Abstract
Subclinical thyroid disease is defined biochemically by an abnormal thyrotropin (TSH) level and normal serum-free thyroxine level. The prevalence of this condition varies according to the reference range for TSH and geographic or demographic factors. Recently, several studies, including our community-based cohort studies, have reported on the incidence of subclinical thyroid disease in Korea. Using these studies, we reviewed the prevalence and risk factors of subclinical thyroid disease, focusing on subclinical hypothyroidism.
Subclinical thyroid disease is defined as elevated or depressed serum thyrotropin (TSH) level with a normal serum-free thyroxine (T4) level [1]. However, the optimal TSH cutoff values and the clinical significance of these subclinical abnormalities are still controversial [2]. Elevated or depressed TSH is associated with nonspecific and nonserious symptoms in most cases [3]. However, some studies have shown that the risk of cardiovascular disease increases in patients with abnormal TSH [4,5]. Therefore, the clinical impact of this condition should be verified because of its high prevalence in the general population; 3% to 12% for subclinical hypothyroidism [6,7,8,9,10,11,12,13,14,15,16,17,18] and 1% to 6% for subclinical hyperthyroidism [7,9,10,12,13,14,17,18].
The treatment of these subclinical conditions is even more controversial. Usually, treatment of subclinical hypothyroidism is considered in patients with pregnancy, infertility, associated symptoms, or high risk of progression to overt hypothyroidism [19]. In subclinical hyperthyroidism, treatment tends to be considered when the patient is old or when she or he has a risk of cardiovascular diseases [20] or osteoporosis [21] or a high risk of progression to overt hyperthyroidism [22]. Therefore, prediction of progression to overt thyroid diseases is very important for screening or treatment of these conditions. Progression to overt thyroid disease varies depending on the baseline TSH level and the presence of underlying thyroid disease. The annual progression rate of overt hypothyroidism has been reported to be 1% to 4% [23], while that of subclinical hyperthyroidism was reported to be 0.5% to 1% [24]. Another study reported that 1% or 2% of patients who show a TSH level lower than 0.1 mIU/L progress to overt hyperthyroidism per year [17]. Persons with serum TSH greater than 0.1 mIU/L in subclinical hyperthyroid status [25] or less than 8 mIU/L in subclinical hypothyroid status tend to recover spontaneously [22].
The well-known risk factors of subclinical thyroid diseases or their progression are baseline TSH level [22], old age, female sex, and the presence of thyroid autoantibodies [12,13,14,16,17]. Therefore, clinical decisions regarding screening or treatment of subclinical thyroid diseases should be based on the different risk factors depending on geographical area with various iodine status [26].
Subclinical hypothyroidism is more prevalent in relatively iodine-rich areas; 6.1% to 18.0% compared with 0.9% to 3.8% in iodine-deficient areas [26,27,28]. However, previous studies have reported a lower incidence of subclinical hypothyroidism ranging from 0.4% to 5.0% in Korea [29,30,31,32,33], despite the fact that Korea is one of the most iodine-rich areas [34]. These studies were based on people who had visited the healthcare system, thus it may not reflect the incidence of the whole community. Recently, we reported the incidence of subclinical thyroid disease in Korea based on two population-based cohort studies, the Ansung cohort and the Korean Longitudinal Study on Health and Aging (KLoSHA) study [35,36]. Using this updated data, we reviewed the prevalence and risk factors of subclinical thyroid disease in Korea as well as in other countries.
The prevalence of subclinical hypothyroidism varies with population, age, sex, race, region, and method of TSH measurement. TSH is heterogeneous with respect to both glycosylation and biological activity [2]. Thus, the normal reference range for TSH should be standardized, and an appropriate quality control procedure should be established by each laboratory [19]. Previously, a panel of experts defined the reference range of normal serum TSH concentration as 0.45 to 4.5 mIU/L based on a statistically defined reference range [19]. On the other hand, one German study that included only healthy persons with normal thyroid function and morphology based on sonography [37] proposed a normal TSH range of 0.3 to 3.7 mIU/L, which is quite lower than the range reported in the United States. The difference may be related to mild iodine-deficient status in German populations [37]. In general, subclinical hypothyroidism is defined as a TSH level greater than 4.0 to 6.0 mIU/L [12,14,16]. Table 1 summarizes the reference range of TSH and the incidence of subclinical hypothyroidism from various studies [38]. The proposed reference ranges of TSH were slightly different for each ethnic or regional population, and its prevalence in large population-based studies in many different countries has been reported to be 3% to 10% based on individual reference ranges (Table 1). In the Whickham, England survey conducted in 1977 and including 2,779 people [16], subclinical hypothyroidism was defined as elevated serum TSH above the 97.5 percentile (6 mIU/L) in the absence of obvious clinical features of hypothyroidism. They also showed that TSH level was increased to greater than 6 mIU/L in 7.5% of females and 2.8% of males [16]. TSH level did not vary with age in men but increased markedly in women over the age of 45 years.
The National Health and Nutrition Examination Survey (NHANES) III, which was performed from 1988 to 1994, included 16,533 people without thyroid disease in the United States [12]. A reference population of 13,344 people was selected based on exclusion of those who were pregnant, taking androgens or estrogens, had thyroid antibodies or showed biochemical hyperthyroidism or hypothyroidism. The 2.5th and 97.5th percentiles of TSH were 0.45 and 4.12 mIU/L, respectively [12]. With the criteria of subclinical hypothyroidism at TSH level greater than 4.5 mIU/L, 4.3% of subjects were defined as having subclinical hypothyroidism [12]. The prevalence of subclinical hypothyroidism and the positivity of antithyroid antibodies were about 2-fold greater in females, increased with age, and were about three times greater in whites than in blacks [12].
Other studies of elderly populations in the United States have reported a higher prevalence of subclinical hypothyroidism than that in NHANES III, ranging from 7.3% to 12.4% (Table 1). The Colorado thyroid disease prevalence study reported that the prevalence of abnormal TSH level greater than 5.1 mIU/L was 9.5% [14]. The Framingham study showed a prevalence of 9.9% with the upper range of TSH greater than 5.0 mIU/L [11,39]. In the Healthy Aging Study with a mean age of 74.7 years, the incidence of subclinical hypothyroidism was 12.40% for a TSH level greater than 4.5 mIU/L [15].
It is known that subclinical hypothyroidism is more prevalent in iodine-sufficient areas [26,27,28]. In a study with elderly subjects in Denmark, 3.8% of subjects in the area of low iodine intake had high serum TSH, while 18% in the area of high iodine intake showed subclinical hypothyroidism [26]. In another study with 1,061 Japanese participants, the frequency of high urine iodine correlated with hypothyroidism in the absence of autoantibodies [28]. Teng et al. [27] also observed an increase in the prevalence of subclinical hypothyroidism with increasing iodine intake in the Chinese population. Three regions with different levels of iodine intake were included; the urinary excretion of iodine was 85 µg/L in Panshan, 243 µg/L in Zhangwu, and 651 µg/L in Huanghua [27], for which the prevalence of subclinical hypothyroidism was 0.9%, 2.9%, and 6.1%, respectively. Overall, previous studies reported the prevalence of subclinical hypothyroidism to be less than 3.0% in iodine-insufficient areas, whereas the prevalence increased to over 6.0% in iodine-sufficient areas (Table 1).
In addition to the iodine intake, the prevalence of subclinical hypothyroidism may vary according to the age or sex of the population analyzed. The prevalence of subclinical hypothyroidism in an elderly group in Italy was 6.7% [7], which was higher than 3.8% in any other age group in that country [6]. Other European study has shown a higher prevalence of subclinical hypothyroidism, 10.8%, in women [8].
Korea is one of the most iodine-sufficient regions; however, previous studies based on health care examination reported the prevalence of subclinical hypothyroidism to be only 0.1% to 5.0% (Table 2). This result might be related to the fact that subjects who receive a health care examination do not represent the general population of Korea. As expected, in recent studies from two different population-based cohorts, the prevalence of subclinical hypothyroidism was reported as 11.7% in the Ansung cohort, which consisted of adults between 40 and 70 years, and 17.3% in the KLoSHA study, which included elderly subjects older than 65 years [36]. The prevalence of subclinical hypothyroidism increases with age in both men and women, with statistical significance in males in the Ansung cohort. There was no significant association with prevalence of subclinical hypothyroidism and age or sexin the KLoSHA study, which included elderly people 65 years of age or older (Fig. 1)[36].
Subclinical hypothyroidism showed a higher prevalence in women (6% to 10%) than in men (2% to 4%) in all previous studies (Table 1). It has been reported in up to 20% of women older than 60 years [11,14,17]. The reason for female predominance is still unclear. In one small study with postmenopausal women, hormone replacement therapy increased the level of thyroid binding protein and TSH [40], suggesting that estrogen is a risk factor. Higher prevalence of autoimmune thyroid diseases in women might be one possible explanation; in the Whickham survey, women older than 45 years showed a higher prevalence of subclinical hypothyroidism; however, this phenomenon virtually disappeared when persons with thyroid antibodies were excluded [16]. Pedersen et al. [41] performed a cross-sectional study of thyroperoxidase antibodies (TPO Ab) and thyroglobulin antibodies (Tg Ab) in 4,649 Danish subjects. The presence of both antibodies was more frequent in females, and the prevalence increased with age and showed a correlation with subclinical hypothyroidism.
Age is one of the most significant risk factors according to several epidemiologic studies (Table 1). Reanalysis of NHANES III showed a progressive increase in both median and 97.5 percentile levels of TSH with age [42]. The 97.5 percentile level of TSH in the 80+ group was 7.49 mIU/L, which was higher than the 4.5 mIU/L in all subjects. Therefore, if we reclassify the subjects with subclinical hypothyroidism according to the age-specific reference, more than 70% of aged subjects with subclinical hypothyroidism have a euthyroid status. The age-related difference in TSH distribution was also replicated in another cross-sectional study [43]. A 13-year follow-up of the Busselton Health surveys revealed that the mean serum TSH increased from 1.49 to 1.81 mIU/L, whereas the level of free T4 remained unchanged [44]. People with lower baseline TSH value showed the largest TSH increase [44], suggesting that the age-related TSH increase reflects an altered TSH set point or reduced TSH bioactivity rather than true thyroid dysfunction [44]. Several mechanisms have been proposed to explain this phenomenon. An age-related TSH increase could be a normal physiologic response to compensate for the decrease in TSH biological activity due to age-related changes in TSH glycosylation [42]. This might also be associated with decreased thyroxine turnover with aging [12,41]. Decreased sensitivity of the thyroid gland to TSH is another possible mechanism [42]. While the increased prevalence of thyroid autoantibodies with aging is also a possible mechanism, for the reasons mentioned above, there is a growing opinion on the importance of age-specific reference ranges of TSH [39,45,46].
Serum TPO Ab and Tg Ab are commonly found in patients with autoimmune thyroid disease [47,48]. The prevalence of subclinical hypothyroidism was associated with the presence of TPO Ab (odds ratio [OR], 8.4; 95% CI, 5.8 to 12.1; P<0.0001) [12]. The Whickham survey and NHANES III studies showed similar tendencies for higher prevalence of subclinical hypothyroidism in subgroups positive for TPO Ab [11,17]. In the Whickham survey, subjects with positive thyroid antibodies showed higher TSH levels than subjects without antibodies, and overall, 60% of subjects with TSH level greater than 6 mIU/L and 80% of those with TSH greater than 10 mIU/L had thyroid antibodies [16]. In the NHANES III study, TPO Ab were present in women (17%) and men (8.7%), and the positivity increased with age and was more prevalent in whites than in blacks [12]. Moreover, in the 20-year follow-up Whickham survey, the ORs of developing overt hypothyroidism were four times higher in subjects with positive antithyroid antibodies and increased TSH level than in those with only elevated TSH level [25]. In our study, the percentage of positive TPO Ab was higher in the group with subclinical hypothyroidism (Ansung, 21.4% vs. 7.4%, P<0.001; KLoSHA, 23.3% vs. 10.1%, P<0.001) [36].
Another risk factor is the iodine intake. Excessive iodine intake could decrease thyroid function via a direct toxic effect [49] or via immunological alterations [50]. As shown in Table 1, subclinical hypothyroidism is more common in iodine-sufficient areas (4.3% to 12.4%) [11,12,13,14,15] than in iodine-deficient areas (3.8% to 6.7%) [6,7]. In cross-sectional baseline studies from Denmark and Japan, the prevalence of hypothyroidism was positively associated with iodine intake [26,28]. A recent study in China, which consisted of three cohorts in three regions with different iodine intake, also showed a significant association between iodine intake and hypothyroidism. In addition, a follow-up study showed that, among subjects with high levels of TPO Ab or Tg Ab, the rate of progression to hypothyroidism was directly correlated with iodine intake [27].
With regard to other risk factors of subclinical hypothyroidism, racial differences [12,37], cigarette smoking [35], and cold environmental temperature [51] have also been suggested. Studies performed in the United States have shown that serum TSH concentrations are higher in whites than in blacks [12,13]. The NHANES III survey demonstrated that active smokers had a TSH distribution shifted toward lower levels [52]. Pedersen et al. [53] showed that smoking was negatively associated with thyroid autoantibodies, particularly with positive Tg Ab. We also demonstrated that cigarette smoking was negatively associated with subclinical hypothyroidism [35]. Through competitive inhibition of thyrotoxic iodine binding to sodium-iodide symporter in thyroid follicular cells, thiocyanate inhibits iodide transport, organification and release of thyroid hormone from the thyroid gland [54]. Nicotine may also shift the autoimmune profile from cytotoxic Th1 and Th17 responses to protective Th2 responses [55], hence reducing the occurrence of autoimmune thyroiditis [56]. In our study, TSH level was the lowest in current smokers, in whom high iodine intake was not a risk factor for subclinical hypothyroidism [35]. Recently, anatabine, a compound in tobacco, was shown to significantly reduce Tg Ab levels in patients with Hashimoto thyroiditis participating in a randomized placebo-controlled study [57]. This result may support the application of anatabine in the treatment of hypothyroidism [57].
Lastly, subclinical hypothyroidism has been reported to be related to cold weather [51]. We observed that seasonal changes in TSH level increased 1.4-fold during the winter-spring season in Korea and then normalized during the summer-fall season [51]. This seasonal variation in TSH could be explained by adaptive thermogenesis, which is minimized at ambient temperature [58]. Alterations in photoperiod also could affect the TSH level via melatonin, which regulates multiple neuroendocrine pathways in mammals [59]. Hence, in Korea, which shows a continental climate with substantial temperature differences between winter and summer, the seasonal change should be considered in the interpretation of TSH level and subclinical thyroid disease [51].
Subclinical hyperthyroidism is a less common condition than subclinical hypothyroidism, and the reported prevalence in adult populations is between 0.2% to 9.7%, generally about 2% to 3% (Table 3). While subclinical hyperthyroidism is the opposite condition to subclinical hypothyroidism, the high-risk subjects are similar, and it is also more common in women and the elderly [39]. In the NHANES III study, the proportion of subclinical hyperthyroidism cases based on a TSH cutoff of 0.4 mIU/L was significantly higher in women (0.4%) than in men (0.2%), and it also increased with age. Other studies with elderly subjects in the United States have shown a higher prevalence of subclinical hypothyroidism, about 2.2% to 2.5% (Table 3).
The major difference in risk factors between hyperthyroidism and hypothyroidism is the amount of iodine intake; hyperthyroidism is more common in iodine-insufficient areas [26,27]. Thyroid autonomy is inversely correlated with the iodine intake [60]. Laurberg et al. [26] investigated the prevalence of subclinical thyroid disease in two areas of different iodine intake. Jutland, an area with lower iodine intake, showed higher prevalence of subclinical hyperthyroidism (9.7%) than Iceland, which has a higher iodine intake (1%). A similar trend was observed in a study in China. Teng et al. [27] showed a difference in prevalence among three regions with different levels of iodine intake. The prevalence of subclinical hyperthyroidism was 3.7% to 3.9% in a relatively iodine-insufficient area and 1.1% in an iodine-sufficient area [27]. The prevalence of subclinical hyperthyroidism in Korea is lower than that of other countries, possibly because Korea is one of the most iodine-sufficient areas. Previous data from Korea indicated that the prevalence of subclinical hyperthyroidism is between 0.64% and 4.6% (Table 2). We reported the prevalence of subclinical hyperthyroidism in Korea as 1.0% in the Ansung cohort and 0.2% in KLoSHA [20].
There is some controversy about the effect of ethnicity. The prevalence of subclinical hyperthyroidism was higher in blacks (0.4%) than in whites (0.1%) or Mexican Americans (0.3%) in the NHANES III survey [12], while a different study found the prevalence of subclinical hyperthyroidism to be greater in whites (3.9%) than in blacks (1.0%) [13].
Subclinical thyroid disease is defined by the reference range of TSH. The importance of this disease is now widely studied, and the data continue to accumulate. In summary, subclinical thyroid disease is more prevalent in the elderly and in women. The iodine intake is inversely correlated with thyroid function; deficient iodine intake is related with hyperthyroid status, while excessive or sufficient intake of iodine is associated with hypothyroidism. Thyroid autoantibodies, smoking status, environmental temperature and ethnicity are also risk factors for subclinical hypothyroidism. Understanding the prevalence and risk factors of subclinical thyroid disease could be a help to identify the patients for screening and/or follow-up.
References
2. Biondi B, Cooper DS. The clinical significance of subclinical thyroid dysfunction. Endocr Rev. 2008; 29:76–131. PMID: 17991805.

3. Bell RJ, Rivera-Woll L, Davison SL, Topliss DJ, Donath S, Davis SR. Well-being, health-related quality of life and cardiovascular disease risk profile in women with subclinical thyroid disease: a community-based study. Clin Endocrinol (Oxf). 2007; 66:548–556. PMID: 17371474.
4. Rodondi N, den Elzen WP, Bauer DC, Cappola AR, Razvi S, Walsh JP, Asvold BO, Iervasi G, Imaizumi M, Collet TH, Bremner A, Maisonneuve P, Sgarbi JA, Khaw KT, Vanderpump MP, Newman AB, Cornuz J, Franklyn JA, Westendorp RG, Vittinghoff E, Gussekloo J. Thyroid Studies Collaboration. Subclinical hypothyroidism and the risk of coronary heart disease and mortality. JAMA. 2010; 304:1365–1374. PMID: 20858880.

5. Gencer B, Collet TH, Virgini V, Bauer DC, Gussekloo J, Cappola AR, Nanchen D, den Elzen WP, Balmer P, Luben RN, Iacoviello M, Triggiani V, Cornuz J, Newman AB, Khaw KT, Jukema JW, Westendorp RG, Vittinghoff E, Aujesky D, Rodondi N. Thyroid Studies Collaboration. Subclinical thyroid dysfunction and the risk of heart failure events: an individual participant data analysis from 6 prospective cohorts. Circulation. 2012; 126:1040–1049. PMID: 22821943.
6. Aghini-Lombardi F, Antonangeli L, Martino E, Vitti P, Maccherini D, Leoli F, Rago T, Grasso L, Valeriano R, Balestrieri A, Pinchera A. The spectrum of thyroid disorders in an iodine-deficient community: the Pescopagano survey. J Clin Endocrinol Metab. 1999; 84:561–566. PMID: 10022416.

7. Iervasi G, Molinaro S, Landi P, Taddei MC, Galli E, Mariani F, L'Abbate A, Pingitore A. Association between increased mortality and mild thyroid dysfunction in cardiac patients. Arch Intern Med. 2007; 167:1526–1532. PMID: 17646607.

8. Hak AE, Pols HA, Visser TJ, Drexhage HA, Hofman A, Witteman JC. Subclinical hypothyroidism is an independent risk factor for atherosclerosis and myocardial infarction in elderly women: the Rotterdam Study. Ann Intern Med. 2000; 132:270–278. PMID: 10681281.

9. Gussekloo J, van Exel E, de Craen AJ, Meinders AE, Frolich M, Westendorp RG. Thyroid status, disability and cognitive function, and survival in old age. JAMA. 2004; 292:2591–2599. PMID: 15572717.

10. Walsh JP, Bremner AP, Bulsara MK, O'Leary P, Leedman PJ, Feddema P, Michelangeli V. Subclinical thyroid dysfunction and blood pressure: a community-based study. Clin Endocrinol (Oxf). 2006; 65:486–491. PMID: 16984241.

11. Sawin CT, Castelli WP, Hershman JM, McNamara P, Bacharach P. The aging thyroid. Thyroid deficiency in the Framingham Study. Arch Intern Med. 1985; 145:1386–1388. PMID: 4026469.

12. Hollowell JG, Staehling NW, Flanders WD, Hannon WH, Gunter EW, Spencer CA, Braverman LE. Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab. 2002; 87:489–499. PMID: 11836274.

13. Bagchi N, Brown TR, Parish RF. Thyroid dysfunction in adults over age 55 years. A study in an urban US community. Arch Intern Med. 1990; 150:785–787. PMID: 2109585.

14. Canaris GJ, Manowitz NR, Mayor G, Ridgway EC. The Colorado thyroid disease prevalence study. Arch Intern Med. 2000; 160:526–534. PMID: 10695693.

15. Rodondi N, Newman AB, Vittinghoff E, de Rekeneire N, Satterfield S, Harris TB, Bauer DC. Subclinical hypothyroidism and the risk of heart failure, other cardiovascular events, and death. Arch Intern Med. 2005; 165:2460–2466. PMID: 16314541.

16. Tunbridge WM, Evered DC, Hall R, Appleton D, Brewis M, Clark F, Evans JG, Young E, Bird T, Smith PA. The spectrum of thyroid disease in a community: the Whickham survey. Clin Endocrinol (Oxf). 1977; 7:481–493. PMID: 598014.

17. Parle JV, Franklyn JA, Cross KW, Jones SC, Sheppard MC. Prevalence and follow-up of abnormal thyrotrophin (TSH) concentrations in the elderly in the United Kingdom. Clin Endocrinol (Oxf). 1991; 34:77–83. PMID: 2004476.
18. Boekholdt SM, Titan SM, Wiersinga WM, Chatterjee K, Basart DC, Luben R, Wareham NJ, Khaw KT. Initial thyroid status and cardiovascular risk factors: the EPIC-Norfolk prospective population study. Clin Endocrinol (Oxf). 2010; 72:404–410. PMID: 19486022.

19. Surks MI, Ortiz E, Daniels GH, Sawin CT, Col NF, Cobin RH, Franklyn JA, Hershman JM, Burman KD, Denke MA, Gorman C, Cooper RS, Weissman NJ. Subclinical thyroid disease: scientific review and guidelines for diagnosis and management. JAMA. 2004; 291:228–238. PMID: 14722150.
20. Sgarbi JA, Villaca FG, Garbeline B, Villar HE, Romaldini JH. The effects of early antithyroid therapy for endogenous subclinical hyperthyroidism in clinical and heart abnormalities. J Clin Endocrinol Metab. 2003; 88:1672–1677. PMID: 12679455.

21. Faber J, Jensen IW, Petersen L, Nygaard B, Hegedus L, Siersbaek-Nielsen K. Normalization of serum thyrotrophin by means of radioiodine treatment in subclinical hyperthyroidism: effect on bone loss in postmenopausal women. Clin Endocrinol (Oxf). 1998; 48:285–290. PMID: 9578817.

22. Imaizumi M, Sera N, Ueki I, Horie I, Ando T, Usa T, Ichimaru S, Nakashima E, Hida A, Soda M, Tominaga T, Ashizawa K, Maeda R, Nagataki S, Akahoshi M. Risk for progression to overt hypothyroidism in an elderly Japanese population with subclinical hypothyroidism. Thyroid. 2011; 21:1177–1182. PMID: 21877935.

24. Bahn RS, Burch HB, Cooper DS, Garber JR, Greenlee MC, Klein I, Laurberg P, McDougall IR, Montori VM, Rivkees SA, Ross DS, Sosa JA, Stan MN. American Thyroid Association. American Association of Clinical Endocrinologists. Hyperthyroidism and other causes of thyrotoxicosis: management guidelines of the American Thyroid Association and American Association of Clinical Endocrinologists. Endocr Pract. 2011; 17:456–520. PMID: 21700562.

25. Vanderpump MP, Tunbridge WM, French JM, Appleton D, Bates D, Clark F, Grimley Evans J, Hasan DM, Rodgers H, Tunbridge F, Young ET. The incidence of thyroid disorders in the community: a twenty-year follow-up of the Whickham Survey. Clin Endocrinol (Oxf). 1995; 43:55–68. PMID: 7641412.

26. Laurberg P, Pedersen KM, Hreidarsson A, Sigfusson N, Iversen E, Knudsen PR. Iodine intake and the pattern of thyroid disorders: a comparative epidemiological study of thyroid abnormalities in the elderly in Iceland and in Jutland, Denmark. J Clin Endocrinol Metab. 1998; 83:765–769. PMID: 9506723.

27. Teng W, Shan Z, Teng X, Guan H, Li Y, Teng D, Jin Y, Yu X, Fan C, Chong W, Yang F, Dai H, Yu Y, Li J, Chen Y, Zhao D, Shi X, Hu F, Mao J, Gu X, Yang R, Tong Y, Wang W, Gao T, Li C. Effect of iodine intake on thyroid diseases in China. N Engl J Med. 2006; 354:2783–2793. PMID: 16807415.

28. Konno N, Makita H, Yuri K, Iizuka N, Kawasaki K. Association between dietary iodine intake and prevalence of subclinical hypothyroidism in the coastal regions of Japan. J Clin Endocrinol Metab. 1994; 78:393–397. PMID: 8106628.

29. Chung JH, Kim BJ, Choi YH, Shin MH, Kim SH, Min YK, Lee MS, Lee MG, Kim KW. Prevalence of thyrotoxicosis and hypothyroidism in the subjects for health check-up. J Korean Soc Endocrinol. 1999; 14:301–313.
30. Choi JK, Kim JS, Hwang CH, Kim DH, Hwang SW, Cho B, Jul BY. Subclinical hypothyroidism and coronary risk factors in women above 55 years. J Korean Acad Fam Med. 2002; 23:96–103.
31. Oh MK, Cheon KS, Jung SM, Ryu DS, Park MS, Cheong SS, Kim JS, Park BG. Prevalence of thyroid diseases among adult for health check-up in a Youngdong area of Kwangwon province. J Korean Acad Fam Med. 2001; 22:1363–1374.
32. Jung CH, Sung KC, Shin HS, Rhee EJ, Lee WY, Kim BS, Kang JH, Kim H, Kim SW, Lee MH, Park JR, Kim SW. Thyroid dysfunction and their relation to cardiovascular risk factors such as lipid profile, hsCRP, and waist hip ratio in Korea. Korean J Intern Med. 2003; 18:146–153. PMID: 14619383.

33. Lee WY, Oh KW, Rhee EJ, Jung CH, Kim SW, Yun EJ, Tae HJ, Baek KH, Kang MI, Choi MG, Yoo HJ, Park SW. Relationship between subclinical thyroid dysfunction and femoral neck bone mineral density in women. Arch Med Res. 2006; 37:511–516. PMID: 16624651.

34. Park HY, Lee SI, Kim WB, Kim SY, Cho BY, Lee HK, Koh CS. A study on the urinary iodine excretion in normal subjects and patients with thyroid disease. J Korean Soc Endocrinol. 1995; 10:386–394.
35. Cho NH, Choi HS, Kim KW, Kim HL, Lee SY, Choi SH, Lim S, Park YJ, Park do J, Jang HC, Cho BY. Interaction between cigarette smoking and iodine intake and their impact on thyroid function. Clin Endocrinol (Oxf). 2010; 73:264–270. PMID: 20105185.
36. Choi HS, Park YJ, Kim HK, Choi SH, Lim S, Park DJ, Jang HC, Cho NH, Cho BY. Prevalence of subclinical hypothyroidism in two population based-cohort: Ansung and KLoSHA cohort in Korea. J Korean Thyroid Assoc. 2010; 3:32–40.
37. Völzke H, Alte D, Kohlmann T, Ludemann J, Nauck M, John U, Meng W. Reference intervals of serum thyroid function tests in a previously iodine-deficient area. Thyroid. 2005; 15:279–285. PMID: 15785248.

38. de Benoist B, McLean E, Andersson M, Rogers L. Iodine deficiency in 2007: global progress since 2003. Food Nutr Bull. 2008; 29:195–202. PMID: 18947032.

39. Sawin CT, Geller A, Kaplan MM, Bacharach P, Wilson PW, Hershman JM. Low serum thyrotropin (thyroid-stimulating hormone) in older persons without hyperthyroidism. Arch Intern Med. 1991; 151:165–168. PMID: 1985591.

40. Arafah BM. Increased need for thyroxine in women with hypothyroidism during estrogen therapy. N Engl J Med. 2001; 344:1743–1749. PMID: 11396440.

41. Pedersen IB, Knudsen N, Jorgensen T, Perrild H, Ovesen L, Laurberg P. Thyroid peroxidase and thyroglobulin autoantibodies in a large survey of populations with mild and moderate iodine deficiency. Clin Endocrinol (Oxf). 2003; 58:36–42. PMID: 12519410.

42. Surks MI, Hollowell JG. Age-specific distribution of serum thyrotropin and antithyroid antibodies in the US population: implications for the prevalence of subclinical hypothyroidism. J Clin Endocrinol Metab. 2007; 92:4575–4582. PMID: 17911171.

43. Vadiveloo T, Donnan PT, Murphy MJ, Leese GP. Age- and gender-specific TSH reference intervals in people with no obvious thyroid disease in Tayside, Scotland: the Thyroid Epidemiology, Audit, and Research Study (TEARS). J Clin Endocrinol Metab. 2013; 98:1147–1153. PMID: 23345094.

44. Bremner AP, Feddema P, Leedman PJ, Brown SJ, Beilby JP, Lim EM, Wilson SG, O'Leary PC, Walsh JP. Age-related changes in thyroid function: a longitudinal study of a community-based cohort. J Clin Endocrinol Metab. 2012; 97:1554–1562. PMID: 22344200.

45. Aggarwal N, Razvi S. Thyroid and aging or the aging thyroid? An evidence-based analysis of the literature. J Thyroid Res. 2013; 2013:481287. PMID: 24106641.

46. Garber JR, Cobin RH, Gharib H, Hennessey JV, Klein I, Mechanick JI, Pessah-Pollack R, Singer PA, Woeber KA. American Association of Clinical Endocrinologists and American Thyroid Association Taskforce on Hypothyroidism in Adults. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Endocr Pract. 2012; 18:988–1028. PMID: 23246686.

48. Mariotti S, Caturegli P, Piccolo P, Barbesino G, Pinchera A. Antithyroid peroxidase autoantibodies in thyroid diseases. J Clin Endocrinol Metab. 1990; 71:661–669. PMID: 2168432.

49. Mahmoud I, Colin I, Many MC, Denef JF. Direct toxic effect of iodide in excess on iodine-deficient thyroid glands: epithelial necrosis and inflammation associated with lipofuscin accumulation. Exp Mol Pathol. 1986; 44:259–271. PMID: 3720915.
50. Sharma RB, Alegria JD, Talor MV, Rose NR, Caturegli P, Burek CL. Iodine and IFN-gamma synergistically enhance intercellular adhesion molecule 1 expression on NOD.H2h4 mouse thyrocytes. J Immunol. 2005; 174:7740–7745. PMID: 15944276.
51. Kim TH, Kim KW, Ahn HY, Choi HS, Won H, Choi Y, Cho SW, Moon JH, Yi KH, Park do J, Park KS, Jang HC, Kim SY, Park YJ. Effect of seasonal changes on the transition between subclinical hypothyroid and euthyroid status. J Clin Endocrinol Metab. 2013; 98:3420–3429. PMID: 23771919.

52. Belin RM, Astor BC, Powe NR, Ladenson PW. Smoke exposure is associated with a lower prevalence of serum thyroid autoantibodies and thyrotropin concentration elevation and a higher prevalence of mild thyrotropin concentration suppression in the third National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab. 2004; 89:6077–6086. PMID: 15579761.

53. Pedersen IB, Laurberg P, Knudsen N, Jorgensen T, Perrild H, Ovesen L, Rasmussen LB. Smoking is negatively associated with the presence of thyroglobulin autoantibody and to a lesser degree with thyroid peroxidase autoantibody in serum: a population study. Eur J Endocrinol. 2008; 158:367–373. PMID: 18299471.

54. Fukayama H, Nasu M, Murakami S, Sugawara M. Examination of antithyroid effects of smoking products in cultured thyroid follicles: only thiocyanate is a potent antithyroid agent. Acta Endocrinol (Copenh). 1992; 127:520–525. PMID: 1283478.

55. Nizri E, Irony-Tur-Sinai M, Lory O, Orr-Urtreger A, Lavi E, Brenner T. Activation of the cholinergic anti-inflammatory system by nicotine attenuates neuroinflammation via suppression of Th1 and Th17 responses. J Immunol. 2009; 183:6681–6688. PMID: 19846875.

57. Schmeltz LR, Blevins TC, Aronoff SL, Ozer K, Leffert JD, Goldberg MA, Horowitz BS, Bertenshaw RH, Troya P, Cohen AE, Lanier RK, Wright C 4th. Anatabine supplementation decreases thyroglobulin antibodies in patients with chronic lymphocytic autoimmune (Hashimoto's) thyroiditis: a randomized controlled clinical trial. J Clin Endocrinol Metab. 2014; 99:E137–E142. PMID: 24178792.

58. Silva JE. The thermogenic effect of thyroid hormone and its clinical implications. Ann Intern Med. 2003; 139:205–213. PMID: 12899588.

59. Bechtold DA, Loudon AS. Hypothalamic thyroid hormones: mediators of seasonal physiology. Endocrinology. 2007; 148:3605–3607. PMID: 17639034.

60. Delange F, de Benoist B, Pretell E, Dunn JT. Iodine deficiency in the world: where do we stand at the turn of the century? Thyroid. 2001; 11:437–447. PMID: 11396702.

Fig. 1
Prevalence of subclinical hypothyroidism according to age in the (A) Ansung cohort and (B) Korean Longitudinal Study on Health and Aging study (KLoSHA). Adapted from Choi et al. J Korean Thyroid Assoc 2010;3:32-40 [36].
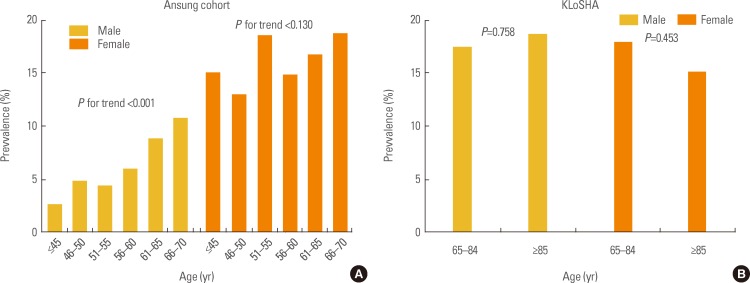
Table 1
Prevalence and Risk Factors of Subclinical Hypothyroidism
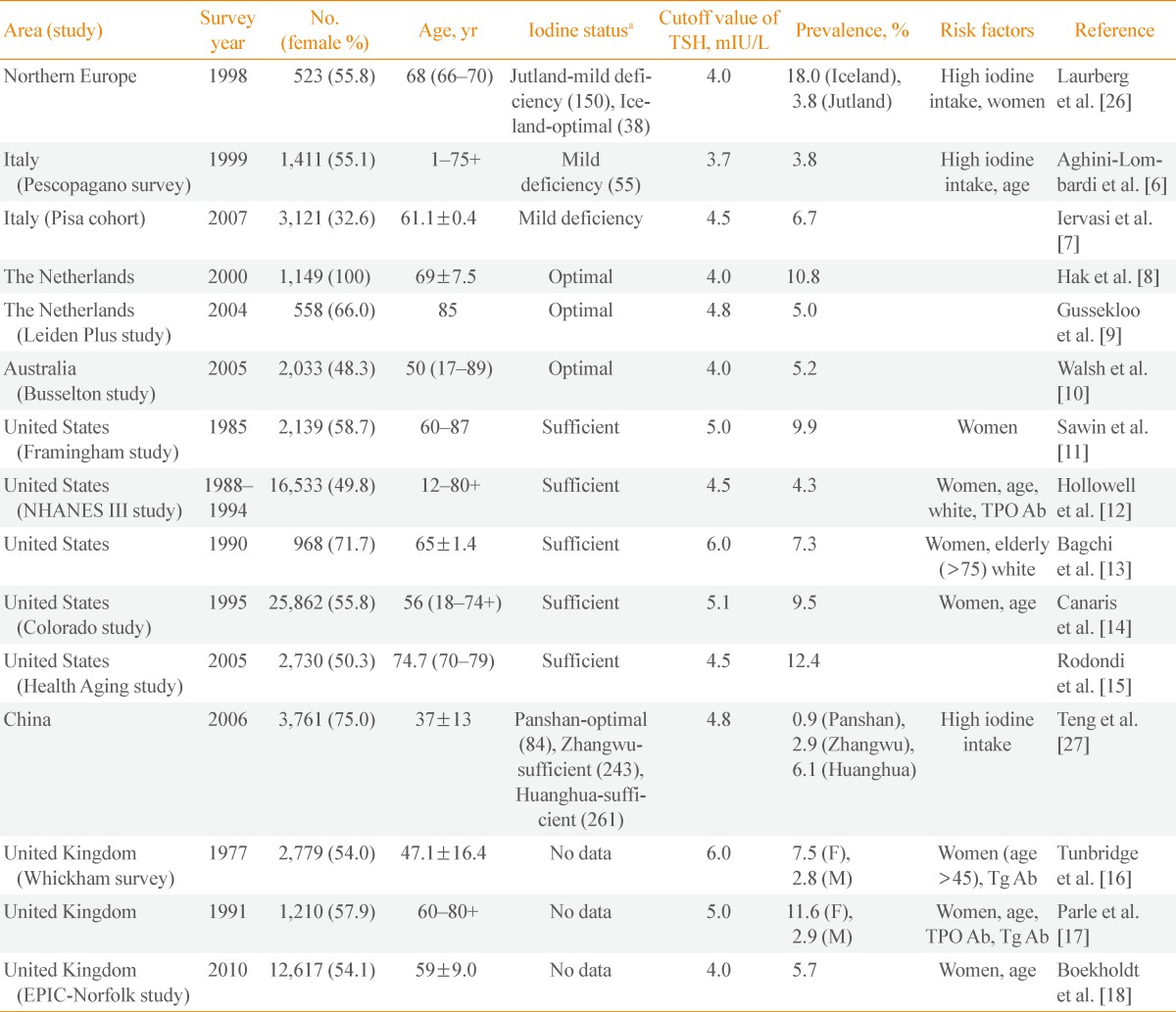
TSH, thyrotropin; NHANES, National Health and Nutrition Examination Survey; TPO Ab, thyroperoxidase antibody; F, female; M, male; Tg Ab, thyroglobulin antibody.
aUrinary excretion (µg/L). Data from de Benoist et al. Food Nutr Bull 2008;29:195-202 [38]. The numbers in the parenthesis were reported in the reference articles.
Table 3
Prevalence and Risk Factors of Subclinical Hyperthyroidism
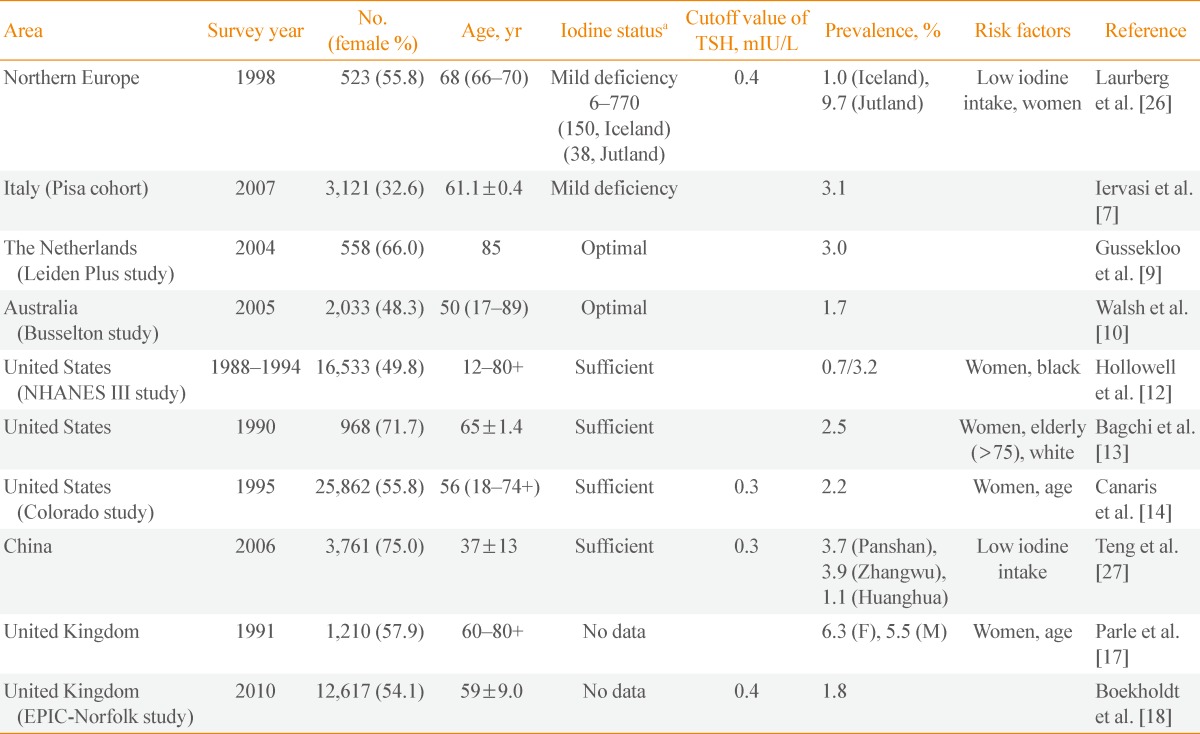
TSH, thyrotropin; NHANES, National Health and Nutrition Examination Survey.
aData from de Benoist et al. Food Nutr Bull 2008;29:195-202 [38].




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download