Abstract
Microglandular adenosis of the breast is a benign proliferative lesion, and is a rare subtype of adenosis. The pathologic findings and clinical symptoms can mimic those of breast cancer. Microglandular adenosis has been frequently associated with invasive carcinoma and in situ carcinoma of the breast. Many reports have described a spectrum of the lesions, ranging from microglandular adenosis to cancer arising from microglandular adenosis. However, most of them have focused on the pathology, and there are a few cases that report imaging findings. In the present case, we report the imaging and pathologic findings of a ductal carcinoma in situ arising in microglandular adenosis. A 57-year-old woman detected a palpable mass in her left breast. Mammogram showed an ill-defined irregular isodense mass, and sonogram showed hyperechoic irregular mass with indistinct margin. The patient underwent breast conserving surgery and adjuvant radiotherapy.
Microglandular adenosis (MGA) of the breast is a rare benign proliferative disease of the breast (1). The condition arises due to infiltrative proliferation of small round glands lacking myoepithelial cell layers, which may mimic breast cancer. This subtype of adenosis has been more frequently associated with invasive carcinoma and in situ carcinoma than the other forms of adenosis in the breast. Several reports have described a spectrum of the lesions, ranging from MGA to atypical MGA and cancer arising from MGA (23). In the present work, we report the imaging and pathologic findings of a ductal carcinoma in situ arising in MGA.
A 57-year-old woman visited our hospital for the operation of a palpable mass in the left breast. She detected the mass incidentally 1 month ago and underwent core biopsy concerning the mass in a local clinic. The histology report said that the section showed nodular growth consisted of lobules of mature adipocytes and hyperplastic lobules, suggestive of hamartoma. She wanted to remove the mass because of its large size. The mass was firm and ill-defined on palpation. Surgeon performed a simple lumpectomy about the mass, based on the pathology report. The mass was hardly separated from the surrounding tissue during the operation. The confirmed pathology was a ductal carcinoma in situ arising in MGA. We reviewed the imaging study.
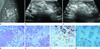
Mammogram showed an ill-defined irregular isodense mass in the left breast (Fig. 1A). Some amount of fat was present within the lesion but no distinct margin or pseudocapsule was present. Suspicious microcalcifications were not noted in the mass. On a sonogram, a 4.5-cm irregular mass was demonstrated with indistinct margin in the left outer peripheral breast. The echogenicity of the mass was predominantly hyperechoic with some heterogeneously isoechoic areas (Fig. 1B, C). Abnormal lymph nodes were not noted in both axillae.
Consequently, the patient underwent breast conserving surgery of the left breast. The cut surface of the mass showed the adipose tissue with an irregular, whitish, firm area in the central portion. Microscopic findings showed round or solid glandular structures, infiltrating the fibrous septa and fatty mammary stroma. The small glands were lined by a single or multiple layers of atypical epithelial cells, as suggestive of atypical MGA. The large coalescent glands showed expansile growth with obliterated lumens and marked cellular atypia, consistent with in situ carcinoma (Fig. 1D, E). Invasion was not present. The tumor cells were positive for cytokeratin 7, S-100 protein (Fig. 1F), but negative for estrogen receptor, progesterone receptor and Her2/Neu. Most of the myoepithelial cells were negative for the smooth muscle actin on immunohistochemical staining (Fig. 1G). Permanent pathology confirmed a ductal carcinoma in situ arising in MGA. The patient was treated with adjuvant radiotherapy after the operation.
Microglandular adenosis (MGA) of the breast is an extremely rare proliferative benign condition. All of the reported cases of MGA are women subjects with the age range from 28 to 82 years, with the majority being in between 45-55 years old (45). The most common symptom of MGA is a palpable breast mass or breast thickening. Pathologically, the lesion consists of small round glandular structures with open lumens containing eosinophilic secretions, distributed in a hypocellular dense collagenous or fatty mammary stroma. A single layer of cuboidal epithelial cells lines the round glands without surrounding myoepithelial cell layer. Immunohistochemical stains demonstrate the glands of MGA, expressing S-100 protein and lacking myoepithelial layer with negative smooth muscle actin. Based on these clinical and histologic findings, MGA is likely to be confused with well-differentiated breast cancer, which presents as an aggressive histological feature. Progression to breast carcinoma has been reported in up to 27% of patients with MGA (456).
There have been several reports on imaging findings of MGA and carcinoma from MGA. Kim et al. (7) reported that MGA showed ill-defined low echoic lesion on sonogram. Sabaté et al. (8) reported the imaging findings of MGA in the BRCA1 mutation carrier. The mass showed parallel hypoechoic mass with relatively well-defined margins, irregular borders, discrete microlobulations and angular margins. The mass was presented as hyperintense on T2-weighted image and showed homogeneous intermediate contrast enhancement with gradual delay enhancement. Lee et al. (9) reported the imaging findings of invasive ductal carcinoma arising in atypical MGA. The mass was presented as hyperdense mass with indistinct margins and pleomorphic microcalcifications on a mammogram. It was incompressible on sonogram with irregular shape and hyperechogenicity.
In our case, fat component was demonstrated on both, the mammogram and sonogram. The majority of the mass has shown hyperechogenicity on a sonogram, which is an unusual finding to diagnose malignancy, because hyperechogenicity of the breast lesion is the most sensitive benign sonographic characteristics. Fat component and hyperechogenicity could be evocative of hamartoma. Choi and Ko (10) reported the invasive ductal carcinoma in a mammary hamartoma and summarized previously described 15 cases. Among the 15 cases, 12 carcinomas were located within the hamartoma and the remaining three carcinomas involved both the hamartoma and adjacent normal breast tissue. The majority of the cases showed typical appearance of hamartoma on mammogram and/or sonogram, and had suspicious findings within the hamartoma. Typical hamartoma is a circumscribed fibrofatty mass with pseudocapsule, and is easily separated from the surrounding tissue from the operation. However, the mass, presented in our case, had no typical findings of hamartoma on both, imaging and operation. The only finding similar to hamartoma was the fat component. The mass had severe adhesion with surrounding tissue on the operation, and there was not any pseudocapsule on pathology. Therefore, hamartoma related lesion could be excluded on differentiation.
Linda et al. (11) reported that 0.4% of the malignant lesions were hyperechoic and all of them were palpable. They suggested finding suspicious sonographic signs, because the hyperechoic malignant lesions were more likely to have noncircumscribed margin and nonparallel orientation. On the review of the imaging study in our case, we found the suspicious findings of an ill-defined margin and irregular shape, which we had missed on the initial evaluation. Our case was composed with intraductal carcinoma, infiltrating the fatty mammary stroma and fibrous septa. We presumed that the adipose tissue within the mass caused the sonographic hyperechogenicity.
In summary, MGA is a benign proliferative disease of the breast and has a spectrum from typical MGA to carcinoma arising from MGA. Although additional study is necessary to set up the characteristic imaging findings of this tumor, they may tend to show suspicious finding in typical MGA, as well as in carcinoma arising from MGA. Any suspicious finding should not be overlooked in an attempt to diagnose malignancy.
Figures and Tables
Fig. 1
A 57-year-old woman with a left palpable mass.
A. Mammogram shows an ill-defined irregular isodense mass on the left breast (arrows). Some amount of fat is present within the lesion.
B, C. Sonogram shows an irregular mass with indistinct margin (arrows). The echogenicity of the mass is predominantly hyperechoic with some heterogeneously isoechoic areas.
D. Microscopic finding shows solid type intraductal carcinoma extending to microglandular adenosis (H&E, × 100).
E. Transition from uncomplicated microglandular adenosis (arrows) to in situ carcinoma (arrowheads) is clearly noted (H&E, × 200).
F. The glands of microglandular adenosis and the tumor cells strongly express S-100 protein (× 200).
G. Most of myoepithelial cells are negative for smooth muscle actin on immunohistochemical staining (× 400).
References
1. Rosen PP. Adenosis and microglandular adenosis. In : Rosen PP, editor. Rosen's Breast pathology. Philadelphia: Lippincott Williams & Wilkins;2001. p. 152–161.
2. Koenig C, Dadmanesh F, Bratthauer GL, Tavassoli FA. Carcinoma arising in microglandular adenosis: an immunohistochemical analysis of 20 intraepithelial and invasive neoplasms. Int J Surg Pathol. 2000; 8:303–315.
3. Khalifeh IM, Albarracin C, Diaz LK, Symmans FW, Edgerton ME, Hwang RF, et al. Clinical, histopathologic, and immunohistochemical features of microglandular adenosis and transition into in situ and invasive carcinoma. Am J Surg Pathol. 2008; 32:544–552.
4. Rosen PP. Microglandular adenosis. A benign lesion simulating invasive mammary carcinoma. Am J Surg Pathol. 1983; 7:137–144.
5. Tavassoli FA, Norris HJ. Microglandular adenosis of the breast. A clinicopathologic study of 11 cases with ultrastructural observations. Am J Surg Pathol. 1983; 7:731–737.
6. James BA, Cranor ML, Rosen PP. Carcinoma of the breast arising in microglandular adenosis. Am J Clin Pathol. 1993; 100:507–513.
7. Kim DJ, Sun WY, Ryu DH, Park JW, Yun HY, Choi JW, et al. Microglandular adenosis. J Breast Cancer. 2011; 14:72–75.
8. Sabaté JM, Gómez A, Torrubia S, Matias-Guiu X, Alonso C, Pericay C, et al. Microglandular adenosis of the breast in a BRCA1 mutation carrier: radiological features. Eur Radiol. 2002; 12:1479–1482.
9. Lee YH, Dai YC, Lin IL, Tu CW. Young-aged woman with invasive ductal carcinoma arising in atypical microglandular adenosis: a case report. Pathol Int. 2010; 60:685–689.
10. Choi N, Ko ES. Invasive ductal carcinoma in a mammary hamartoma: case report and review of the literature. Korean J Radiol. 2010; 11:687–691.
11. Linda A, Zuiani C, Lorenzon M, Furlan A, Girometti R, Londero V, et al. Hyperechoic lesions of the breast: not always benign. AJR Am J Roentgenol. 2011; 196:1219–1224.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download