Abstract
Background
The aim of this study was to assess the clinical differences between acromegalic patients with microadenoma and patients with macroadenoma, and to evaluate the predictive value of growth hormone (GH) levels for early detection of macroadenoma.
Methods
We performed a retrospective analysis of 215 patients diagnosed with a GH-secreting pituitary adenoma. The patients were divided into two groups: the microadenoma group and the macroadenoma group, and the clinical parameters were compared between these two groups. The most sensitive and specific GH values for predicting macroadenoma were selected using receiver operating characteristic (ROC) curves.
Results
Compared with the microadenoma group, the macroadenoma group had a significantly younger age, higher body mass index, higher prevalence of hyperprolactinemia and hypogonadism, and a lower proportion of positive suppression to octreotide. However, there were no significant differences in the gender or in the prevalence of diabetes between the two groups. The tumor diameter was positively correlated with all GH values during the oral glucose tolerance test (OGTT). All GH values were significantly higher in the macroadenoma group than the microadenoma group. Cut-off values for GH levels at 0, 30, 60, 90, and 120 minutes for optimal discrimination between macroadenoma and microadenoma were 5.6, 5.7, 6.3, 6.0, and 5.8 ng/mL, respectively. ROC curve analysis revealed that the GH value at 30 minutes had the highest area under the curve.
Acromegaly is associated with significant morbidity and mortality due to excess growth hormone (GH) secretion and increased circulating insulin-like growth factor 1 (IGF-1) concentrations [1]. Acromegaly is most frequently caused by a GH-secreting pituitary adenoma. Neurosurgery is considered as the first-line treatment for acromegalic patients and the efficiency of this treatment ranges between 61% to 91% for all microadenoma cases. However, because surgical success depends on the tumor size, location, and initial GH concentration, the efficiency of neurosurgical treatment in macroadenoma cases is lower and is reported to be between 23% to 53% [2,3]. In general, larger tumors are associated with higher GH and IGF-1 levels than smaller tumors [4]. Rieger et al. [5] reported that GH levels prior to surgery appeared to be an important indicator of tumor invasiveness, and thus, GH levels should be considered as a prognostic factor that can be used to plan adjuvant tumor treatment. Until now, the predictive value of glucose-suppressed GH levels for detection of macroadenoma has not been evaluated.
The preoperative clinical and biochemical characteristics of 215 acromegalic patients were reviewed, and the differences between patients with microadenoma and those with macroadenoma were the focus of this study. The study also evaluated the possible predictive value of GH levels for early detection of macroadenoma using the oral glucose tolerance test (OGTT) at the time of diagnosis.
A retrospective study was conducted at the Samsung Medical Center, Seoul, Korea and 254 patients who had been diagnosed with acromegaly between 1994 and 2008 were included in this study. Of the 254 patients, 39 were excluded from the study for the following reasons: incomplete records of biochemical analysis (28 patients) and previous pituitary surgery or radiotherapy (11 patients). The study population therefore consisted of 215 patients who initially had been diagnosed with acromegaly. None of these patients had received any treatment for acromegaly. Magnetic resonance imaging (MRI) was used to detect and measure pituitary adenomas. The patients were divided into a microadenoma group and a macroadenoma group, and an adenoma size of 10 mm was used to classify patients. Thirty-two patients presented with microadenoma at the time of acromegaly diagnosis and 183 patients presented with macroadenoma. The baseline clinical characteristics, OGTT and octreotide suppression test (OST) results were evaluated. Clinical parameters and hormone levels were compared between the two groups of study patients.
The diagnosis of acromegaly was based on a plasma GH concentration >1 ng/mL after oral administration of 75 g glucose, in association with a raised IGF-1 concentration for age and sex. The OST was performed by injecting 100 µg octreotide subcutaneously and measuring serum GH levels 0, 1, 2, and 5 hours after octreotide administration. A 50% decrease in the lowest GH level over basal GH level was regarded as a positive suppression of octreotide. Hyperprolactinemia was defined as a serum prolactin (PRL) level greater than 20 ng/mL. Hypogonadism was defined by a testosterone concentration less than 8 nmol/L in males. In females, hypogonadism was defined by a serum estradiol concentration less than 70 nmol/L or by the absence of a normal menstrual cycle in premenopausal women or inadequate response to a luteinizing hormone (LH)-releasing hormone test. Thyroid stimulating hormone (TSH) deficiency was defined as low or inappropriately normal TSH with a free T4 level below the reference range.
The insulin tolerance test was performed using a blood sample that was collected after an overnight fast. Next, insulin (0.1 to 0.2 U/kg) was injected between 9:00 AM and 10:30 AM to achieve blood glucose levels less than 40 mg/dL and until symptoms of hypoglycemia developed. Blood samples were taken from the patients at 0, 30, 60, and 90 minutes after injection.
Diabetes mellitus (DM) was diagnosed according to the American Diabetes Association 2003 criteria [6]. Several models have been applied to estimate the insulin sensitivity and secretion based on plasma glucose and insulin concentrations recorded during the OGTT. To measure basal insulin secretion, homeostasis model assessment for β-cell function (HOMA-β) was calculated as follows: HOMA-β=fasting insulin (mU/L)β/[fasting glucose (mg/dL)/18-3.5]. A homeostasis model was used to assess insulin resistance (HOMA-IR). HOMA-IR was calculated as follows: HOMA-IR=fasting insulin (mU/L)×fasting glucose (mg/dL)/405 [7]. The previously recorded fasting glucose and insulin values were used to calculate the quantitative insulin-sensitivity check index (QUICKI). QUICK=1/[log (I0)+log (G0)], where I0 is fasting insulin (µU/mL) and G0 is fasting glucose (mg/dL) [8].
The relationship between tumor diameter and GH values was investigated during OGTT. Additionally, the most sensitive and specific GH values (cutoff values) for predicting macroadenoma were determined. Positive predictive value (PPV) and negative predictive value (NPV) for macroadenoma predictions were calculated using two-by-two tables.
Serum GH levels were measured by immunoradiometric assay (IRMA), using commercially available kits (Daiichi, Tokyo, Japan). The sensitivity of the assay was 0.1 ng/mL. The intra- and interassay coefficients of variation (CVs) were less than 1.4% and 1.3%, respectively. Serum IGF-1 concentrations were determined by an IRMA (Immunotech, Marseille Cedex, France). The sensitivity of the assay was 2 ng/mL. The intra- and interassay CVs were less than 6.3% and 6.8%, respectively. Expected values based on age were: 21 to 30 years, 232 to 385 ng/mL; 31 to 40 years, 177 to 382 ng/mL; 41 to 50 years, 124 to 310 ng/mL; 51 to 60 year, 71 to 263 ng/mL. The serum levels of PRL, TSH, ACTH, cortisol, LH, and follicle stimulating hormone were measured using commercial methods.
Study groups were compared using the independent sample t test or Mann-Whitney U test. Pearson chi-square test and Fisher exact test were applied as appropriate. Data in the tables are presented as mean±SD. Correlation coefficients between tumor diameter and biochemical parameters were calculated by measuring the Spearman coefficients. Receiver operating characteristic (ROC) curves were used to select the most sensitive and specific GH values (cutoff values) for detecting macroadenomas. The statistical software packages used for this study were SPSS version 12 (SPSS Inc., Chicago, IL, USA) and GraphPad Prism version 4 (GraphPad Software, La Jolla, CA, USA). A P<0.05 was considered statistically significant.
The final study population consisted of 215 patients (110 males and 105 females). Sellar MRI revealed a pituitary microadenoma in 32 patients (14.9%) and pituitary macroadenomas in 183 patients (85.1%). The clinical, biochemical, and hormonal characteristics in each group are shown in Table 1. Patients with macroadenoma were significantly younger in age (P=0.002) and had greater height (P=0.013), weight (P=0.006), and body mass index (P=0.032) than those with microadenoma. Gender was not significantly different between the two groups. The mean basal GH and IGF-1 levels were significantly more elevated in the group with macroadenoma (P<0.001 and P=0.025, respectively). Hyperprolactinemia was more prevalent in the group with macroadenoma (P=0.005), but there was a higher proportion of patients with microadenoma (P=0.028) that had positive octreotide suppression. There were no significant differences in the prevalence of DM, glucose levels, fasting insulin levels, fasting C-peptide, HOMA-β, HOMA-IR, and QUICK between these two groups. In regards to pituitary axis insufficiency, hypogonadism was observed only in the group with macroadenoma, and there were no differences between the two groups in the incidence of hypothyroidism and hypocortisolism.
There was a positive correlation between tumor diameter and all GH values during OGTT (P<0.001), but there was no correlation between tumor diameter and IGF-1 (P=0.169) (Table 2). When patients who presented with macroadenoma and microadenoma were analyzed separately, the positive correlation between tumor diameter and GH values during OGTT was maintained in the macroadenoma group (GH 0 minute, ρ=0.375, P<0.001; GH 30 minutes, ρ=0.364, P<0.001; GH 60 minutes, ρ=0.279, P=0.001; GH 90 minutes, ρ=0.336, P<0.001; GH 120 minutes, ρ=0.294, P<0.001). However, the correlation between tumor diameter and GH values during OGTT was shown to no longer be statistically significant in the microadenoma group (GH 0 minute, ρ=0.114, P=0.563; GH 30 minutes, ρ=-0.169, P=0.4; GH 60 minutes, ρ=-0.229, P=0.240; GH 90 minutes, ρ=-0.219, P=0.272; GH 120 minutes, ρ=-0.138, P=0.484).
The GH levels after ingestion of oral glucose are shown in Fig. 1. The GH values measured at each time were significantly higher in the macroadenoma group compared to the microadenoma group (GH 0 minute, 27.8±20.7 ng/mL vs. 6.4±5.0 ng/mL; GH 30 minutes, 28.5±30.5 ng/mL vs. 5.6±4.4 ng/mL; GH 60 minutes, 31.4±31.8 ng/mL vs. 6.4±4.6 ng/mL; GH 90 minutes, 28.8±25.2 ng/mL vs. 6.6±4.8 ng/mL; GH 120 minutes, 29.3±25.8 ng/mL vs. 6.2±4.9 ng/mL; P<0.001).
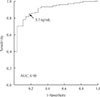
To investigate the efficacy of GH values in predicting the presence of macroadenoma in acromegalic patients, the sensitivity, specificity, PPV, and NPV were calculated at each timepoint after oral glucose was administered (Table 3). Cutoff values for GH at 0, 30, 60, 90, and 120 minutes for predicting macroadenoma from GH-secreting pituitary adenoma were 5.6, 5.7, 6.3, 6.0, and 5.8 ng/mL, respectively. The appropriate cutoff value for GH at 30 minutes was 5.7 ng/mL (sensitivity 91.7%, specificity 71.4%, PPV 94.3%, NPV 62.5%) (Fig. 2).
The aim of this study was to demonstrate the clinical and biochemical differences between acromegalic patients that presented with microadenoma and patients with macroadenoma and to ascertain whether measuring GH levels after glucose loading can predict tumor size at the time of diagnosis.
The onset of acromegaly frequently occurs during the fourth decade of life [1]. However, the age at diagnosis in macroadenomas and microadenomas remains controversial. Etxabe et al. [9] claim that patients with invasive tumors tend to present with disease at a later age, while other studies reported opposite findings [10,11]. In our study, the mean age of macroadenoma and microadenoma onset support the latter view. In addition, our study showed that patients with macroadenomas had significantly higher random GH and IGF-1 levels. This observation might reflect that patients with macroadenoma, who also tend to have severe GH levels, were diagnosed at an earlier age.
Colao et al. [12] reported the prevalence of diabetes in acromegalic patients at around 19% to 56%, depending on the specific population. Out of the 215 acromegalic patients included in this study, DM was identified in 33.3% of the patients, which is 3.5 times higher than in the general Korean population (9.1%) [13]. IR is considered to be the main pathogenic factor of glycemic abnormalities in acromegaly. Excessive GH levels induce IR by impairing the ability of insulin to both suppress liver glucose production and stimulate glucose utilization [3]. No significant differences in HOMA-β, HOMA-IR, and QUICKI were noted between the microadenoma and macroadenoma groups. Clemmons et al. [14] found a positive correlation between the degree of carbohydrate intolerance and serum concentrations of GH and IGF-1 in patients with acromegaly. In contrast, Rodrigues et al. [15] showed that acromegalic patients with and without diabetes had similar absolute GH and IGF-1 levels. According to our results, the glucose tolerance status was similar among subjects with microadenomas and macroadenomas, although both GH and IGF-1 levels were significantly higher in patients with macroadenoma. The precise mechanism underlying increased glucose and IR caused by an excess of GH is not fully understood in acromegalic patients. GH is known to stimulate the proliferation of β-cells [16]. Kasayama et al. [17] reported that impaired β-cell function is important in determining glucose tolerance in acromegalic patients. Therefore, both IR and pancreatic β-cell dysfunction appear to be involved in the pathogenesis of glucose homeostasis abnormalities. A recent study reported that DM is more common among patients with macroadenomas [11]; however, there was no significant difference in the prevalence of diabetes between the two groups in our study. One possible explanation for the discrepancies among these results might be the independent factor of age or diabetes duration. The younger age distribution in the macroadenoma group could affect the glucose tolerance status and prevalence of diabetes. However, the duration of diabetes was not investigated in this study.
Hypogonadotropic hypogonadism was found in 14.8% of the patients with macroadenomas but was not found in any of the patients with microadenomas. Gonad function impairment is a common clinical finding with acromegaly [18]. As previously reported, ganadotropin deficiency can be related to a tumor mass, hyperprolactinemia, or excess GH/IGF-1 [10,19,20]. In our study, 20 patients (74%) with gonadal dysfunction had no mass that affected gonadal function (lateral and/or downward macroadenoma development; no suprasellar expansion compressing the pituitary stalk; normal pituitary visible in each case). Hypogonadism was also found to be more common in the macroadenoma group; however, there was no significant correlation between hypogonadism and hyperprolactinemia (P=0.058). Hypogonadal patients tended to have slightly higher concentrations of both GH and IGF-1 levels, but this finding was not statistically significant. These data suggest that excess GH/IGF-1 may interfere with gonadal function, as has been suggested in the study by Colao et al. [20].
Our study demonstrated that there is a positive relationship between GH levels and tumor diameter in patients with acromegaly. The basal IGF-1 level was significantly higher in the macroadenoma group than in the microadenoma group, however, no significant association was observed between tumor diameter and IGF-1 levels. These results are likely influenced by the serum IGF-1 concentrations, which plateau at very high GH concentrations [21]. Therefore, GH levels may be more predictive values of disease severity. Based on this hypothesis, an ROC curve was created that shows the most sensitive and specific GH cutoff value for predicting macroadenoma. Among the GH values studied, the GH value measured at 30 minutes after OGTT was found to be the best predictor for macroadenoma in patients with acromegaly. The appropriate cutoff value for GH at 30 minutes was 5.7 ng/mL and yielded a 91.7% sensitivity, 71.4% specificity, 94.3% PPV, and 62.5% NPV. The overall predictive power of GH was not very high; however, OGTT is routinely used to diagnose acromegaly and is a relatively simple test to perform, and it is an accurate and readily available test in outpatient clinics. Colao et al. [22] found that patients with higher baseline GH and IGF-1 levels and larger tumors at the time of diagnosis required higher doses of somatostatin analogues (SSA). Furthermore, clinicians may opt for SSA as the primary choice of adjuvant medical treatment for active disease. Therefore initial GH levels, especially GH levels found at 30 minutes, could be considered a useful additional parameter to predict macroadenoma at the time of acromegaly diagnosis.
One of the limitations of this study was the retrospective design. There were fewer patients in the microadenoma group than the macroadenoma group, and it was difficult to secure a large study sample size because of the rareness of an acromegaly diagnosis. Third, subjects were recruited from a single tertiary care center in Korea and the GH and IGF-1 results and tumor size measurements were performed at the same center.
In conclusion, the present study demonstrated the differences between microadenoma and macroadenoma in acromegalic patients. Compared to patients with microadenoma, the patients with macroadenoma were significantly younger and had higher BMI, a higher prevalence of hyperprolactinemia and hypogonadism, and a lower proportion of positive suppression to octreotide. However, there were no statistically significant differences in the gender or in the prevalence of diabetes between two groups. In addition, the study provides insight into GH levels during OGTT. A GH level of 5.7 ng/mL or higher, found at 30 minutes, was the most useful predictor for detecting macroadenoma. Based on these results, clinicians could check GH levels at the 30 minutes time interval during OGTT. Finally, the results indicate that GH levels can be used to diagnose macroadenoma and this method could be applied to establish early treatment plans at the time of an acromegaly diagnosis.
Figures and Tables
Fig. 1
Growth hormone (GH) levels during oral glucose tolerance test in patients with microadenoma and macroadenoma, aP<0.001 vs. microadenoma.

Fig. 2
Receiver operating characteristic (ROC) curve showing macroadenoma prediction using the growth hormone (GH) level at the 30 minutes interval during the oral glucose tolerance test. The cutoff value for GH level at 30 minutes was 5.7 ng/mL, and the area under the ROC curve was 0.90 (95% confidence interval, 0.85 to 0.96). AUC, area under the curve.
References
1. Melmed S. Medical progress: acromegaly. N Engl J Med. 2006; 355:2558–2573.
2. Nomikos P, Buchfelder M, Fahlbusch R. The outcome of surgery in 668 patients with acromegaly using current criteria of biochemical 'cure'. Eur J Endocrinol. 2005; 152:379–387.
3. Swearingen B, Barker FG 2nd, Katznelson L, Biller BM, Grinspoon S, Klibanski A, Moayeri N, Black PM, Zervas NT. Long-term mortality after transsphenoidal surgery and adjunctive therapy for acromegaly. J Clin Endocrinol Metab. 1998; 83:3419–3426.
4. Petersenn S, Buchfelder M, Gerbert B, Franz H, Quabbe HJ, Schulte HM, Grussendorf M, Reincke M. Participants of the German Acromegaly Register. Age and sex as predictors of biochemical activity in acromegaly: analysis of 1,485 patients from the German Acromegaly Register. Clin Endocrinol (Oxf). 2009; 71:400–405.
5. Rieger A, Rainov NG, Ebel H, Sanchin L, Shibib K, Helfrich C, Hoffmann O, Burkert W. Factors predicting pituitary adenoma invasiveness in acromegalic patients. Neurosurg Rev. 1997; 20:182–187.
6. Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care. 2003; 26:Suppl 1. S5–S20.
7. Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985; 28:412–419.
8. Katz A, Nambi SS, Mather K, Baron AD, Follmann DA, Sullivan G, Quon MJ. Quantitative insulin sensitivity check index: a simple, accurate method for assessing insulin sensitivity in humans. J Clin Endocrinol Metab. 2000; 85:2402–2410.
9. Etxabe J, Gaztambide S, Latorre P, Vazquez JA. Acromegaly: an epidemiological study. J Endocrinol Invest. 1993; 16:181–187.
10. Nabarro JD. Acromegaly. Clin Endocrinol (Oxf). 1987; 26:481–512.
11. Mercado M, Espinosa de los Monteros AL, Sosa E, Cheng S, Mendoza V, Hernandez I, Sandoval C, Guinto G, Molina M. Clinical-biochemical correlations in acromegaly at diagnosis and the real prevalence of biochemically discordant disease. Horm Res. 2004; 62:293–299.
12. Colao A, Ferone D, Marzullo P, Lombardi G. Systemic complications of acromegaly: epidemiology, pathogenesis, and management. Endocr Rev. 2004; 25:102–152.
13. Choi YJ, Kim HC, Kim HM, Park SW, Kim J, Kim DJ. Prevalence and management of diabetes in Korean adults: Korea National Health and Nutrition Examination Surveys 1998-2005. Diabetes Care. 2009; 32:2016–2020.
14. Clemmons DR, Van Wyk JJ, Ridgway EC, Kliman B, Kjellberg RN, Underwood LE. Evaluation of acromegaly by radioimmunoassay of somatomedin-C. N Engl J Med. 1979; 301:1138–1142.
15. Rodrigues TC, Costenaro F, Fedrizzi D, Oliveira MD, Lima PB, Boschi V, Czepielewski MA. Diabetes mellitus in a cohort of patients with acromegaly. Arq Bras Endocrinol Metabol. 2011; 55:714–719.
16. Nielsen JH, Galsgaard ED, Moldrup A, Friedrichsen BN, Billestrup N, Hansen JA, Lee YC, Carlsson C. Regulation of beta-cell mass by hormones and growth factors. Diabetes. 2001; 50:Suppl 1. S25–S29.
17. Kasayama S, Otsuki M, Takagi M, Saito H, Sumitani S, Kouhara H, Koga M, Saitoh Y, Ohnishi T, Arita N. Impaired beta-cell function in the presence of reduced insulin sensitivity determines glucose tolerance status in acromegalic patients. Clin Endocrinol (Oxf). 2000; 52:549–555.
18. Melmed S, Ho K, Klibanski A, Reichlin S, Thorner M. Clinical review 75: recent advances in pathogenesis, diagnosis, and management of acromegaly. J Clin Endocrinol Metab. 1995; 80:3395–3402.
19. Asa SL, Ezzat S. The pathogenesis of pituitary tumours. Nat Rev Cancer. 2002; 2:836–849.
20. Colao A, De Rosa M, Pivonello R, Balestrieri A, Cappabianca P, Di Sarno A, Rochira V, Carani C, Lombardi G. Short-term suppression of GH and IGF-I levels improves gonadal function and sperm parameters in men with acromegaly. J Clin Endocrinol Metab. 2002; 87:4193–4197.
21. Bates AS, Evans AJ, Jones P, Clayton RN. Assessment of GH status in acromegaly using serum growth hormone, serum insulin-like growth factor-1 and urinary growth hormone excretion. Clin Endocrinol (Oxf). 1995; 42:417–423.
22. Colao A, Pivonello R, Auriemma RS, Briganti F, Galdiero M, Tortora F, Caranci F, Cirillo S, Lombardi G. Predictors of tumor shrinkage after primary therapy with somatostatin analogs in acromegaly: a prospective study in 99 patients. J Clin Endocrinol Metab. 2006; 91:2112–2118.




 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download