Abstract
Purpose
Materials and Methods
Results
Figures and Tables
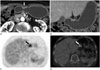 | Fig. 1A 69-year-old male with a poorly differentiated adenocarcinoma of the lesser curvature side of stomach antrum (T2 stage).
A, B. Axial and coronal CT scan show ulcerofungating with submucosal enhancing lesion (black and white arrows) in the stomach antrum.
C, D. Positron emission tomography (PET) and PET/CT fusion image show thickened stomach wall with focal strong fluorodeoxyglucose uptake lesion (black and white arrows) in the stomach antrum. The mean maximum standardized uptake values were both 4.2.
|
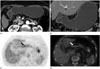 | Fig. 2A 75-year-old male with a moderately differentiated adenocarcinoma of the lesser curvature side of stomach antrum (T1a stage).
A, B. Axial and coronal CT scan show no detectable lesion in the stomach.
C, D. Positron emission tomography (PET) and PET/CT fusion image show focal fluorodeoxyglucose uptake lesion (black and white arrow) in lesser curvature side of stomach antrum. The mean maximum standardized uptake values were both 3.0.
|
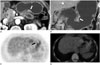 | Fig. 3A 55-year-old male with a moderately-differentiated adenocarcinoma of the posterior wall of stomach body (T3).
A, B. Axial and coronal CT scan show focal wall thickening and enhancement in the lower body of the stomach, near the angle area (white arrow).
C, D. Positron emission tomography (PET) and PET/CT fusion image obtained at the same level show no discernible fluorodeoxyglucose uptake in the stomach (black arrow).
|
Table 1
Area Under the Alternative-Free Response Receiver-Operating Characteristic Curve (Az) of S-CT Set and Combined Set in the Detection of Primary Gastric Cancers According to Invasiveness*

Note.-*Az values are mean ± standard deviation.
†Values between S-CT set and combined set were significantly different (1, p < 0.0001; 2, p = 0.0001).
‡Values between S-CT set and combined set were not significantly different (1, p = 0.6034; 2, p = 0.3546).
AGC = advanced gastric cancer, EGC = early gastric cancer, S-CT = hydro-stomach CT
Table 2
Diagnostic Performance of S-CT Set, 18F-FDG PET/CT Set, and Combined Set According to Invasiveness in Consensus Reading*

Note.-*Numbers are absolute values with percentages in parentheses. Values between each sets without superscript "†" or "‡" or "§" mark were not available or not significantly different.
†Values between S-CT set and combined set were significantly different (p < 0.0001).
‡Values between 18F-FDG PET/CT set and combined set were significantly different (1, p < 0.0001; 2, p = 0.0137; 3, p = 0.0126; 4, p = 0.0134; 5, p = 0.0108).
§Values between S-CT set and 18F-FDG PET/CT set were significantly different (1, p < 0.0001; 2, p = 0.0137; 3, p = 0.0126; 4, p = 0.0041; 5, p = 0.0134; 6, p = 0.0108; 7, p = 0.0111).
AGC = advanced gastric cancer, EGC = early gastric cancer, NPV = negative predictive value, PPV = positive predictive value, S-CT = hydro-stomach CT, 18F-FDG PET/CT = 18F-fluorodeoxyglucose positron emission tomography/computed tomography




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download