Abstract
Glossopharyngeal neuralgia with syncope is a rare syndrome. It is characterized by paroxysms of neuralgic pain in the throat and pharynx, and is associated with transient loss of consciousness caused by brief periods of cardiac arrhythmia. Glossopharyngeal neuralgia is caused by neoplasm, infection, vascular malformation, or neurovascular compression. Herein, we present a rare case of glossopharyngeal neuralgia with syncope, which was caused by an extrinsic compression of lower cranial nerves in the right cerebellomedullary cistern by the ipsilateral posterior inferior cerebellar artery. In this case, high-resolution MR imaging played a key role in the diagnosis of neurovascular compression induced glossopharyngeal neuralgia.
Glossopharyngeal neuralgia (GPN) is a rare condition with a reported incidence of 0.7/100000/year (1). It is characterized by paroxysms of lancinating pain localized unilaterally in the base of the tongue, soft palate, throat, and in the lateral and posterior regions of the pharynx, radiating to the ipsilateral ear. The pain can be triggered by swallowing, coughing, yawning, and chewing, and it usually lasts only for seconds (2). Occasionally, GPN can be associated with cardiac syncope, which is caused by asystole or bradycardia in most cases, and possibly by a vasodepressor reaction as well (2, 3). This syndrome may be referred to as vagoglossopharyngeal neuralgia when the cardiac symptoms accompany pain attacks (3). The causes of GPN are diverse and are divided in the two groups. Idiopathic type of GPN is attributed to the compression of cisternal part of glossopharyngeal nerve by intracranial vessels (4). Secondary type is associated with various lesions that affect the glossopharyngeal nerve at different levels of its path at brainstem structures, cisternal branches, and extracranial divisions (5). Here, we present a case of GPN with recurrent syncope by neurovascular compression.
A 62-year-old woman presented with a 3-year history of paroxysmal pain attacks originating from the right lower jaw and radiating to the right ear, when swallowing and brushing teeth. The duration of pain attacks lasted no longer than 1 minute. She had sudden loss of consciousness preceded by a sudden pain in the aforementioned regions, and the duration of this symptom was only a few seconds. In addition, she had recurrent episodes of 'light-headedness', which were always preceded by or appeared simultaneously with the characteristic pain. However, these episodes were not always associated with syncope. There were no concomitant symptoms, suggestive of seizure disorder. General physical, cardiovascular, and neurological examination findings were all normal. High-resolution magnetic resonance (MR) imaging for evaluation of cranial nerves revealed that the root-entry zone of the right glossopharyngeal and vagus nerves was in contact with vascular loop formed by the right posterior inferior cerebellar artery (PICA) in the right cerebellopontomedullary angle, resulting in posterior displacement of the nerves (Fig. 1). Normal left glossopharyngeal and vagus nerves were observed. MR angiography of major intracranial arteries demonstrated a tortuous vascular loop of the right PICA in the right cerebellopontomedullary angle. Once the diagnosis of GPN had been made, the patient was commenced on amitriptyline. The symptoms were improved after starting the medication.
GPN is a rare form of pain when compared to trigeminal neuralgia (0.2-1.3% of cases of facial pain). The pain of GPN is described as a cluster of unilateral attacks of sharp, stabbing, and shooting pain in the pharynx, tonsil, and posterior tongue base, radiating to the ipsilateral inner ear or mandibular angle (5). GPN usually occurs without any obvious cause, and it is divided into two types. Idiopathic GPN is caused by vascular compression of the glossopharyngeal nerve, and most of the cases belong to this type. For this lesion microvascular decompression is successful in the majority of the cases (6). Secondary type are demonstrable lesion such as Eagle's syndrome, cerebellopontine angle tumors, parapharyngeal space lesions, multiple sclerosis, arachnoiditis, posterior fossa arteriovenous malformation, direct carotid puncture, metastatic head and neck tumors, and Chiari I malformation (7).
GPN is occasionally associated with cardiac syncope, and the most acceptable mechanism is the close connection between the vagus nerve and glossopharyngeal nerve, particularly with respect to visceral sensations, which may favor the creation of a vagoglossopharyngeal reflex arch. Afferent nerve impulses from irritative or ischemic lesions arising from the peripheral course of the glossopharyngeal nerve may reach the nucleus of the tractus solitaries of the midbrain through this nerve, and via collaterals to reach the dorsal nucleus of the vagus nerve. This is the main autonomic nucleus, supplying parasympathetic fibers to the heart, bronchi, and abdominal nerve. Furthermore, the carotid sinus nerve (Hering's nerve) is responsible for the conduction of impulses from the body and the carotid sinus, and it runs in the main trunk of the glossopharyngeal nerve and ends in the dorsal nucleus of the vagus nerve (7). It has been supposed that neuralgic pain may activate the vagoglossopharyngeal reflex, resulting in bradycardia, hypotension, and syncope, and this could explain the disparity in the latent period between the beginning of neuralgia and syncope seen in the previously reported cases (8).
In the present case, GPN with recurrent syncope was caused by compression of the glossopharyngeal nerve at the root entry zone by vascular loop of the right PICA, and high-resolution MR imaging was helpful in making a diagnosis and initiating an appropriate treatment.
High-resolution MR imaging of the brain is highly sensitive and is a specific modality for revealing neurovascular compression, because it clearly demonstrates the course of the glossopharyngeal nerve as well as its relationship to arterial structures that cause vascular compression (9). The most common offending vessel has been reported to be the PICA, followed by the vertebral artery, and other vessels or combination of vessels. In the normal anatomy, there is no contact between the glossopharyngeal nerve and adjacent vessels (10).
The first line of treatment for GPN is medicine, and the surgical treatment such as microvascular decompression is proposed when appropriate medication fails to relieve the pain (5).
In conclusion, we report a rare clinical syndrome of GPN with syncope caused by neurovascular compression which was revealed by high-resolution MR imaging. This report suggests that an awareness of the pathophysiology and the normal imaging anatomy of lower cranial nerves and adjacent vessels in the cerebellopontomedullary angle may be greatly helpful for making a diagnosis in patients with atypical orofacial pain.
Figures and Tables
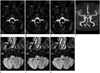
Fig. 1
A 62-year-old woman with paroxysmal pain attacks originating from the right lower jaw.
A-C. Axial 3D T2 VISTA images reveal the root-entry zone of the right glossopharyngeal and vagus nerves (white arrows) in contact with vascular loop of the right posterior inferior cerebellar artery (PICA) (black arrows) in the right cerebellopontomedullary angle, resulting posterior displacement of nerve complex. Normal left glossopharyngeal and vagus nerves (arrowheads) is seen, and no vessel is in contact with these nerves at this location.
D. 3D TOF MR angiography demonstrates the right PICA which arises from the vertebral artery and forms tortuous vascular looping (arrow).
E-G. 3D FLAIR VISTA images depict posterior displacement of the right glossopharyngeal and vagus nerves (arrows). On the left side, normal glossopharyngeal and vagus nerves is also seen (arrowheads).
References
1. Manzoni GC, Torelli P. Epidemiology of typical and atypical craniofacial neuralgias. Neurol Sci. 2005; 26:Suppl 2. s65–s67.
2. Johnston RT, Redding VJ. Glossopharyngeal neuralgia associated with cardiac syncope: long term treatment with permanent pacing and carbamazepine. Br Heart J. 1990; 64:403–405.
3. Kanpolat Y, Savas A, Batay F, Sinav A. Computed tomography-guided trigeminal tractotomy-nucleotomy in the management of vagoglossopharyngeal and geniculate neuralgias. Neurosurgery. 1998; 43:484–489. discussion 490.
4. Kandan SR, Khan S, Jeyaretna DS, Lhatoo S, Patel NK, Coakham HB. Neuralgia of the glossopharyngeal and vagal nerves: long-term outcome following surgical treatment and literature review. Br J Neurosurg. 2010; 24:441–446.
5. Singh PM, Kaur M, Trikha A. An uncommonly common: glossopharyngeal neuralgia. Ann Indian Acad Neurol. 2013; 16:1–8.
6. Gaitour E, Nick ST, Roberts C, Gonzalez-Toledo E, Munjampalli S, Minagar A, et al. Glossopharyngeal neuralgia secondary to vascular compression in a patient with multiple sclerosis: a case report. J Med Case Rep. 2012; 6:213.
7. Ferrante L, Artico M, Nardacci B, Fraioli B, Cosentino F, Fortuna A. Glossopharyngeal neuralgia with cardiac syncope. Neurosurgery. 1995; 36:58–63. discussion 63.
8. Elias J, Kuniyoshi R, Carloni WV, Borges MR, Peixoto CA, Pimentel D. Glossopharyngeal neuralgia associated with cardiac syncope. Arq Bras Cardiol. 2002; 78:510–519.
9. Boch AL, Oppenheim C, Biondi A, Marsault C, Philippon J. Glossopharyngeal neuralgia associated with a vascular loop demonstrated by magnetic resonance imaging. Acta Neurochir (Wien). 1998; 140:813–818.
10. Hiwatashi A, Matsushima T, Yoshiura T, Tanaka A, Noguchi T, Togao O, et al. MRI of glossopharyngeal neuralgia caused by neurovascular compression. AJR Am J Roentgenol. 2008; 191:578–581.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download