Abstract
Purpose
The aim of this study was to investigate and compare the effects of udenafil and mannitol in an experimental renal ischemia-reperfusion (I/R) injury model.
Materials and Methods
A total of 64 female Wister Albino rats were used. Right nephrectomy was performed in all groups. In the control group; I/R injury was not performed. In the I/R group; left renal pedicle was clamped for 45 minutes and then underwent 60 minutes and 24 hours of reperfusion. In the mannitol group; 1 mL 20% mannitol was given intravenously 15 minutes before clamping. In the udenafil group; 10-mg/kg udenafil was given orally 1 hour before clamping. Creatinine (Cr), blood urea nitrogen (BUN), Cr clearance, malondialdehyde, neutrophil gelatinase associated lipocalin (NGAL), histological examination and DNA damage (Comet Assay method) levels were compared in tissue, serum and urine samples.
Renal ischemia-reperfusion (I/R) injury leads to acute renal damage, acute renal failure (ARF) and chronic renal failure. I/R injury is often seen in intensive care units, secondary to trauma, shock, sepsis, renal transplantation, cardiovascular and urologic surgery [12]. ARF can be seen in 5% of hospitalized patients and approximately 10% of these patients will need renal transplantation [3]. Also ARF patients had more than 2 days of hospitalization when compared to patients without ARF and mortality rates are 4.1 times higher [4]. In renal transplantation I/R injury usually leads to primary renal dysfunction, delayed graft failure, increased late acute rejection and allograft dysfunction [56]. In partial nephrectomy, renal arteries are clamped before tumor removal and during this time I/R injury leads to functional renal tissue damage [7].
The pathogenesis of I/R injury is not defined clearly and several independent multifactorial mechanisms play a role in this process. During ischemia tissue blood flow and oxygen levels decrease and cellular damage occurs depending on the duration and the severity of ischemia.
During reperfusion ischemic tissue is supported by blood flow but paradoxically leads to additional damage [8910]. In I/R injury reactive oxygen radicals and the acute inflammatory process are the main factors causing the damage. The most sensitive cellular structures are membrane lipids, proteins, nucleic acids and DNA molecules [11].
Mannitol is an osmotic diuretic and used to prevent acute renal injury because of its potential renal protective effect. This renoprotective effect is provided by the removal of obstructing tubular casts, dilution of nephrotoxic substances in the tubular fluid and reduction in the swelling of tubular elements via osmotic extraction of water [12]. Also mannitol increases renal blood flow and consequently clears free radicals during the reperfusion period [12]. Some studies have shown that the prophylactic administration of mannitol reduces renal damage however some other studies have reported that it has no benefit at all and there are also other studies showing that it increases renal damage [1213].
Nitric oxide (NO) has a pivotal role in I/R injury which is relatively complicated. Indictable nitric oxide synthases are related to the damage and endothelial NOS (eNOS) has protective effects. Udenafil is a phosphodiesterase type 5 (PDE-5) inhibitor and increases the level of cyclic guanosine monophosphate which is the second messenger for NO and eNOS. Also udenafil was known to have fixed and consistent pharmacokinetics in rats and have been used orally in the previous experimental studies for similar purposes [1415].
The aim of this study was to compare the renoprotective effects of mannitol and udenafil on renal I/R injury in an experimental model which may potentially help the field of deceased donor transplantation and partial nephrectomy where clamping is required.
This study was approved by the Pamukkale University Animal Ethics Committee. Sixty-four female Wistar Albino rats weighing 281–330 g were randomly divided into 4 groups. Right nephrectomy was performed in all groups. Left kidney remained intact and no additional procedures were performed in the control group. In I/R groups left renal artery was occluded by a vascular clamp for 45 minutes and the clamp was removed for reperfusion for 60 minutes (n=8) and 24 hours (n=8) for short-term effects of I/R injury and functional studies, respectively. In the mannitol group; 1 mL 20% mannitol was given intravenously 15 minutes before clamping and same procedures were performed, as mentioned above. In the udenafil group; 10-mg/kg udenafil was given orally 1 hour before clamping with an orogastric tube and the procedure was repeated. At the end of the 60 minutes reperfusion period left nephrectomy was performed and tissue samples were divided into 3 pieces for biochemical, histopathological and comet assay examinations. Urine samples were collected by intravesical aspiration and blood samples were collected by intracardiac aspiration. In 24-hour reperfusion groups, rats were placed into metabolic cages after anesthesia recovery. At the end of the 24-hour reperfusion period rats were reanesthetized and blood and urine samples were collected. Tissue samples were kept at −80℃, blood and urine samples were centrifuged and kept at −20℃.
Serum creatinine (Cr), blood urea nitrogen (BUN), and
urine Cr levels were calculated with the automatic system at Pamukkale University Medical Faculty Hospital Biochemistry Laboratory. Cr clearance was calculated by using the formula: urine Cr×24-hour urine volume/serum Cr×1440. Serum Cr levels were indicated as “Day 0” 45 minutes ischemia and 60 minutes reperfusion period and “Day 1” 45 minutes ischemia and 24 hours reperfusion period.
Samples of malondialdehyde (MDA) were prepared by standards in the prospectus of lipid peroxidation (MDA) Colorimetric/FluoROMetric Assay Kit product by BioVision. Results were collected by spectrophotometric measurement of the color generated by the reaction of thiobarbituric acid (TBARS) with MDA.
Samples were diluted 1:10 as calculation of medium target protein concentration was aimed. Standard and controls were prepared according to the instructions of Boster Immunoleader Rat Lipocalin-2/neutrophil gelatinase associated lipocalin (NGAL) ELISA Kit. NGAL antibody, Avidin-Biotin-Peroxidase Complex (ABC), phosphate buffer saline (PBS) were also prepared. TMB (3,3′,5,5′-Tetramethyl benzidine) color developing agent and TMB stop solutions were used as ready-to-use products. Then 0.1 mL of standard, control, blind and samples were put into the plates and incubated at 37℃ for 90 minutes. Biotinized antibodies were added and incubated at 37℃ for 60 minutes. Plates were washed 3 times with 0.01M PBS. Then ABC working solution was added and incubated at 37℃ for 30 minutes. Plates were washed 5 times with 0.01M PBS. TMB color developing agent was added and incubated at 37℃ for 30 minutes in dark room. TMB stop solution was added. Absorbance values was read at 450 nm wavelength with Kayto RT-2100c Microplate Reader device.
Comet sampling was performed by using Cell Biolabs OxiSelect Comet Assay Kit and Santacruz cOmPlete EDTA-Free protease inhibitor cocktail tablets. DNA damage was determined by using the program Comet Assay IV system (AutoComet) by one physiologist blindly.
All data were expressed as the mean±standard deviation. Differences between groups were evaluated by Kruskal-Wallis variance analysis followed by a Mann-Whitney U-test with Bonferroni correction for binary comparisons. The Pearson chi-square test was used to compare renal pathology grades. Values of p<0.05 were considered statistically significant. All data were processed using the SPSS ver. 17.0 (SPSS Inc., Chicago, IL, USA) statistical package.
Serum Cr levels of the udenafil and mannitol groups on day 0 and day 1 were found to be lower than the I/R group. This decrease was significant for the udenafil group (Fig. 1, Table 2). Also, an increase in serum Cr was observed for all groups on the 1st day and for the udenafil group the rate of increase was minimum. (control=40%, I/R=64%, mannitol=57%, udenafil=35%). When the serum BUN levels of the groups were compared to the I/R group, results were detected higher for the mannitol group and in contrast significantly lower for the udenafil group (Table 2). When Cr clearance levels of the groups were compared, I/R group had the lowest Cr clearance. Cr clearance was detected higher for the mannitol and udenafil groups. Cr clearance for udenafil group was detected higher than both the I/R and mannitol groups, and these findings were also statistically significant (Fig. 1, Table 2).
The serum and tissue MDA levels were the highest in the I/R group and MDA levels were found significantly lower for both the mannitol and udenafil groups (Table 2). The serum, urine and tissue NGAL levels were detected higher in the I/R group. NGAL levels were detected lower for mannitol and udenafil groups and this decrease was significant for the udenafil group (Table 2).
When pathological damage rates of the groups examined, acute damage rate was detected the highest for the mannitol group. Damage rate was detected lower for the udenafil group (Table 2).
In the comet assay examination of the groups no significant difference was detected after the evaluation of head intensity, tail intensity, and tail length (Table 2).
For many years in practice mannitol has been widely used for decreasing ARF in intensive care units and surgical operations. However, its effectiveness has become highly questionable after recent studies [13]. Tissue protective effects of PDE-5 inhibitors have been shown on kidney and other organs against I/R injury. Udenafil which is a new member of the PDE-5 family has also proved its effectiveness in preventing the renal I/R injury [14]. Although both of mannitol and udenafil have been shown to be effective separetely and mannitol is widely used in daily practice, there have been no studies comparing the effects of these agents. This is the first study comparing the efficacy of both the mannitol and udenafil in an I/R experimental model. Additionally, comet-assay method which is an indicator of DNA damage has been used for evaluation of tissue damage in such a model for the first time.
The effectiveness of several PDE-5 inhibitors including sildenafil, tadalafil, udenafil and zaprinast for preventing the renal damage caused by I/R were investigated before. In several studies recovery of renal function was shown with the decrease in Cr and BUN levels, increase in glomerular filtration rate (GFR) or by using scintigraphic studies [15161718]. The positive and renoprotective effects of mannitol were also shown by Burke et al. [19] and Dawson [20] in the acute renal damage models. We detected that there was an improvement in renal function parameters in mannitol and udenafil groups which was consistent with those studies and this improvement was significantly higher in the udenafil group compared to the mannitol group which was not demonstrated previously.
NGAL can be detected in the urine in the second hour after ischemic renal damage and its level continue to rise proportionally with the intensity of damage. Also it is an indicator which can be detected in the serum before the Cr level increases. In an experimental renal I/R injury model, NGAL and kidney injury molecule levels decreased with a concordant increase in GFR levels in the group treated with tadalafil [18]. A significant difference in the NGAL levels was also demonstrated in studies investigating the effects of renal transplantation. NGAL levels of the patients who had transplantation and the patients who are waiting for transplantation have been found to be significantly higher than the healthy population (7,6 fold and 9 fold, respectively). Twelve months later, a considerable decrease in the NGAL levels (1.5 times the healthy population) has been detected in transplantation group when compared to the patients who were on the waiting list (10 times the healthy population) [21]. In our study, tissue NGAL level was found to be highest in the I/R group, and this difference was statistically significant when compared to the control group. Tissue NGAL levels of the udenafil and mannitol groups were determined to be lower the I/R group. The decrease in the udenafil group was more explicit and found to be statistically significant. We also showed that the increase in the tissue NGAL level starts within the first 60 minutes with the damage occurring after the reperfusion. In our study, the serum and urine NGAL level after reperfusion was detected highest in the I/R group. The serum and urine NGAL levels in the udenafil and mannitol groups were considerably lower when compared to the I/R group. Although not statistically significant the serum NGAL level of the udenafil group was detected to be lower than the mannitol group.
Free oxygen radicals arise with the I/R injury and cause damage to membrane lipids. Lipid peroxidation is linked to tissue damage, and MDA is an indicator that brings out lipid peroxydation. Kucuk et al. [22] have compared the effectiveness of sildenafil and tadalafil which are inhibitors of PDE5 in the renal I/R injury model. In their study, they have found that both of the PDE5 inhibitors have decreased the tissue MDA level more than the I/R group and that this effect is better in the sildenafil group. Fu et al. [23] made by using L-citrulline in the study, have shown a considerable increase in tissue MDA levels. They have found that MDA levels have decreased significantly in the L-citrulline group. In accordance with the previous studies, our study showed that both mannitol and udenafil decreases the MDA level and inhibits lipid peroxidation.
The alterations in renal histopathology in renal I/R models have also been investigated. In previous studies investigating the effects of different agents including PDE-5 inhibitors, several authors showed that there was a considerable decrease of damage in treatment groups compared to the I/R groups [1617242526]. In our study, the histopathological renal damage in the early period was found to be higher in the I/R, mannitol and udenafil groups when compared to the control group. Although the level of damage in the I/R and mannitol groups more obvious and the level of damage in the udenafil group is less when compared to the I/R and mannitol groups, the difference was not statistically significant.
One of the important effects of I/R injury occurs on DNA which was shown in previous studies. Dias et al. [27] have studied the effects of sildenafil on oxidative stress, DNA damage and renal functions in an experimental renovascular hypertension model. When comparing measurements of tail density and tail momentum in Comet sampling analysis, a significant increase (2 fold) in DNA damage in the placebo group was demonstrated when compared to the group treated with sildenafil. In the current study we have found that in the mannitol group and the udenafil group, DNA damage was lower. The difference in the udenafil group was significant. Karahalil et al. [28] have examined the I/R injury related DNA damage that occurs in preischemia, postischemia, and postreperfusion of patients who have had lower extremity surgery. In the Comet sampling analysis, tail density between the groups has been examined, and it has been found that that there was no significant difference among the 3 groups. In our study, looking at the most important parameter, the tail density, the differences were not found to be significant although there was an increase in density in the I/R group and a decrease in the mannitol and udenafil groups. According to the results obtained from the post I/R Comet sampling analysis, no significant DNA damage has occurred in tissue cells. The reason of that may be cytotoxic damage did not reach to genotoxic levels or the lack of Comet Assay method to detect renal tissue genotoxic damage.
Although routinely used in daily practice to prevent ischemia-related injury during partial nephrectomy in urology clinics, there are controversies regarding mannitol. In their review, Hersey and Poullis [29] have investigated the effects of mannitol use in open abdominal aortic aneurysm surgeries on the protection of kidneys. In some of the studies included in that review, it is revealed that mannitol causes a remission in the renal functions during the acute stage, but it does not affect the overall renal failure incidence. Power et al. [30] have investigated the effects of mannitol use before partial nephrectomy ischemia on renal functions. Considering the long-term effects (after at least six months), they have concluded that the patients using mannitol do not exhibit a significant difference regarding renal functions as compared to the patients who were not administered mannitol. Karajala et al. [12] have revealed in their study that diuretic agents including mannitol, are not to be used for protective purposes; they are to be used merely to set the liquid balance in treatment. In another meta-analysis, the use of mannitol did not have additional benefits in decreasing acute renal damage of the patients who received adequate fluid treatment. Also, mannitol was proved to increase additional renal damage in contrast-induced nephropathy [13].
This study showed that udenafil improves renal functions, decreases lipid peroxidation and reduces histological and DNA damage in the renal I/R injury models. It has significantly better effects on decreasing renal damage than mannitol which is currently most widely used agent for the same purposes. The use of other phosphodiesterase type-5 inhibitors in other studies revealed similar results. Although previous studies have shown these effects separately with various agents including different PDE-5 inhibitors, to our knowledge our study is the first demonstrating a wide range of biochemical, histological and ultrastructural changes in an experimental I/R injury model. Also, as an experimental study, our results show the short term effects of both agents. To support the short term effects and to investigate the long term effects, further experimental and clinical trials are needed.
ACKNOWLEDGMENTS
This study was supported by Pamukkale University Scientific Research Project Coordination Unit (2014TPF009).
References
1. Ahmadiasl N, Banaei S, Alihemmati A, Baradaran B, Azimian E. The anti-inflammatory effect of erythropoietin and melatonin on renal ischemia reperfusion injury in male rats. Adv Pharm Bull. 2014; 4:49–54. PMID: 24409409.
2. Sancaktutar AA, Bodakci MN, Hatipoglu NK, Soylemez H, Basarılı K, Turkcu G. The protective effects of pomegranate extracts against renal ischemia-reperfusion injury in male rats. Urol Ann. 2014; 6:46–50. PMID: 24669122.

3. Legrand M, Mik EG, Johannes T, Payen D, Ince C. Renal hypoxia and dysoxia after reperfusion of the ischemic kidney. Mol Med. 2008; 14:502–516. PMID: 18488066.

4. Liangos O, Wald R, O'Bell JW, Price L, Pereira BJ, Jaber BL. Epidemiology and outcomes of acute renal failure in hospitalized patients: a national survey. Clin J Am Soc Nephrol. 2006; 1:43–51. PMID: 17699189.

5. Fadili W, Habib Allah M, Laouad I. Chronic renal allograft dysfunction: risk factors, immunology and prevention. Arab J Nephrol Transplant. 2013; 6:45–50. PMID: 23282234.
6. Geddes CC, Woo YM, Jardine AG. The impact of delayed graft function on the long-term outcome of renal transplantation. J Nephrol. 2002; 15:17–21. PMID: 11936421.
7. Martin GL, Warner JN, Nateras RN, Andrews PE, Humphreys MR, Castle EP. Comparison of total, selective, and nonarterial clamping techniques during laparoscopic and robot-assisted partial nephrectomy. J Endourol. 2012; 26:152–156. PMID: 22192110.

9. Orvieto MA, Zorn KC, Mendiola FP, Gong EM, Lucioni A, Mikhail AA, et al. Ischemia preconditioning does not confer resilience to warm ischemia in a solitary porcine kidney model. Urology. 2007; 69:984–987. PMID: 17482956.

10. Pasupathy S, Homer-Vanniasinkam S. Ischaemic preconditioning protects against ischaemia/reperfusion injury: emerging concepts. Eur J Vasc Endovasc Surg. 2005; 29:106–115. PMID: 15649715.

11. Wilhelm J. Metabolic aspects of membrane lipid peroxidation. Acta Univ Carol Med Monogr. 1990; 137:1–53. PMID: 1966309.
12. Karajala V, Mansour W, Kellum JA. Diuretics in acute kidney injury. Minerva Anestesiol. 2009; 75:251–257. PMID: 18636060.
13. Yang B, Xu J, Xu F, Zou Z, Ye C, Mei C, et al. Intravascular administration of mannitol for acute kidney injury prevention: a systematic review and meta-analysis. PLoS One. 2014; 9:e85029. PMID: 24454783.

14. Shim HJ, Kim YC, Kim EJ, Kim DG, Kwon JW, Kim WB, et al. Pharmacokinetic changes of DA-8159, a new erectogenic, after intravenous and oral administration to rats with acute renal failure induced by uranyl nitrate. Biopharm Drug Dispos. 2004; 25:137–142. PMID: 15083502.

15. Yang JW, Han ST, Kim YS, Song SH, Kim BR, Eom MS, et al. Effects of a cGMP-specific phosphodiesterase inhibitor on expression of endothelial nitric oxide synthase and vascular endothelial growth factor in rats with cyclosporine-induced nephrotoxicity. Transplant Proc. 2010; 42:4625–4632. PMID: 21168749.

16. Medeiros PJ, Villarim Neto A, Lima FP, Azevedo IM, Leão LR, Medeiros AC. Effect of sildenafil in renal ischemia/reperfusion injury in rats. Acta Cir Bras. 2010; 25:490–495. PMID: 21120279.

17. Polcari AJ, Farooq AV, Woods ME, Ripsch MS, Picken M, Turk TM, et al. Effect of the phosphodiesterase-5 inhibitor zaprinast on ischemia-reperfusion injury in rats. J Endourol. 2013; 27:338–342. PMID: 22974473.

18. Sohotnik R, Nativ O, Abbasi A, Awad H, Frajewicki V, Bishara B, et al. Phosphodiesterase-5 inhibition attenuates early renal ischemia-reperfusion-induced acute kidney injury: assessment by quantitative measurement of urinary NGAL and KIM-1. Am J Physiol Renal Physiol. 2013; 304:F1099–F1104. PMID: 23364806.

19. Burke TJ, Cronin RE, Duchin KL, Peterson LN, Schrier RW. Ischemia and tubule obstruction during acute renal failure in dogs: mannitol in protection. Am J Physiol. 1980; 238:F305–F314. PMID: 6769339.

20. Dawson JL. Jaundice and anoxic renal damage: protective effect of mannitol. Br Med J. 1964; 1:810–811. PMID: 14100797.

21. Magnusson NE, Hornum M, Jørgensen KA, Hansen JM, Bistrup C, Feldt-Rasmussen B, et al. Plasma neutrophil gelatinase associated lipocalin (NGAL) is associated with kidney function in uraemic patients before and after kidney transplantation. BMC Nephrol. 2012; 13:8. PMID: 22325322.

22. Küçük A, Yucel M, Erkasap N, Tosun M, Koken T, Ozkurt M, et al. The effects of PDE5 inhibitory drugs on renal ischemia/reperfusion injury in rats. Mol Biol Rep. 2012; 39:9775–9782. PMID: 22736111.

23. Fu X, Li S, Jia G, Gou L, Tian X, Sun L, et al. Protective effect of the nitric oxide pathway in L-citrulline renal ischaemia-reperfusion injury in rats. Folia Biol (Praha). 2013; 59:225–232. PMID: 24485304.
24. Gasanov F, Aytac B, Vuruskan H. The effects of tadalafil on renal ischemia reperfusion injury: an experimental study. Bosn J Basic Med Sci. 2011; 11:158–162. PMID: 21875417.

25. Goksin I, Adali F, Enli Y, Akbulut M, Teke Z, Sackan G, et al. The effect of phlebotomy and mannitol on acute renal injury induced by ischemia/reperfusion of lower limbs in rats. Ann Vasc Surg. 2011; 25:1118–1128. PMID: 22023943.

26. Guzeloglu M, Yalcinkaya F, Atmaca S, Bagriyanik A, Oktar S, Yuksel O, et al. The beneficial effects of tadalafil on renal ischemia-reperfusion injury in rats. Urol Int. 2011; 86:197–203. PMID: 21160160.

27. Dias AT, Rodrigues BP, Porto ML, Gava AL, Balarini CM, Freitas FP, et al. Sildenafil ameliorates oxidative stress and DNA damage in the stenotic kidneys in mice with renovascular hypertension. J Transl Med. 2014; 12:35. PMID: 24502628.

28. Karahalil B, Polat S, Senkoylu A, Bölükbaşi S. Evaluation of DNA damage after tourniquet-induced ischaemia/reperfusion injury during lower extremity surgery. Injury. 2010; 41:758–762. PMID: 20398900.

29. Hersey P, Poullis M. Does the administration of mannitol prevent renal failure in open abdominal aortic aneurysm surgery? Interact Cardiovasc Thorac Surg. 2008; 7:906–909. PMID: 18653498.

30. Power NE, Maschino AC, Savage C, Silberstein JL, Thorner D, Tarin T, et al. Intraoperative mannitol use does not improve long-term renal function outcomes after minimally invasive partial nephrectomy. Urology. 2012; 79:821–825. PMID: 22469577.

Fig. 1
Creatinine and creatinine clearance levels of the control and the study groups. I/R, ischemia-reperfusion; M, mannitol; U, udenafil. *Statiscally significant (p<0.01) when U and M groups compared to I/R group.
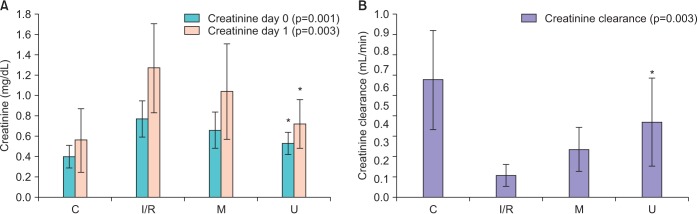
Table 1
Scores used for histopathological evaluation
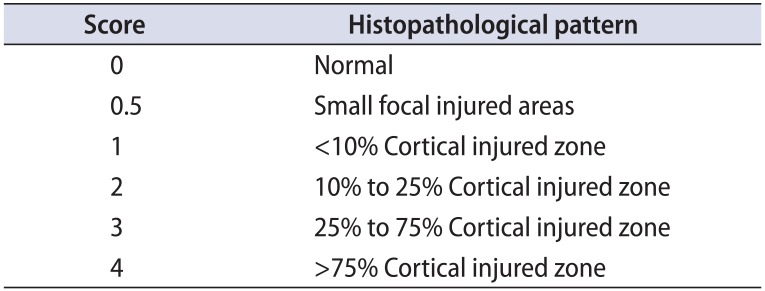
| Score | Histopathological pattern |
|---|---|
| 0 | Normal |
| 0.5 | Small focal injured areas |
| 1 | <10% Cortical injured zone |
| 2 | 10% to 25% Cortical injured zone |
| 3 | 25% to 75% Cortical injured zone |
| 4 | >75% Cortical injured zone |
Table 2
The results of functional, biochemical and pathological examinations in control and study groups
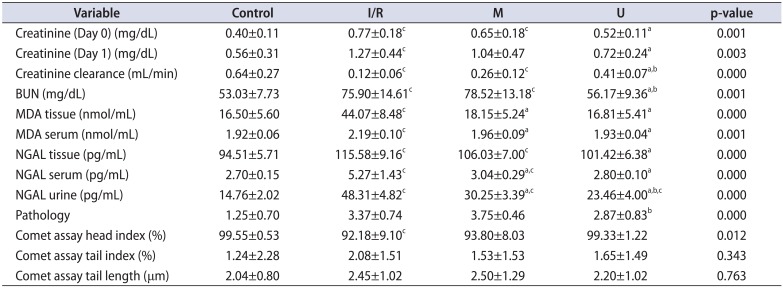
I/R, ischemia-reperfusion; M, mannitol; U, udenafil; BUN, blood urea nitrogen; MDA, malondialdehyde; NGAL, neutrophil gelatinase associated lipocalin.
a:Statistically significant (p<0.01) when U and M groups compared to I/R group. b:Statistically significant (p<0.01) when U group compared to M group. c:Statistically significant (p<0.01) when U, M and I/R groups compared to control group.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download