-induced diabetic rat models. ZUMS J. 2013; 21:22–31.
20. Sakamoto H, Saito K, Oohta M, Inoue K, Ogawa Y, Yoshida H. Testicular volume measurement: comparison of ultrasonography, orchidometry, and water displacement. Urology. 2007; 69:152–157.

21. Ahangarpour A, Oroojan AA, Radan M. Effect of aqueous and hydro-alcoholic extracts of lettuce (Lactuca sativa) seed on testosterone level and spermatogenesis in NMRI mice. Iran J Reprod Med. 2014; 12:65–72.
22. Novelli M, Canistro D, Martano M, Funel N, Sapone A, Melega S, et al. Anti-diabetic properties of a non-conventional radical scavenger, as compared to pioglitazone and exendin-4, in streptozotocin-nicotinamide diabetic mice. Eur J Pharmacol. 2014; 729:37–44.

23. Roy S, Rahaman N, Ahmed F, Metya S, Sannigrahi S. Naringenin attenuates testicular damage, germ cell death and oxidative stress in streptozotocin induced diabetic rats: naringenin prevents diabetic rat testicular damage. J Appl Biomed. 2013; 11:195–208.

24. Rabbani SI, Devi K, Khanam S. Protective role of glibenclamide against nicotinamide-streptozotocin induced nuclear damage in diabetic Wistar rats. J Pharmacol Pharmacother. 2010; 1:18–23.

25. Aitken RJ, Roman SD. Antioxidant systems and oxidative stress in the testes. Adv Exp Med Biol. 2008; 636:154–171.

26. Agarwal A, Aitken RJ, Alvarez JG. Studies on men's health and fertility. 1st ed. New York: Humana Press Publisher;2012. p. 489.
27. Kosar M, Bozan B, Temelli F, Baser KHC. Antioxidant activity and phenolic composition of sumac (Rhus coriaria L.) extracts. Food Chem. 2007; 103:952–959.

28. Abbasi Z, Tabatabaei SR, Mazaheri Y, Barati F, Morovvati H. Effects of sesame oil on the reproductive parameters of diabetes mellitus-induced male rats. World J Mens Health. 2013; 31:141–149.

29. Rossi P, Dolci S. Paracrine mechanisms involved in the control of early stages of Mammalian spermatogenesis. Front Endocrinol (Lausanne). 2013; 4:181.

30. Lin T, Haskell J, Vinson N, Terracio L. Characterization of insulin and insulin-like growth factor I receptors of purified Leydig cells and their role in steroidogenesis in primary culture: a comparative study. Endocrinology. 1986; 119:1641–1647.

31. Wdowiak A, Raczkiewicz D, Stasiak M, Bojar I. Levels of FSH, LH and testosterone, and sperm DNA fragmentation. Neuro Endocrinol Lett. 2014; 35:73–79.
32. Akrayi HFS, Abdullrahman ZFA. Screening in vitro and in vivo the antibacterial activity of Rhus coriaria extract against S. Aureus. IJRRAS. 2013; 15:390–397.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download