Abstract
Fibrothecomas are mesenchymal tumors deriving from the ovarian stromal and consisting of theca-like elements and fibrous tissue. They are common, but their malignant counterpart is extraordinarily rare. Classical malignant fibrothecomas are said to show four or more mitotic figures per 10 high power fields. We have experienced a rare case of malignant ovarian fibrothecoma in a 72-year-old postmenopausal woman. We report a case with brief review of literature.
Fibrothecomas are benign neoplasms which belong to the estrogenic group of sex cord tumors with differentiation in fibroblastic or theca cells. Fibroma is purely composed of mature fibroblastic cells producing abundant collagen and thecoma contains many cells resembling theca cells and/or lutein cells as well as a number of fibroblastic cells. However, in some instances, differentiation between fibroma and thecoma is often difficult, justifying use of the term 'fibrothecoma'. Malignant ovarian fibrothecoma is a rare entity.
Classical malignant fibrothecomas are said to show four or more mitotic figures per 10 high power fields (HPF). These tumors occur generally in older menopausal patient. Histologically, these tumors have a stromal origin and are composed of variable proportion of spindle cells forming collagen and theca cells containing lipids. They may be hormonally active and thus responsible for estrogenic manifestations.
We report a rare case of malignant ovarian fibrothecoma in a 72-year-old postmenopausal woman.
A 72-year-old gravida 3 para 2 postmenopausal woman with no significant medical history presented to our gynecology outpatient clinic with constipation and abdominal distention. No associated fever, vomiting, or localized abdominal pain occurred. The pelvic examination revealed a palpable non-tender pelvic mass. Transvaginal ultrasonogram shows a 11.3×11.7×10.2 cm sized well defined heterogenous echoic solid mass with some tiny cystic changes abutting on atrophic uterus in right adnexa (Fig. 1). The result of laboratory studies, including analysis of tumor markers, such as CA-125, CA19-9, squamous cell carcinoma related antigen were all within normal limits.
At laparotomy, there was no ascites and a lobulated bulging mass measuring 15 cm was observed on the right ovary. The left ovary, uterus, omentum, and pelvic lymph node were unremarkable. A frozen section of the ovarian mass revealed a sex-cord stromal tumor with nuclear atypia and mitosis. The patient underwent abdominal total hystrectomy, bilateral salpingo-oophorectomy, omentectomy and pelvic adhesiolysis. Her postoperative recovery was uneventful and she was discharged home on the 6th day of surgery.
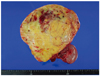
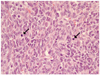
On gross examination, the right ovary was mostly replaced by a solid mass, measuring 16×16 cm, which is composed of yellow-gray solid firm mass with focal myxoid change (Fig. 2). Histological examination revealed the tumor. The tumor is composed of cellular, oval to spindle cells with pale cytoplasm and frequent mitosis (>4/10 HPF) (Fig. 3). The tumor cells are positive for vimentin and alpha-inhibin (Fig. 4). The ovarian tumor was stromal tumor, and diagnosed as a malignant fibrothecoma.
Fibrothecomas are mesenchymal tumors deriving from the ovarian stroma and consisting of theca-like elements and fibrous tissue. The term fibrothecoma has been used on neoplasms which are intermediate between theca cell tumor and fibroma. The vast majority of fibrothecomas behave in a benign fashion and malignant variants are exceedingly rare. Occasional cases of malignant thecoma have been reported [1], but there is doubt as to whether a true malignant variant of this neoplasm exists.
Because of its histologic resemblance to fibrosarcoma and sarcomatoid granulosa cell tumor, and since the precise distinction between fibromas and thecomas can be difficult and sometimes impossible to ascertain, Waxman et al. [1] proposed in 1979 that this type of tumor should be called "ovarian stromal sarcoma".
These tumors occur generally in older menopausal patient. However, some authors report 2 peaks of frequency: the first peak of onset is after menopause and the second is between 20 and 40 years. The occurrence of these tumors before the age of 20 is extremely rare [2]. The tumor is unilateral in 90 per cent of cases and with an average diameter of 6 cm. In 4.5 per cent of cases it may be more than 20 cm [3].
The reported incidence is between 0.4% and 8.0% of all ovarian tumors, and this wide variability can be attributed to the difficulty in differentiating fibrothecoma from ovarian fibromas or hyperplastic stromal nodules [4].
The clinical presentation of ovarian fibrothecoma is relatively nonspecific, namely pelvic pain and metrorrhagia. The clinical examination generally finds a solid, mobile tumor with a regular surface and a variable size [5]. Rarely, fibrothecomas present with endocrine manifestations related to hormonally active tumors. In these cases, estrogenic or even some rare androgenic manifestations are reported [6].
Even though the definitive diagnosis of fibrothecoma is histologic, Conte et al. [7] have described the presence of characteristic sonographic patterns of fibrothecoma. The presence of a homogenous echogenic pattern, with marked posterior acoustic absorption, is highly suggestive of a predominantly fibrous ovarian fibrothecoma; an echogenic pattern, with no posterior acoustic shadowing appears to be correlated to the presence of a mixed fibrothecoma; finally, the finding of a diffusely hypoechoic mass, with no posterior echo enhancement, is characteristic of pure ovarian thecomas. Sonographic findings, even though nonspecific, can provide the clinician with useful information which permits to detect these rare neoplasms preoperatively.
Mak et al. [8] have described the presence of characteristic computed tomography patterns of fibrothecoma. Fibrothecomas usually appear as a homogeneous solid tumor with varying degrees of enhancement. Calcification may be present and, as these tumors enlarge, myxoid or cystic degeneration may occur, resulting in a heterogeneous pattern. CA-125 serum levels are usually normal. Rarely, high serum levels are noted, and they become normal after the removal of tumor. Depending on the prevalence of cells or fibrous tissue, these tumors are pathologically classified as pure thecomas, fibrothecomas and predominantly fibrous fibrothecomas [4].
Macroscopically, fibrothecomas are very similar to fibromas. Microscopically, fibrothecomas are composed of intermingling bundles or anastomosing trabeculae of ovoid theca-like cells, with round and pale nuclei, containing lipid droplets and fibroblastic fusiform elements. Granulosa cells, separated by collagen bundles, have also been observed.
For Prat and Scully [9], the tumor is benign if there less than 3 mitosis per 10 HPF, and malignant fibrothecomas are said to show four or more mitotic figures per 10 HPF. Two clinical associations are worth to mention. The first is called Meigs syndrome. Meigs syndrome is a rare clinical entity characterized with ovarian benign tumor, ascites and hydrothorax. This syndrome is observed with 1%-10% of ovarian fibromas. Hydrothorax and ascites regress quickly after the ablation of the tumor [10]. The second association is called sclerosing peritonitis. Sclerosing peritonitis is a rare inflammatory process that leads to the deposition of a thick fibrous membrane on the peritoneum. Sclerosing peritonitis is a recognized complication of chronic ambulatory peritoneal dialysis [11]. It has also been linked to practolol and other β-blocking agent [12]. The serosa of the small bowel is the most frequently and, most severely affected site. Involvement is usually confined to the peritoneum, which may be thickened as much as 4-5 mm, but occasionally the muscularis is involved. Less commonly, the peritoneum of the large bowel and the visceral peritoneum of the pelvis and upper abdomen are involved. The pathogenesis of the peritoneal reaction in sclerosing peritonitis is unclear but may be hormonally related [13]. The pelvic mass or luteinized thecoma comprises a proliferation of ovarian spindle cells, which enlarge under the influence of progesterone. Sclerosing peritonitis in chronic ambulatory peritoneal dialysis patients has a high mortality rate, but data from patients with luteinized thecomas are more favorable [13]. With thecomas, this process appears to be self-limiting, requiring conservative management.
Treatment of these ovarian tumors is based on surgery. The surgical intention is always that of a complete resection of the mass, or even en bloc resection with adjacent or infiltrated organs and structures. For all patients with a potentially resectable tumor, the surgical access we prefer is a long midline ventral incision with the patients in the supine position. An additional transverse incision was sometimes adopted in huge tumors in order to provide maximum exposure. Factors influencing survival outcome of tumorous mass include, location, tumor size, adjacent involvement, mitotic index. However, it is generally agreed that the most important factor influencing survival outcome of malignancies is the surgeon's skill in resecting the tumor [14]. Considering the difficulty of achieving complete resection with clear margins in giant tumors, especially those with involvement of critical abdominal vessels as vena cava, aorta, and mesenteric vessels, a lager tumor size was found to be a negative prognostic factor. If the surgeon is able to achieve safe tumor resection without microscopic residual disease, patient may expect long-term survival, regardless of tumor size. In fact, it has showed that the status of gross surgical margins is the most important factor influencing prognosis. When neoplastic infiltration of adjacent organs is suspected, en bloc resection with the involved organs, such as stomach, intestines, pancreas, kidney, liver, spleen, adrenal gland, is always preferable [15].
In conclusion, We have experienced a rare case of malignant ovarian fibrothecoma in a 72-year-old postmenopausal woman. We report a case with brief review of literature.
Figures and Tables
Fig. 1
Well defined heterogenous echoic solid mass (11.3×11.7×10.2 cm) with some tiny cystic changes abuting on atrophic uterus in right adnexa.
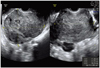
Fig. 2
The specimen received for frozen section consists of a circumscribed ovarian mass (16×16 cm), which is composed of yellow-gray solid firm mass with focal myxoid change.
References
1. Waxman M, Vuletin JC, Urcuyo R, Belling CG. Ovarian lowgrade stromal sarcoma with thecomatous features: a critical reappraisal of the so-called "malignant thecoma". Cancer. 1979. 44:2206–2217.
2. Laufer L, Barki Y, Mordechai Y, Maor E, Mares A. Ovarian fibroma in a prepubertal girl. Pediatr Radiol. 1996. 26:40–42.
3. Mawad NM, Hassanein OM. Ovarian fibro-thecoma in a 19 years old Sudanese girl. Gynaecological case report. Clin Exp Obstet Gynecol. 1994. 21:243–245.
4. Stage AH, Grafton WD. Thecomas and granulosa-theca cell tumors of the ovary: an analysis of 51 tumors. Obstet Gynecol. 1977. 50:21–27.
5. Sivanesaratnam V, Dutta R, Jayalakshmi P. Ovarian fibroma: clinical and histopathological characteristics. Int J Gynaecol Obstet. 1990. 33:243–247.
6. Siekierska-Hellmann M, Sworczak K, Babińska A, Wojtylak S. Ovarian thecoma with androgenic manifestations in a postmenopausal woman. Gynecol Endocrinol. 2006. 22:405–408.
7. Conte M, Guariglia L, Benedetti Panici P, Scambia G, Rabitti C, Capelli A, et al. Ovarian fibrothecoma: sonographic and histologic findings. Gynecol Obstet Invest. 1991. 32:51–54.
8. Mak CW, Tzeng WS, Chen CY. Computed tomography appearance of ovarian fibrothecomas with and without torsion. Acta Radiol. 2009. 50:570–575.
9. Prat J, Scully RE. Cellular fibromas and fibrosarcomas of the ovary: a comparative clinicopathologic analysis of seventeen cases. Cancer. 1981. 47:2663–2670.
10. Le Bouëdec G, Glowaczower E, de Latour M, Fondrinier E, Kauffmann P, Dauplat J. Demons-Meigs' syndrome A case of thecoma and ovarian fibroma. J Gynecol Obstet Biol Reprod (Paris). 1992. 21:651–654.
11. Bradley JA, Hamilton DN, McWhinnie DL, Briggs JD, Junor BJ. Sclerosing peritonitis after CAPD. Lancet. 1983. 2:572–573.
12. Brown P, Baddeley H, Read AE, Davies JD, McGarry J. Sclerosing peritonitis, an unusual reaction to a beta-adrenergic-blocking drug (practolol). Lancet. 1974. 2:1477–1481.
13. Clement PB, Young RH, Hanna W, Scully RE. Sclerosing peritonitis associated with luteinized thecomas of the ovary. A clinicopathological analysis of six cases. Am J Surg Pathol. 1994. 18:1–13.
14. Chiappa A, Zbar AP, Bertani E, Biffi R, Luca F, Crotti C, et al. Primary and recurrent retroperitoneal soft tissue sarcoma: prognostic factors affecting survival. J Surg Oncol. 2006. 93:456–463.
15. Avancès C, Mottet N, Mahatmat A, Chapuis E, Serre I, Culine S. Prognostic factors for first recurrence in patients with retroperitoneal sarcoma. Urol Oncol. 2006. 24:94–96.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download