Abstract
Due to the advancement in laparoscopic surgery in gynecology, laparoscopic surgery of patients with gynecologic malignancy is widely used. A 69-year-old woman who had elevated serum level of CA-125 and malignant ascites was transferred from general surgery after laparoscopic peritoneal biopsy. She complained about 2.5 cm sized abdominal wall mass which developed only 1week after surgery. With the impression of ovarian cancer with port-site metastasis, we performed debulking operation and abdominal wall mass excision. The final pathology confirmed primary peritoneal serous papillary adenocarcinoma. Whenever a female patient has elevated serum level of CA-125 and ascites cytology shows adenocarcinoma, she should be referred to gynecologic oncologists first. And considering the primary peritoneal or ovarian cancer, primary debulking operation by gynecologic oncologist is recommended.
Owing to advancement of laparoscopic surgical technique, laparoscopic approach is now considered as a standard care for many benign gynecologic diseases. But in case of advanced malignant ovarian or peritoneal cancer, we should concern about port-site metastases after laparoscopic procedures. According to some gynecologic oncology literature the reported incidence of port-site metastases varies from less than 1% to up to 19.6% depending on primary malignancy, stage of disease and indication for the laparoscopic procedure [1-5].
We report a patient who developed port-site metastasis of primary peritoneal cancer at left lower quadrant of abdomen only 1 week after laparoscopic biopsy of peritoneal seeding mass.
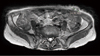
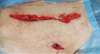
A 69-year-old woman visited our hospital, diagnosed as malignant ascites at local hospital. She visited local clinic due to dyspepsia and abdominal discomfort which developed several weeks ago. Results of esophagogastroduodenoscopy and colonoscopy were normal. Computerized tomography (CT) scanning of the abdomen and pelvis demonstrated moderate amount of ascites and omental infiltration but bilateral normal sized ovaries. 18 F-fluorodeoxyglucose positron emission tomography-CT scan showed extensive peritoneal carcinomatosis. Serum level of the tumor-associated protein CA-125 was elevated over 600 U/mL. Ascites cytology revealed metastatic adenocarcinoma. She visited outpatient department of general surgery of our hospital. Laparoscopic biopsy of peritoneum was performed by general surgeon on Feburary 12, 2011. The 10 mm trocar was applied at umbilicus and 5 mm trocars were applied at left and right lower quadrant of abdomen. Extensive peritoneal carcinomatosis and about 300 mL of ascites were found. The pathologic examination established histologic diagnosis of papillary serous adenocarcinoma. She was referred to gynecologic oncologist and debulking operation was scheduled. When she admitted for operation, she complained about 2.5 cm sized hard palpable mass at her left lower quadrant of abdomen where the laparoscopic trocar was inserted. Follow-up magnetic resonance imaging of the abdomen and pelvis demonstrated extrafascial 2.5 cm sized newly developed metastatic abdominal wall mass after laparoscopic surgery (Fig. 1). Serum level of the tumor-associated protein CA-125 was elevated over 4,009.7 U/mL. On March 8, 2011, she underwent a total abdominal hysterectomy with bilateral salpingo-oophrectomy, bilateral pelvic and para-aortic lymph node dissection, total omentectomy, and abdominal wall mass removal (Fig. 2). The pathologist diagnosed abdominal wall mass as grade 2 primary peritoneal serous papillary adenocarcinoma, and found the same pathologic feature on other specimens. The patient discharged after combination chemotherapy of paclitaxel and carboplatin.
The first detailed descriptions of laparoscopic management and treatment of patients with gynecologic cancers were reported approximately 30 years ago [6]. Since that time, an increasing number of advanced laparoscopic techniques have been used in the management of gynecologic malignancies including second-look laparoscopy, laparoscopic lymphadenectomy, laparoscopically assisted vaginal hysterectomy, and laparoscopically assisted radical vaginal hysterectomy [7].
In case of ovarian malignancies, the advancement in laparoscopic surgery offered some advantages over previous laparotomy including smaller incisions, improved visualization, less blood loss, reduction in the need for analgesics, decreased morbidity and rapid recovery.
But increasing amount of literature describes postoperative tumor growth at the specific puncture sites associated with trocar placement [1,3].
The true incidence of port-site metastases in patients with gynecologic malignancies has not been clearly defined and wide range of incidence was reported by several studies. Childers et al. [3] reported port-site metastases in 1 of 88 patients (1.4% per procedure) undergoing a laparoscopic procedure for ovarian cancer and 1% per procedure for gynecologic cancers in general, while Kruitwagen et al. [1] reported the incidence of port-site metastases to be 16% in patients with ovarian cancer undergoing laparoscopic procedures 9 to 35 days prior to the initial debulking procedure. And van Dam et al. [8] reported port-site metastases in 9% of patients (9/104) undergoing laparoscopic procedures for primary or recurrent ovarian cancer.
Although the concept of tumor cell contamination of the surgical wound was described over 40 years ago [9], the mechanism by which wound metastases occurs is still not completely understood. Hypotheses specific to laparoscopy include exfoliation and spread of tumor cells by laparoscopic instruments, direct implantation at the trocar site by frequent changes of instruments, direct implantation from the passage of the specimen, the presence of the pneumoperitoneum, which can create a "chimney effect" that causes an increase in the passage of tumor cells at port-sites, and preferential growth of malignant cells at areas of laparoscopic peritoneal perforation [10].
The prognostic effect of port-site metastases is not fully understood yet, but Huang et al. [4] reported it is associated with poor outcome. Therefore it is of worthy to prevent port-site metastases, and whenever a female patient has elevated serum level of CA-125 and ascites cytology shows adenocarcinoma, we should consider the possibility of primary peritoneal cancer or normal sized ovarian cancer [11] in case of imaging studies show normal sized ovaries. To prevent the laparoscopic port-site metastases of these patients, all the suspicious patients must be referred to gynecologic oncologists first. If the gynecologic oncologist decided to do the laparoscopic biopsy rather than debulking surgery, the plastic retrieval bag should be used when removing the specimen through 11 mm or 12 mm trocar to prevent cancer cell implantation. But in case of large amount of ascites are present, it would be better to do the primary debulking surgery rather than to do laparoscopic biopsy.
Figures and Tables
References
1. Kruitwagen RF, Swinkels BM, Keyser KG, Doesburg WH, Schijf CP. Incidence and effect on survival of abdominal wall metastases at trocar or puncture sites following laparoscopy or paracentesis in women with ovarian cancer. Gynecol Oncol. 1996. 60:233–237.
2. Abu-Rustum NR, Rhee EH, Chi DS, Sonoda Y, Gemignani M, Barakat RR. Subcutaneous tumor implantation after laparoscopic procedures in women with malignant disease. Obstet Gynecol. 2004. 103:480–487.
3. Childers JM, Aqua KA, Surwit EA, Hallum AV, Hatch KD. Abdominal-wall tumor implantation after laparoscopy for malignant conditions. Obstet Gynecol. 1994. 84:765–769.
4. Huang KG, Wang CJ, Chang TC, Liou JD, Hsueh S, Lai CH, et al. Management of port-site metastasis after laparoscopic surgery for ovarian cancer. Am J Obstet Gynecol. 2003. 189:16–21.
5. Chung MJ, Lee JM, Kim YJ, Park IS, Cho YL, Lee YS. Trocar-site metastasis after laparoscopic gynecologic oncologic surgery. Korean J Obstet Gynecol. 2007. 50:117–125.
6. Rosenoff SH, DeVita T Jr, Hubbard S, Young RC. Peritoneoscopy in the staging and follow-up of ovarian cancer. Semin Oncol. 1975. 2:223–228.
7. Jennings TS, Dottino P, Rahaman J, Cohen CJ. Results of selective use of operative laparoscopy in gynecologic oncology. Gynecol Oncol. 1998. 70:323–328.
8. van Dam PA, DeCloedt J, Tjalma WA, Buytaert P, Becquart D, Vergote IB. Trocar implantation metastasis after laparoscopy in patients with advanced ovarian cancer: can the risk be reduced? Am J Obstet Gynecol. 1999. 181:536–541.
9. Thomas CG. Tumor Cell Contamination of the Surgical Wound: Experimental and Clinical Observations. Ann Surg. 1961. 153:697–704.
10. Wang PH, Yuan CC, Lin G, Ng HT, Chao HT. Risk factors contributing to early occurrence of port site metastases of laparoscopic surgery for malignancy. Gynecol Oncol. 1999. 72:38–44.
11. Choi CH, Kim TJ, Kim WY, Ahn GH, Lee JW, Kim BG, et al. Papillary serous carcinoma in ovaries of normal size: a clinicopathologic study of 20 cases and comparison with extraovarian peritoneal papillary serous carcinoma. Gynecol Oncol. 2007. 105:762–768.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download