Abstract
Purpose
Thulium vaporesection of the prostate (ThuVARP) is a new and safe approach for patients receiving anticoagulant therapy in whom transurethral resection of the prostate (TURP) may possess a high bleeding risk. We aimed to demonstrate the efficacy and safety of ThuVARP in patients receiving oral antiplatelet/anticoagulant (OAP/OAC) therapy.
Materials and Methods
A total of 103 patients who underwent ThuVARP between 2011 and 2013 were enrolled in the study. Patients were divided into 2 groups. Group A consisted of 47 patients who underwent low molecular weight heparin (LMWH) bridging and group B consisted of 56 patients who were operated on while receiving OAP/OAC therapy.
Results
The drop in hemoglobin levels in the pre- and postoperative periods was significantly higher in group A than in group B. When subgroups were analyzed, the mean drop in hemoglobin was significantly lower in the warfarin and ticlopidine subgroups of group B than in group A. International Prostate Symptom Scores were significantly lower 3, 12, 18, and 24 months after surgery in group A than in group B. Quality of life scores, maximal flow rate values, and postmicturition residual urine volumes (mL) were similar between the 2 groups. A total of 38 and 41 patients in groups A and B, respectively, had no complications.
Transurethral resection of the prostate (TURP) is considered the best option for treating benign prostatic enlargement (BPE); however, it is associated with significant bleeding [1]. In recent years, we have seen a gradual increase in life expectancy and, particularly in more developed countries, an increased incidence of cardiovascular diseases, metabolic syndrome, and cerebrovascular stroke, all of which are conditions that require oral antiplatelet or anticoagulation (OAP/OAC) therapy [2]. Owing to the severe bleeding risk in these patients, TURP is contraindicated. Furthermore, suspending OAP/OAC therapy is also not recommended. Recently, an increasing number of procedures have been introduced as alternatives to TURP [3]. For example, the use of laser technology has advantages in patients receiving OAP/OAC therapy who have severe comorbidities and bleeding disorders [4].
Thulium vaporesection of the prostate (ThuVARP) with a 2-µm continuous-wave (CW) thulium-doped yttrium-aluminum-garnet (Tm:YAG) laser has been established as a new approach for BPE treatment [5]. This laser has a peak absorption spectrum closer to that of water than the holmium laser, which causes increased tissue vaporization and results in reduced penetration depth. However, because the thulium laser operates in CW mode, it permits smaller and more precise cutting and consequently decreased risk of bleeding [6]. The aim of this study was to demonstrate the efficacy and safety of vaporesection with the 120-W Tm:YAG laser (Revolix Duo) in patients with BPE receiving systemic OAP/OAC therapy and to evaluate medium-term functional outcomes.
Between October 2011 and December 2013, a total of 103 patients with lower urinary tract symptoms due to BPE were enrolled in the study, and data were retrospectively analyzed. We divided the patients into 2 groups. In group A, OAP/OAC therapy was interrupted for 10 days before the operation and patients were switched to low molecular weight heparin (LMWH) until 2 weeks after the procedure. In group B, the OAP/OAC therapy was continued throughout the whole perioperative period. Group B patients were also divided into subgroups according to the OAP/OAC therapy that they were using: warfarin, acetyl salicylic acid (ASA), clopidogrel, ticlopidine, or ASA+clopidogrel.
According to the standard protocol of the university hospital in which the study was carried out, each patient was informed of the procedure upon admission and signed an informed consent allowing data collection for research purposes. The study design was in accordance with the Helsinki Declaration, conformed to the Committee on Publication Ethics guidelines, and was approved by the Institutional Review Board of the Department of Human Pathology, Section of Urology, University of Messina, Messina, Italy in which it was performed. All the design, analysis, data interpretation, drafting, and revisions followed the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement as well as the guidelines for reporting observational studies, available through the EQUATOR (Enhancing the Quality and Transparency of Health Research) network [7]. All operations were performed by a single surgeon (CM) with extensive experience in laser prostatectomy and prostate resection. The ThuVARP technique used in this study was the same described in our previous publication [2].
The procedure was performed by using the 120-W 2-µm CW Tm:YAG laser (Revolix Duo). The Revolix Duo has 2 laser generators: (1) a 120-W CW mode for soft tissue use and (2) a 20-W pulsed head to holmium for lithotripsy. A 550-µm core nude-ended fiber (RigiFib; OHG, LISA Laser, Katlenburg-Lindau, Germany) was used in combination with a 26-F continuous-flow laser resectoscope (Karl Storz GmbH & Co. KG, Tuttlingen, Germany).
We first marked the distal resection border near the verumontanum. Incisions were made on the bladder neck at the 5 and 7 o'clock positions and vaporesection was performed on the median lobe. Finally, vaporesection of the lateral lobe was performed until the prostatic capsule was reached. By performing this technique, small tissue chips are obtained, which were evacuated through the resectoscope sheath. At the end of every procedure, a 20-F Foley catheter was placed and bladder irrigation was performed in the case of hematuria. The catheter was routinely removed 48 to 72 hours after the procedure regardless of preoperative prostate size when the urine was clear. Cefazolin (2 g) was administered as a short protocol 1 hour before the procedure. Data collected included operative time, hospital stay, catheterization time, and hemoglobin values pre- and postoperatively. The postoperative hemoglobin measurement was done at 6 hours postoperatively. In addition, maximal flow rate (Qmax), postmicturition residual urine (PMR), International Prostate Symptom Score (IPSS), and quality of life (QoL) scores were assessed immediately before surgery (baseline) and at 3, 6, 12, 18, and 24 months after treatment. Prostate-specific antigen (PSA) values were assessed preoperatively and at 12 and 24 months. All perioperative and postoperative complications were recorded.
Statistical analyses were performed by using SPSS ver. 20 (IBM Corp, Armonk, NY, USA). The statistical significance was set at p<0.05. Baseline variables were described by using mean and standard deviation or percentages, as appropriate. Mann-Whitney U-tests were used to evaluate the difference between quantitative measurements that had nonparametric distributions. Chi-square tests and Fisher exact tests were used for categorical data.
A total of 103 patients who underwent laser prostatectomy were included in the study. In group A, anticoagulant therapy was interrupted and switched for LMWH in 47 patients, whereas in group B, 56 patients underwent surgery while still on their anticoagulant therapy regimen. The mean ages of groups A and B were 68.5±6 and 69.6±6.8 years, respectively. The mean operation, hospitalization, and catheterization durations are given in Table 1 for each group. The mean preoperative prostate volumes were 66.8±14.1 and 65.3±9.5 mL for groups A and B, respectively. The mean amounts of resected prostate tissue were 43.8±10.9 and 43.1±6.7 mL for groups A and B, respectively. These values, along with subgroup values are also given in Table 1. In group B, 7 patients were on warfarin, 23 patients were on ASA, 14 were on ticlopidine, 8 were on clopidogrel, and 4 patients were on ASA + clopidogrel combination treatment.
The mean preoperative hemoglobin level of patients in group A was 13.7±1 and that of patients in group B was 13.5±1.5. The mean postoperative hemoglobin level of patients in group A was 13±1 and that of patients in group B was 13.2±1.5. Neither pre- nor postoperative hemoglobin values were statistically significantly different between groups A and B. However, the drop in hemoglobin values in the pre- and postoperative periods was significantly higher in group A: 0.7±0.5 and 0.3±0.5 in groups A and B, respectively (p<0.005). When subgroup analyses were performed within group B, the mean drop in hemoglobin was significantly lower in patients who were on warfarin and ticlopidine compared with patients in group A (p<0.005, p<0.005, respectively). The mean pre- and postoperative hemoglobin values along with the mean drop in hemoglobin are given for each group in Fig. 1.
When the IPSS values were compared between groups A and B, group A had significantly lower IPSS values at 3, 12, 18, and 24 months after surgery (p<0.005, p<0.05, p<0.005, p<0.001, respectively). However, in the preoperative period, the IPSS was 15.9 in group A and 18.6 in group B and the difference was not statistically significant. No statistically significant differences were found between groups A and B or subgroups of group B in QoL scores, Qmax values, or PMR volumes (mL). During the whole follow-up period, none of the patients had a urinary catheter. The mean IPSS, Qmax, and PMR values along with QoL scores for groups A and B and the subgroups of group B are given in Table 2 and Fig. 2.
PSA values were evaluated preoperatively and at 12 and 24 months. The mean PSA values in the preoperative period were 3.1±1.5 and 3.1±1.9 in groups A and B, respectively. At 12 months after the operation, the values were 2.3±0.9 and 2.4±1.2, and at 24 months, the values were 2.2±0.8 and 2.3±0.9 for groups A and B, respectively. The differences between the 2 groups were not statistically significant.
In group A, 38 patients had no complications, whereas 4 had transient urge incontinence, 4 had urinary tract infection (UTI), and 1 patient needed recatheterization for slight hematuria for 5 days which resolved without further intervention. No complications occurred in 41 patients in group B; however, in 6 patients, transient urge incontinence occurred; 7 patients had a UTI; 1 patient needed recatheterization for 4 days due to hematuria; and 1 patient needed transfusion due to intraoperative bleeding.
According to the latest guidelines of the European Association of Urology, TURP is the best treatment option for patients with nonneurogenic lower urinary tract symptoms affected by BPE [8]. However, morbidity with TURP remains fairly high, with associated bleeding risk and electrolytic disorders. In particular, patients receiving OAP/OAC treatment carry a major risk for hemorrhage, and discontinuation of these treatments carries the risk of thromboembolic events. This may be considered a double-edged sword: the management of BPE is problematic for the urologist owing to the increased use of OAP/OAC for primary or secondary prevention of cardiovascular complications [9].
In their study, Capodanno et al. [10] expressed that this commonly used bridging practice is potentially harmful in patients undergoing noncardiac surgeries and can result in worse ischemic outcomes, especially in the first 30 days, along with significant risk of bleeding. Rose et al. [11] pointed out the importance of these risks and proposed an immediate change in daily practice to abandon bridging therapy in most of these patients. To clarify this issue for urological procedures, several studies were conducted on TURP in patients receiving OAP/OAC therapy regimens. Taylor et al. [12] analyzed 163 cases and found a higher rate of perioperative bleeding in patients who continued OAP/OAC therapy than in those who were not on an OAP/OAC therapy. Also, cerebrovascular and cardiovascular events occurred in patients who had discontinued their OAP/OAC treatments. This study demonstrates the importance of studies in which patients are evaluated while they are still receiving OAP/OAC treatments.
To question the safety of bridging therapy with LMWH and the discontinuation of prophylactic anticoagulants, Dotan et al. [13] compared the bleeding rates of patients who were on chronic OAP/OAC treatment and switched to LMWH perioperatively with those of non–warfarin-treated patients. They found a bleeding rate of 20% in patients who switched to perioperative LMWH, and this situation is presumed to result in longer hospitalization duration. However, they concluded that bridging is a safe option for these patients. High rates of bleeding were also reported by Parr et al. [14] in a cohort of 12 patients with more than 30% of them having transfusions. However, they reported that several such patients who switched from warfarin to heparin before surgery presented to the Emergency Department with major thromboembolic events and some even required open-heart surgery. Thus, they concluded that there is no need for bridging therapy and that bleeding problems can be easier to manage than thromboembolic events [14]. Ong et al. [15] reported that in a population of 293 patients on chronic OAP/OAC therapies who underwent TURP, bleeding complications occurred in 10% of patients, of whom 8% required prolonged bladder irrigation, 2% required re-catheterization, 2% required reoperation, and 2% needed blood transfusion.
Although TURP has been the gold standard for the surgical management of BPE for the past several decades, in recent years, a new series of lasers and techniques have become well established as an alternative treatment for BPE. In particular, holmium and thulium lasers have shown safety and efficacy in numerous studies throughout the past decade [21617]. Studies reporting the safety of laser prostatectomy in patients on OAP/OAC therapies are also becoming more common [18192021]. In their study in 2007, Ruszat et al. [21] reported that in 116 patients on OAP/OAC therapy who were compared with a group of 92 control cases, none of the patients required transfusions. Owing to slight hematuria, 17% of these patients required prolonged bladder irrigation; however, this frequency was 5% among the control cases. In our patient groups, one patient in each group was catheterized as a result of bleeding after the retrieval of the urinary catheter and required prolonged irrigation. Ruszat et al. [21] reported no statistically significant differences among the 2 groups in terms of IPSS, QoL scores, or Qmax. These findings agree with our study. In our patient cohort, although these variables all improved after surgery, there were no statistically significant differences between the groups showing that bridging OAP/OAC with LMWH or continuing OAP/OAC affected functional outcomes.
In various studies, authors have compared different techniques and different surgical modalities for safety and efficacy. For example, Fu et al. [5] compared ThuVARP and monopolar TURP and stated that the efficacy of the two modalities was similar, although ThuVARP resulted in less blood loss and a shorter hospital stay. Bach et al. [22] compared the 120-W and 70-W Tm:YAG lasers in terms of hemostatic properties. The device that we used, the 120-W Tm:YAG laser, provided better ablation rates and fewer bleeding problems compared with the 70-W device. The 120-W thulium laser has a wavelength of 2013 nm, which is close to the absorption peak of water. Laser energy is electively absorbed by water, which is ubiquitous in all tissues. Thus, the effects are independent of tissue vascularization, which provides good hemostasis, comparable to that of the holmium:YAG laser. Additionally, the CW laser beam in Tm:YAG lasers provides precise incisions and sufficient vaporization [23]. In the study by Bach et al. [22] in 2010 and the study by Fu et al. [5] in 2010 about ThuVARP, the authors did not indicate the vaporized prostate volume. In our study written by Netsch et al. [24] in 2015 comparing ThuVARP and ThuVEP, the amount of vaporized tissue was 42.5 mL. The amount of vaporized tissue in our current study was 43.1 and 43.8 mL in the different groups.
PSA is a tool that can be used to predict prostate volume and prostate growth [25]. Additionally, according to the PLESS (Proscar Long-term Efficacy and Safety) Study, PSA is valuable in predicting changes in symptoms, QoL, and Qmax [26]. In our study, PSA values were lower in the postoperative period compared with preoperative values in both groups A and B. This drop was due to tissue vaporization and the decrease in prostate volume, which also supports the amelioration of symptoms and improved QoL after surgery.
As expressed earlier, the bleeding complications during TURP in patients receiving OAP/OAC therapies are high. With technological advancements and new laser devices, the bleeding rate is expected to decrease as a result of the high ablative and coagulative power of these modalities. In our study, apart from showing low bleeding rates similar to those reported in Fu et al. [5]'s study, we also compared patients on continuous OAP/OAC therapy and patients who underwent LMWH bridging. The 2 patient groups showed no significant difference in terms of blood loss. Although the subgroup comparisons showed statistically significant differences, the drop in hemoglobin levels was similar and the differences were not clinically relevant. These results show that bridging therapy with LMWH has a small impact on bleeding complications that have clinical relevance. Although no cardiopulmonary adverse events occurred in our patient cohort, the high level of evidence in the literature about the seriousness of these events draws great attention to patients undergoing bridging therapy with LMWH. When the low risk due to bleeding complications in our patient group is combined with the high risk of potential cardiopulmonary adverse events in the case of LMWH bridging, continuing OAP/OAC therapy during ThuVARP seems to be the better way of patient management.
Apart from bleeding complications, we did not encounter any bladder neck contracture or urethral stricture in the long-term follow-up. These complications are also rarely reported in the literature. Fu et al. [5] reported one case of urethral stricture and no cases of bladder neck contracture in their study. In a recent study by Yu et al. [27], the authors reported a UTI rate of 10%. In our study, 4 of 47 patients in group A (8.5%) and 7 of 56 patients in group B (12.5%) had UTI. If the whole group is considered, the rate of UTI was 10.6%. These results agree with the results of Yu et al. [27]. In Fu et al. [5]'s study, the rate of UTI in ThuVARP group was 6.9%.
The main limitation of this study was its retrospective design. Another minor limitation was the lack of comparison with a group of patients undergoing TURP, which is considered the gold standard operation for BPE. However, in the absence of good prospective trials, the results obtained from well-analyzed retrospective studies can play significant roles in clinical practice and can set up new ideas for future randomized prospective studies.
Continuing OAP/OAC therapy regimens for certain endoscopic procedures has been strongly encouraged in recent years owing to the high risk of cardiopulmonary thromboembolic events in these patients. Our study showed the safety profile of continuing different OAP/OAC regimens in terms of bleeding problems in patients undergoing ThuVARP. We strongly recommend abandoning LMWH bridging and maintaining the patient's current OAP/OAC therapy regimen. A larger cohort of patients and a longer follow-up period are needed to add more valuable contributions to our results.
References
1. Rassweiler J, Teber D, Kuntz R, Hofmann R. Complications of transurethral resection of the prostate (TURP)--incidence, management, and prevention. Eur Urol. 2006; 50:969–979. PMID: 16469429.

2. Macchione L, Mucciardi G, Gali' A, Di Benedetto A, Butticè S, Magno C. Efficacy and safety of prostate vaporesection using a 120-W 2-μm continuous-wave Tm:YAG laser (RevoLix 2) in patients on continuous oral anticoagulant or antiplatelet therapy. Int Urol Nephrol. 2013; 45:1545–1551. PMID: 23892531.

3. Rieken M, Ebinger Mundorff N, Bonkat G, Wyler S, Bachmann A. Complications of laser prostatectomy: a review of recent data. World J Urol. 2010; 28:53–62. PMID: 20052586.

4. Herrmann TR, Bach T, Imkamp F, Georgiou A, Burchardt M, Oelke M, et al. Thulium laser enucleation of the prostate (ThuLEP): transurethral anatomical prostatectomy with laser support. Introduction of a novel technique for the treatment of benign prostatic obstruction. World J Urol. 2010; 28:45–51. PMID: 20063164.

5. Fu WJ, Zhang X, Yang Y, Hong BF, Gao JP, Cai W, et al. Comparison of 2-microm continuous wave laser vaporesection of the prostate and transurethral resection of the prostate: a prospective nonrandomized trial with 1-year follow-up. Urology. 2010; 75:194–199. PMID: 19819535.
6. Cornu JN, Ahyai S, Bachmann A, de la Rosette J, Gilling P, Gratzke C, et al. A systematic review and meta-analysis of functional outcomes and complications following transurethral procedures for lower urinary tract symptoms resulting from benign prostatic obstruction: an update. Eur Urol. 2015; 67:1066–1096. PMID: 24972732.

7. von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies. Int J Surg. 2014; 12:1495–1499. PMID: 25046131.

8. Gratzke C, Bachmann A, Descazeaud A, Drake MJ, Madersbacher S, Mamoulakis C, et al. EAU Guidelines on the assessment of non-neurogenic male lower urinary tract symptoms including benign prostatic obstruction. Eur Urol. 2015; 67:1099–1109. PMID: 25613154.

9. Reich O, Bachmann A, Siebels M, Hofstetter A, Stief CG, Sulser T. High power (80 W) potassium-titanyl-phosphate laser vaporization of the prostate in 66 high risk patients. J Urol. 2005; 173:158–160. PMID: 15592063.

10. Capodanno D, Musumeci G, Lettieri C, Limbruno U, Senni M, Guagliumi G, et al. Impact of bridging with perioperative low-molecular-weight heparin on cardiac and bleeding outcomes of stented patients undergoing non-cardiac surgery. Thromb Haemost. 2015; 114:423–431. PMID: 26017898.

11. Rose AJ, Allen AL, Minichello T. A call to reduce the use of bridging anticoagulation. Circ Cardiovasc Qual Outcomes. 2016; 9:64–67. PMID: 26715651.

12. Taylor K, Filgate R, Guo DY, Macneil F. A retrospective study to assess the morbidity associated with transurethral prostatectomy in patients on antiplatelet or anticoagulant drugs. BJU Int. 2011; 108(Suppl 2):45–50. PMID: 22085127.

13. Dotan ZA, Mor Y, Leibovitch I, Varon D, Golomb J, Duvdevani M, et al. The efficacy and safety of perioperative low molecular weight heparin substitution in patients on chronic oral anticoagulant therapy undergoing transurethral prostatectomy for bladder outlet obstruction. J Urol. 2002; 168:610–613. PMID: 12131319.

14. Parr NJ, Loh CS, Desmond AD. Transurethral resection of the prostate and bladder tumour without withdrawal of warfarin therapy. Br J Urol. 1989; 64:623–625. PMID: 2627636.

15. Ong WL, Koh TL, Fletcher J, Gruen R, Royce P. Perioperative management of antiplatelets and anticoagulants among patients undergoing elective transurethral resection of the prostate: a single institution experience. J Endourol. 2015; 29:1321–1327. PMID: 26154769.
16. Pariser JJ, Famakinwa OJ, Pearce SM, Chung DE. High-power thulium laser vaporization of the prostate: short-term outcomes of safety and effectiveness. J Endourol. 2014; 28:1357–1362. PMID: 24936718.

18. Berger J, Robert G, Descazeaud A. Laser treatment of benign prostatic hyperplasia in patients on oral anticoagulant therapy. Curr Urol Rep. 2010; 11:236–241. PMID: 20461480.

19. Hirayama T, Shitara T, Fujita T, Iwamura M, Kubo S, Baba S. Holmium laser enucleation of the prostate (HoLEP) in patients with continuous oral anticoagulation: first reported cases in Japan. Nihon Hinyokika Gakkai Zasshi. 2010; 101:754–757. PMID: 21174742.

20. Chung DE, Wysock JS, Lee RK, Melamed SR, Kaplan SA, Te AE. Outcomes and complications after 532 nm laser prostatectomy in anticoagulated patients with benign prostatic hyperplasia. J Urol. 2011; 186:977–981. PMID: 21791350.

21. Ruszat R, Wyler S, Forster T, Reich O, Stief CG, Gasser TC, et al. Safety and effectiveness of photoselective vaporization of the prostate (PVP) in patients on ongoing oral anticoagulation. Eur Urol. 2007; 51:1031–1038. PMID: 16945475.

22. Bach T, Huck N, Wezel F, Häcker A, Gross AJ, Michel MS. 70 vs 120 W thulium:yttrium-aluminium-garnet 2 microm continuous-wave laser for the treatment of benign prostatic hyperplasia: a systematic ex-vivo evaluation. BJU Int. 2010; 106:368–372. PMID: 19912204.
23. Bach T, Herrmann TR, Ganzer R, Burchardt M, Gross AJ. RevoLix vaporesection of the prostate: initial results of 54 patients with a 1-year follow-up. World J Urol. 2007; 25:257–262. PMID: 17530258.

24. Netsch C, Magno C, Buttice S, Macchione L, Mucciardi G, Herrmann TR, et al. Thulium vaporesection of the prostate and thulium vapoenucleation of the prostate in patients on oral anticoagulants: a retrospective three-centre matched-paired comparison. Urol Int. 2016; 96:421–426. PMID: 27197739.

25. Roehrborn CG, McConnell J, Bonilla J, Rosenblatt S, Hudson PB, Malek GH, et al. Serum prostate specific antigen is a strong predictor of future prostate growth in men with benign prostatic hyperplasia. PROSCAR long-term efficacy and safety study. J Urol. 2000; 163:13–20. PMID: 10604304.
26. Roehrborn CG, Boyle P, Bergner D, Gray T, Gittelman M, Shown T, et al. Serum prostate-specific antigen and prostate volume predict long-term changes in symptoms and flow rate: results of a four-year, randomized trial comparing finasteride versus placebo. PLESS Study Group. Urology. 1999; 54:662–669. PMID: 10510925.
27. Yu H, Zhang Z, Zhu Y, Chen J, Jiang X, Meng H, et al. Long-term outcome following thulium vaporesection of the prostate. Lasers Surg Med. 2016; 48:505–510. PMID: 26940981.

Fig. 1
The postoperative hemoglobin (Hb) levels (g/dL) of patients and the drop in hemoglobin levels. Group A, patients switched from OAP/OAC to low molecular weight heparin; group B, patients undergoing surgery while on OAC/OAP therapy; OAP/OAC, oral antiplatelet/anticoagulant; ASA, acetyl salicylic acid.
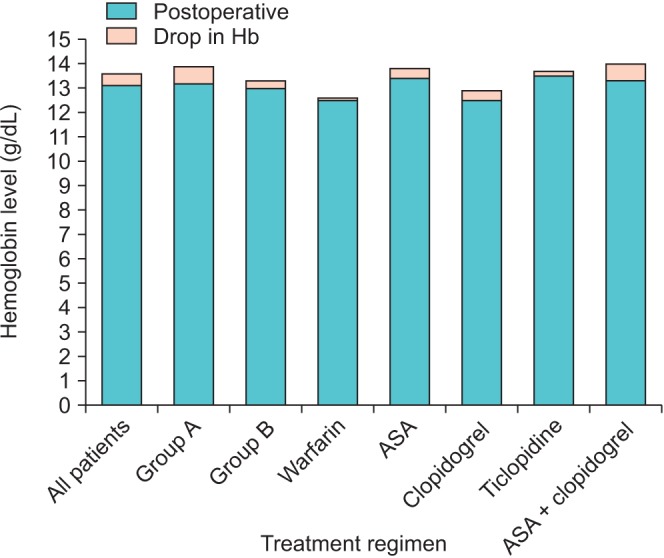
Fig. 2
International Prostate Symptom Scores (A), postmicturition residual urine (B) volumes (mL), maximal flow rate (C) values, and quality of life (D) scores of patients (mean±standard deviation). Group A, patients switched from OAP/OAC to low molecular weight heparin; group B, patients undergoing surgery while on OAC/OAP therapy; OAP/OAC, oral antiplatelet/anticoagulant; ASA, acetyl salicylic acid.
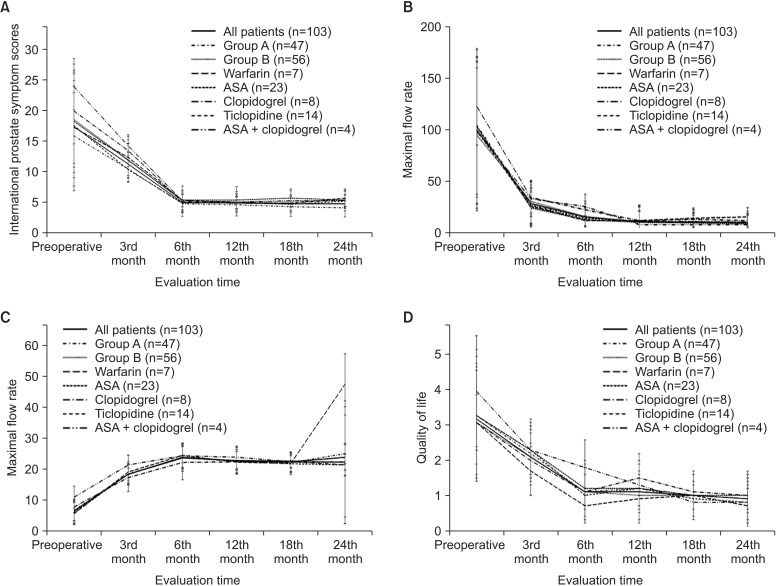
Table 1
Mean preoperative prostate volume, amount of resected prostate tissue, and duration of operation, hospitalization, and catheterization of patients in group A, group B, and subgroups of group B (classified according to individual OAP/OAC therapy regimen)
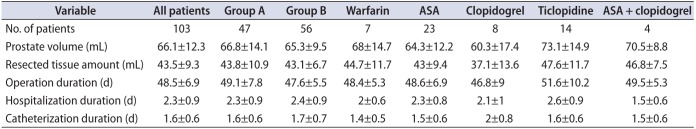
Table 2
Surgical outcomes of patients in group A, group B, and subgroups of group B (classified according to the individual OAP/OAC therapy regimen)
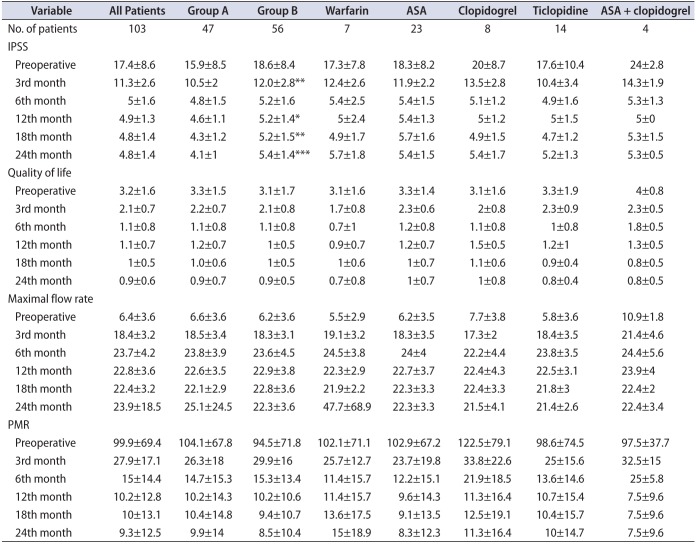
Values are presented as mean±standard deviation.
Group A, patients switched from OAP/OAC to low molecular weight heparin; group B, patients undergoing surgery while on OAC/OAP therapy; OAP/OAC, oral antiplatelet/anticoagulant; ASA, acetyl salicylic acid; IPSS, International Prostate Symptom Score; PMR, postmicturition residual urine volume.
*p<0.05 compared with group A. **p<0.005 compared with group A. ***p<0.001 compared with group A.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download