Abstract
We reported a case of port site metastasis in a 57-year-old patient who underwent video-assisted thoracic surgery (VATS) resection of pulmonary metastasis from breast cancer. Port site metastasis after VATS is very rare in patients with breast cancer. However, when suspicious lesions are detected near the port site in patients who have undergone VATS for pulmonary metastasis, port site metastasis should be considered in the differential diagnosis.
Video-assisted thoracic surgery (VATS) is a minimally invasive technique commonly used worldwide for biopsy or resection of pulmonary lesions. Although VATS procedures are considered safe and effective, they have potential complications such as prolonged air leak, bleeding, infection, and port site metastasis. Port site metastasis is a rare complication after VATS for a malignancy, with a few reports of mesothelioma cases and others (1234). However, to our knowledge, it has not been reported in a breast cancer patient in the English literature. We reported the first case of iatrogenic port site metastasis in a breast cancer patient who received VATS wedge resection for pulmonary metastasis.
We experienced a case of a 57-year-old woman who had undergone modified radical mastectomy for clinical stage T3N1M0 triple negative subtype invasive ductal carcinoma (IDC) of the right breast. After 2 years, 2 pulmonary metastatic nodules were suspected in her left lung on a surveillance positron emission tomography-computed tomography (PET-CT) scan (Fig. 1). She underwent VATS wedge resection and the lung nodules were pathologically confirmed as metastatic IDC.
Six months after VATS, she presented at our hospital with a small palpable mass on the left breast periphery. Ultrasonography (US) revealed an approximately 2.0 cm sized, circumscribed, round, and complex echoic mass with internal vascularity (on color Doppler US) in the lower outer quadrant periphery of the left breast just beneath the VATS port site scar (Fig. 2). The dif
ferential diagnosis included tumor spread, metachronous or contralateral secondary primary cancer; however, since the mass was detected at the site of the postoperative scar on the first follow-up US after VATS resection with no abnormal uptake on the previous post-op PET-CT taken 4 months prior, the possibility of postoperative organizing hematoma was also considered. Consequently, the lesion was initially rated as category 4a (low suspicion for malignancy) according to the Breast Imaging-Reporting and Data System; and as a precaution, follow up US was decided upon.
After 6 months, the follow-up US revealed an increase in the mass size (3.8 × 3 × 3.4 cm), as compared to the previous US (Fig. 3A). In addition, a consecutively performed contrast enhanced CT revealed a necrotic rim enhancing lobulated mass and PET-CT scan showed hot uptake with maximum standardized uptake value of 5.0 at the previous VATS port site (Fig. 3B, C). Subsequent excisional biopsy was confirmed histopathologically as metastatic IDC consistent with primary triple negative breast cancer (Fig. 3D). Despite complete resection of the port site metastatic mass and chemotherapy, recurrent pulmonary metastasis and pleural seeding developed and the patient expired 2 years later.
VATS is currently a routinely performed procedure for the management of metastatic lung tumor, especially when the lesion is located peripherally and is small in size and number. Compared with open thoracotomy, VATS provides many advantages: smaller incisions, better visualization of the pleural cavity, less postoperative pain, less surgical morbidity, shorter length of hospitalization, fewer adhesions, shorter interval between surgery, and adjuvant therapy with better treatment compliance. But there are also complications such as prolonged air leak, bleeding, postoperative wound infections, and metastasis at the port site (5). Port site metastasis is a rare complication of VATS for malignancy with an overall incidence of 0.12–0.57% (56). The port site metastasis is an indicator of multifocal disease with poor prognosis and increasing mortality (6). While the mechanism of port site metastasis has not been clarified, tumor implantation in the port wound is considered as the most likely cause (1). There are only a few reported cases of port site metastasis after VATS procedures for pulmonary malignancies due to pulmonary adenocarcinoma, mesothelioma, metastatic colorectal carcinoma, metastatic endometrial carcinoma, metastatic medullary thyroid carcinoma, metastatic melanoma (1234).
However, to our best knowledge, port site metastasis of breast cancer has not yet been reported in the English literature. In general, for newly developed masses in the contralateral breast after mastectomy for breast cancer, the first differential diagnosis is synchronous or metachronous breast cancer, or second primary breast cancer. This is because women with unilateral breast cancer are at an increased risk for the development of contralateral breast cancers, with a 1–5% incidence of synchronous cancer and a 3–13% incidence of metachronous cancer (78). Also, the risk of developing a second primary breast cancer for women with unilateral breast cancer is 2–6 times higher than the general population (9). In our case, a palpable mass developed at the port site, 6 months after VATS, which was detected at the first postoperative follow-up US, therefore, differential diagnosis included not only contralateral metachronous or second primary breast cancer, but also postoperative organizing hematoma. All of the above lesions can be seen as growing masses, tend to have well-demarcated contours, and can demonstrate similar sonographic findings as complex echoic lesions. However, organizing hematoma usually shows no flow on color Doppler US.
In conclusion, this was a rare case of port site metastasis after resection of pulmonary metastasis from breast cancer by VATS. Thus, radiologists should consider port site metastasis in the differential diagnosis in cases of newly developed masses at the port site following VATS for pulmonary metastasis. Furthermore, clinicians should not hesitate to perform a biopsy for early detection and diagnosis of the potentially fatal complication of port site metastasis.
Figures and Tables
Fig. 1
Positron emission tomography-computed tomography of a 59-year-old woman 2 years after mastectomy. Two pulmonary nodules are seen in the left lung with maximum standardized uptake value of 5.0, suggestive of metastasis (arrows). These nodules were confirmed as metastatic invasive ductal carcinoma from the breast after video-assisted thoracic surgery resection.
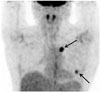
Fig. 2
Ultrasonography (US) performed 6 months after video-assisted thoracic surgery (VATS) resection for evaluation of a palpable mass in the left breast.
A. Transverse US scan reveals a 2.0 × 1.8 × 1.9 cm size, circumscribed round mass in lower outer quadrant of left breast around the port insertion area. The mass shows complex cystic and solid echogenicity. Port site scar after VATS (arrow) is demonstrated as subtle linear hypoechoic tract between the mass and the skin.
B. On color Doppler US, the mass shows internal vascularity.
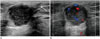
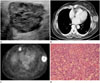
Fig. 3
Follow-up ultrasonography (US), computed tomography (CT), and positron emission tomography-computed tomography (PET-CT) after video-assisted thoracic surgery resection of pulmonary metastasis of breast cancer, and histopathologic image of surgically resected specimen.
A. The mass in the lower outer quadrant periphery of the left breast shows an increase in size, as compared with previous US performed 6 months earlier, measuring 3.8 × 3 × 3.4 cm.
B. Contrast-enhanced CT demonstrates rim enhancing lobulated mass with central necrosis at the port site.
C. PET-CT shows fluorodeoxyglucose (FDG) uptake in the periphery of the mass with maximum standardized uptake value of 5.0. An enlarged right internal mammary lymph node is also noted with increased FDG uptake.
D. Photomicrograph shows prominent nuclear pleomorphism, no identifiable tubule formation, and rampant mitotic activity, indicating invasive ductal carcinoma with nuclear grade 3 and modified Bloom-Richardson histologic grade III/III (hematoxylin and eosin stain, × 200).
References
1. Downey RJ, McCormack P, LoCicero J 3rd. Dissemination of malignant tumors after video-assisted thoracic surgery: a report of twenty-one cases. The Video-Assisted Thoracic Surgery Study Group. J Thorac Cardiovasc Surg. 1996; 111:954–960.
2. Thurer RL. Video-assisted thoracic surgery. Ann Thorac Surg. 1993; 56:199–200.
3. Walsh GL, Nesbitt JC. Tumor implants after thoracoscopic resection of a metastatic sarcoma. Ann Thorac Surg. 1995; 59:215–216.
4. Fry WA, Siddiqui A, Pensler JM, Mostafavi H. Thoracoscopic implantation of cancer with a fatal outcome. Ann Thorac Surg. 1995; 59:42–45.
5. Imperatori A, Rotolo N, Gatti M, Nardecchia E, De Monte L, Conti V, et al. Peri-operative complications of video-assisted thoracoscopic surgery (VATS). Int J Surg. 2008; 6:Suppl 1. S78–S81.
6. Johnstone PA, Rohde DC, Swartz SE, Fetter JE, Wexner SD. Port site recurrences after laparoscopic and thoracoscopic procedures in malignancy. J Clin Oncol. 1996; 14:1950–1956.
7. Jobsen JJ, van der, Ong F, Meerwaldt JH. Synchronous, bilateral breast cancer: prognostic value and incidence. Breast. 2003; 12:83–88.
8. Gao X, Fisher SG, Emami B. Risk of second primary cancer in the contralateral breast in women treated for early-stage breast cancer: a population-based study. Int J Radiat Oncol Biol Phys. 2003; 56:1038–1045.
9. Hankey BF, Curtis RE, Naughton MD, Boice JD Jr, Flannery JT. A retrospective cohort analysis of second breast cancer risk for primary breast cancer patients with an assessment of the effect of radiation therapy. J Natl Cancer Inst. 1983; 70:797–804.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download