1. Renehan AG, Tyson M, Egger M, Heller RF, Zwahlen M. Body-mass index and incidence of cancer: a systematic review and meta-analysis of prospective observational studies. Lancet. 2008; 371:569–578. PMID:
18280327.

2. Wolin KY, Carson K, Colditz GA. Obesity and cancer. Oncologist. 2010; 15:556–565. PMID:
20507889.

3. Marcello MA, Cunha LL, Batista FA, Ward LS. Obesity and thyroid cancer. Endocr Relat Cancer. 2014; 21:T255–T271. PMID:
24741026.
4. De Pergola G, Silvestris F. Obesity as a major risk factor for cancer. J Obes. 2013; 2013:291546. PMID:
24073332.

5. Tresallet C, Seman M, Tissier F, Buffet C, Lupinacci RM, Vuarnesson H, et al. The incidence of papillary thyroid carcinoma and outcomes in operative patients according to their body mass indices. Surgery. 2014; 156:1145–1152. PMID:
24878452.

6. Han JM, Kim TY, Jeon MJ, Yim JH, Kim WG, Song DE, et al. Obesity is a risk factor for thyroid cancer in a large, ultrasonographically screened population. Eur J Endocrinol. 2013; 168:879–886. PMID:
23513231.

7. Marcello MA, Sampaio AC, Geloneze B, Vasques AC, Assumpcao LV, Ward LS. Obesity and excess protein and carbohydrate consumption are risk factors for thyroid cancer. Nutr Cancer. 2012; 64:1190–1195. PMID:
23163848.

8. Zhao ZG, Guo XG, Ba CX, Wang W, Yang YY, Wang J, et al. Overweight, obesity and thyroid cancer risk: a meta-analysis of cohort studies. J Int Med Res. 2012; 40:2041–2050. PMID:
23321160.

9. Rinaldi S, Lise M, Clavel-Chapelon F, Boutron-Ruault MC, Guillas G, Overvad K, et al. Body size and risk of differentiated thyroid carcinomas: findings from the EPIC study. Int J Cancer. 2012; 131:E1004–E1014. PMID:
22511178.

10. Xu L, Port M, Landi S, Gemignani F, Cipollini M, Elisei R, et al. Obesity and the risk of papillary thyroid cancer: a pooled analysis of three case-control studies. Thyroid. 2014; 24:966–974. PMID:
24555500.

11. Strom SS, Wang X, Pettaway CA, Logothetis CJ, Yamamura Y, Do KA, et al. Obesity, weight gain, and risk of biochemical failure among prostate cancer patients following prostatectomy. Clin Cancer Res. 2005; 11(19 Pt 1):6889–6894. PMID:
16203779.

12. Collaborative Group on Hormonal Factors in Breast Cancer. Breast cancer and hormone replacement therapy: collaborative reanalysis of data from 51 epidemiological studies of 52,705 women with breast cancer and 108,411 women without breast cancer. Lancet. 1997; 350:1047–1059. PMID:
10213546.
13. Borugian MJ, Sheps SB, Kim-Sing C, Olivotto IA, Van Patten C, Dunn BP, et al. Waist-to-hip ratio and breast cancer mortality. Am J Epidemiol. 2003; 158:963–968. PMID:
14607804.

14. Protani M, Coory M, Martin JH. Effect of obesity on survival of women with breast cancer: systematic review and meta-analysis. Breast Cancer Res Treat. 2010; 123:627–635. PMID:
20571870.

15. Kim HJ, Kim NK, Choi JH, Sohn SY, Kim SW, Jin SM, et al. Associations between body mass index and clinico-pathological characteristics of papillary thyroid cancer. Clin Endocrinol (Oxf). 2013; 78:134–140. PMID:
22812676.

16. Harari A, Endo B, Nishimoto S, Ituarte PH, Yeh MW. Risk of advanced papillary thyroid cancer in obese patients. Arch Surg. 2012; 147:805–811. PMID:
22914989.

17. Paes JE, Hua K, Nagy R, Kloos RT, Jarjoura D, Ringel MD. The relationship between body mass index and thyroid cancer pathology features and outcomes: a clinicopathological cohort study. J Clin Endocrinol Metab. 2010; 95:4244–4250. PMID:
20519347.

18. Kim JY, Jung EJ, Jeong SH, Jeong CY, Ju YT, Lee YJ, et al. The indices of body size and aggressiveness of papillary thyroid carcinoma. J Korean Surg Soc. 2011; 80:241–244. PMID:
22066042.

19. Edge S, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A. AJCC cancer staging manual. 7th ed. New York: Springer;2010. p. 87–96.
20. Yim JH, Kim WB, Kim EY, Kim WG, Kim TY, Ryu JS, et al. The outcomes of first reoperation for locoregionally recurrent/persistent papillary thyroid carcinoma in patients who initially underwent total thyroidectomy and remnant ablation. J Clin Endocrinol Metab. 2011; 96:2049–2056. PMID:
21508143.

21. Hay ID, Hutchinson ME, Gonzalez-Losada T, McIver B, Reinalda ME, Grant CS, et al. Papillary thyroid microcarcinoma: a study of 900 cases observed in a 60-year period. Surgery. 2008; 144:980–987. PMID:
19041007.

22. Baudin E, Travagli JP, Ropers J, Mancusi F, Bruno-Bossio G, Caillou B, et al. Microcarcinoma of the thyroid gland: the Gustave-Roussy Institute experience. Cancer. 1998; 83:553–559. PMID:
9690549.
23. Appetecchia M, Scarcello G, Pucci E, Procaccini A. Outcome after treatment of papillary thyroid microcarcinoma. J Exp Clin Cancer Res. 2002; 21:159–164. PMID:
12148571.
24. World Health Organization. Obesity: preventing and managing the global epidemic: report of a WHO consultation on obesity. Geneva: World Health Organization;1997.
25. Korean Endocrine Society. Korean Society for the Study of Obesity. Management of obesity, 2010 recommendation. Endocrinol Metab (Seoul). 2010; 25:301–304.
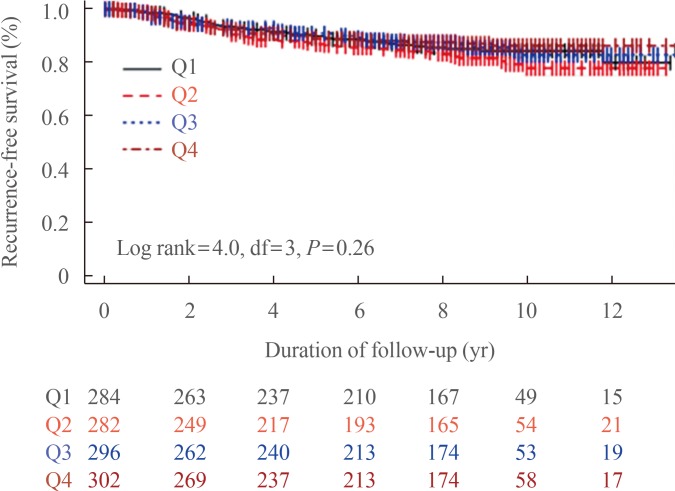
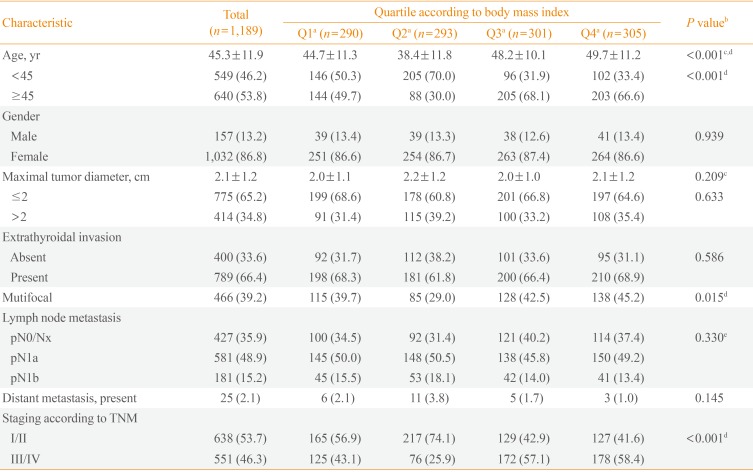
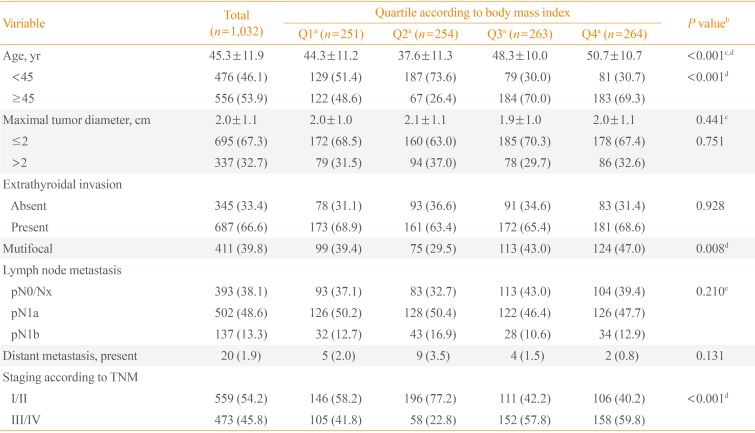
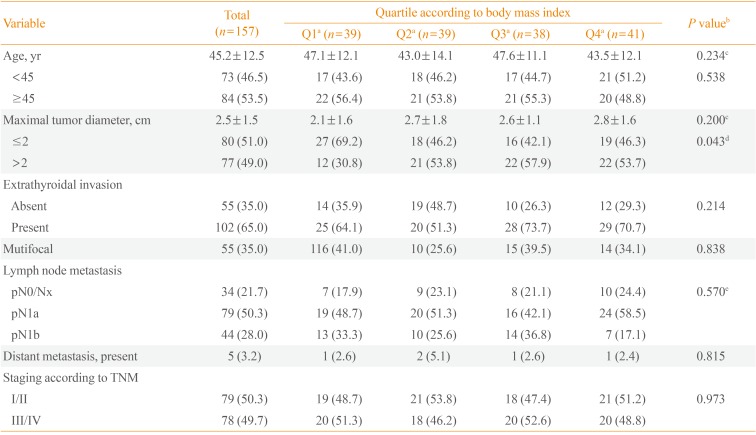




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download