Abstract
Background
This study was conducted to examine the effects of ad libitum consumption of highly palatable food (HPF) during adolescence on the adverse behavioral outcome of neonatal maternal separation.
Methods
Male Sprague-Dawley pups were separated from dam for 3 hours daily during the first 2 weeks of birth (maternal separation, MS) or left undisturbed (nonhandled, NH). Half of MS pups received free access to chocolate cookies in addition to ad libitum chow from postnatal day 28 (MS+HPF). Pups were subjected to behavioral tests during young adulthood. The plasma corticosterone response to stress challenge was analyzed by radioimmunoassay.
Results
Daily caloric intake and body weight gain did not differ among the experimental groups. Ambulatory activities were decreased defecation activity and rostral grooming were increased in MS controls (fed with chow only) compared with NH rats. MS controls spent less time in open arms, and more time in closed arms during the elevated plus maze test, than NH rats. Immobility duration during the forced swim test was increased in MS controls compared with NH rats. Cookie access normalized the behavioral scores of ambulatory and defecation activities and grooming, but not the scores during the elevated plus maze and swim tests in MS rats. Stress-induced corticosterone increase was blunted in MS rats fed with chow only, and cookie access normalized it.
Many studies have indicated a strong correlation between traumatic events during early life and the development of behavioral and neuroendocrine abnormalities later in life. Neonatal maternal separation is considered an animal model of stressful experience early in life. A number of studies have demonstrated its impact both on the development of depression [1,2] and anxiety-like behavior [3,4] later in life and on the activity of the hypothalamic-pituitary-adrenal (HPA) axis; i.e., permanent alterations in the characteristics of the HPA response to stress [5,6,7]. We have reported that repeated maternal separation during the first 2 weeks of life results in the development of depression- and/or anxiety-like behaviors in later life of rats with dysfunctions in HPA axis activity [8].
A recent human study reported that subjects offered palatable foods, such as savory snacks, bakery snacks and pasta, had more pleasant emotions such as satisfaction, enjoyment, and desire [9]. Exposure to a highly preferred diet high in fat was suggested to reduce stress sensitivity [10]. Earlier studies reported that rats consuming a high fat diet for a period of 4 months show decreased sympathetic responses following psychological and immunological stress [11], and that consumption of palatable food improves the stress response in rats subjected to restraint relative to rats fed chow only [12]. Another study showed that rats fed a high fat diet for 5 days are less anxious on the elevated plus maze compared to rats fed a low fat diet [13]. Studies have suggested that the HPA axis dysfunction is implicated in the pathophysiology of anxiety [14] and depression [15]. Taken together, these reports support the relationship between consumption of a highly palatable diet and positive emotional behavior, likely by a modulation effect of palatable food on the HPA axis function.
Here, we have hypothesized that psychoemotional disorders such as anxiety and depression induced by experience of neonatal maternal separation, which is accompanied by HPA axis dysfunction [16,17], is likely to be improved by consumption of highly palatable food (HPF) in later life. In this study, rat pups experienced maternal separation (MS) during the first 2 weeks after birth, and then had free access to Oreo cookies, a HPF rich in fat, in addition to standard chow during adolescence and youth. Anxiety- and depression-like behaviors and their HPA axis function responding to stress were examined at youth, and compared with nonhandled (NH) and MS control groups that were fed standard chow only.
Sprague-Dawley rats were purchased (Samtako Bio, Osan, Korea), and cared in a specific-pathogen-free barrier area with constant control of temperature (22℃±1℃), humidity (55%), and a 12/12 hours light/dark cycle (lights-on at 7:00 AM). Standard laboratory food (Purina Rodent Chow, Purina Co., Seoul, Korea) and membrane filtered purified water were available ad libitum. Animals were cared according to the Guideline for Animal Experiments, 2000, edited by the Korean Academy of Medical Sciences, which is consistent with the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals, revised in 1996. All animal experiments were approved by the Committee for the Care and Use of Laboratory Animals at Seoul National University.
Nulliparous females and proven breeder males were used for breeding in the laboratory of the animal facility, and the pups were reared in a controlled manner to minimize and standardize unwanted environmental stimulation from in utero life. Twelve hours after confirming delivery (postnatal day [PND] 1), pups were culled to five males and five females per litter. Each litter was assigned either for the MS group or for the NH group. MS was performed as we previously described [16,17,18,19]. In brief, MS pups were removed from their dam and home cage and placed closely together in a new cage bedded with woodchips (Aspen shaving, Animal JS Bedding, Cheongyang, Korea) for 180 minutes, and then returned to their home cage and dam. MS was performed at room temperature; i.e., no additional treatment to keep the pups warm during the separation period other than placing them closely together was offered and pup-cooling during MS was expected. MS was performed from 9:00 AM to 12:00 PM daily from PND 1 through 14, and then the pups were left with their dam undisturbed until weaning on PND 22. The NH group remained undisturbed until weaning except for routine cage cleaning. For cage cleaning, all rats were moved to clean the cage twice a week. On the weaning day, two NH and four MS male pups were randomly selected from each NH or MS litter and placed two NH or two MS pups together in each cage. Two MS pups housed together from each litter received free access to chocolate cookies, a HPF, in addition to ad libitum chow from PND 28 (maternal separation fed with chow and cookies [MS+HPF] group), and the remaining two MS pups in each litter (MS group) and NH pups (NH group) received standard chow only. Water was freely available for all of the experimental groups, and the food conditions were continued throughout the whole experimental period.
NH, MS, and MS+HPF rats were subjected to the ambulatory test on PND 54. On each trial, the rat was placed in the center of the activity chamber (43.2 cm in length, 42.2 cm in width, and 30.5 cm in height, MED Associates, St. Albans, VT, USA), a transparent acryl chamber equipped with two horizontal planes of 16 infrared photocell-detector pairs placed in the x, y dimension, spaced 2.5 cm apart, and its ambulatory activity was monitored by the computerized system for 30 minutes. The light condition of the test room was maintained at the same intensity as the animal rooms under day light condition. Ambulatory activity was measured as the total counts of beam interruptions in the horizontal sensor during each consecutive 5 minutes session. Defecation activity of each rat during the ambulation test was measured by the weight of the fecal boli. Grooming activity was further analyzed; i.e., forepaw and head grooming was considered as rostral grooming, and body, legs, and tail/genital grooming as caudal grooming [20]. The activity chamber was cleaned with 70% ethanol after each use to eliminate any olfactory cues of the previously tested rat.
Two days after the ambulatory activity test (PND 56), rats were subjected to the behavioral assessment in an elevated plus maze, a plus shaped acryl maze with two opposite open arms (50 cm in length and 10 cm in width) and two opposite closed arms (50 cm in length, 10 cm in width, and 31 cm in height), extending out from a central platform (10×10 cm). The whole apparatus was elevated 50 cm above the floor. The test procedure was followed as previously described [4]. Each rat was placed in the center of the maze facing one of the open arms, and then allowed to explore the open or closed arms of the maze for 5 minutes. The time spent in the different arms was recorded, respectively. Four paws had to be inside the entrance line to each arm, which signaled the start of the time spent in the specific arm, and the end time was recorded when all four paws were outside the line again. The maze was cleaned with 70% ethanol after each test to prevent influences of the previously tested rat.
Three days after the elevated plus maze test (PND 59), rats were subjected to the forced swim test, according to the method previously described [21]. Each rat was allowed to swim in a glass cylinder (54 cm in height and 24 cm in diameter) filled with water in 40 cm of depth (23℃ to 25℃) for 5 minutes, and the test sessions were recorded by a video camera from the side of the cylinder. Duration of rat's immobility in the water was scored from videotapes by a trained observer (J.Y.K.) who was blinded to the experimental conditions. Immobility was defined as the state in which rats were judged to be making only the movements necessary to keep their head above the surface.
A week after the end of behavioral sessions, rats were placed in a restraint box for 2 hours, in which rats were able to move their four limbs but not to change their body orientation. Tail blood was collected at 0, 30, 60, and 120 minutes time points during the restraint period, and centrifuged at 2,000 rpm for 20 minutes. The plasma samples were frozen in liquid nitrogen, and stored at -80℃ until used for the assay. Plasma levels of corticosterone were determined by radioimmunoassay using 125I-labelled Coat-A-Count kit (Siemens, Mountain View, CA, USA). The sensitivity of the assay was 5.7 ng/mL. The intra-assay coefficient of variation was 4% to 12.2%.
Rats were placed in the test room at least 2 hours prior to each test to minimize unwanted stress effects, and all behavioral assessments were performed between 9:00 AM and 12:00 PM of the day to avoid the influences of circadian variances.
Data were analyzed by one-way analysis of variance, and preplanned comparisons between groups performed by post hoc Fisher's protected least significant difference test, using StatView software (Abacus, Berkeley, CA, USA). The level of significance was set at P<0.05, and all values were presented as means±SEM.
Daily chow intake and body weight gain did not differ between NH and MS rats (Fig. 1A, B). Chow intake of MS rats was decreased with cookie access, but daily caloric intake was not affected (Fig. 1A). Consequently, body weight gain of MS rats was not affected by cookie access (Fig. 1B).
Ambulatory activities of NH, MS, and MS+HPF rats were measured in a computerized activity chamber for 30 minutes on PND 54. Ambulatory counts, the total counts of beam interruptions in the horizontal sensor, were gradually decreased during the test session in all groups (Fig. 2A). Total ambulatory counts and the distance travelled during the 30 minutes of session were decreased in MS rats compared with NH rats (P<0.05), and increased with cookie access in MS rats (MS vs. MS+HPF, P<0.05) (Fig. 2B, C). Defecation activity and grooming behavior was scored during the ambulatory activity test (Fig. 2D, E). Feces weight and rostral grooming were increased whereas caudal grooming decreased by MS experience (NH vs. MS, P<0.05), and these behaviors were normalized with cookie access.
In order to further assess the anxiety-like behaviors, rats were subjected to an elevated plus maze test 2 days after the ambulatory activity test. MS rats spent more time in closed arms, and less time in open arms, compared with NH rats (P<0.05), and cookie access did not affect the behavioral scores of MS rats (Fig. 3A). To assess depression-like behaviors, rats were subjected to forced swim test 3 days after the elevated plus maze test. Immobility duration during the 5 minutes of test session was significantly increased (P<0.05) in MS rats (27.667±5.878 seconds) compared with NH rats (8.222±2.748 seconds), and the immobility score was not affected by cookie access in MS rats (Fig. 3B).
A week after the swim test, rats received 2 hours of restraint stress, and tail blood was collected at 0, 30, 60, and 120 minutes time points during the restraint session, and used for plasma corticosterone assay (Fig. 4). The plasma corticosterone levels of NH rats were significantly increased at 30 and 60 minutes after the onset of the restraint stress (P<0.05 vs. 0 time point), and restored to the basal levels at 120 minutes after the restraint onset. However, in MS rats, stress-induced corticosterone increase was not observed, and the basal levels of corticosterone (0 time point) were higher than in NH rats (138.695±28.892 ng/mL in NH and 236.143±36.524 ng/mL in MS, P<0.05). MS rats with cookie access (MS+HPF) showed a significant increase in plasma corticosterone levels at 30 minutes time point (P<0.05 vs. 0 time point), and their basal plasma corticosterone levels did not differ from NH rats.
Many studies have demonstrated that experience of neonatal MS may lead to the development of depression- and anxiety-like behaviors later in life [2,3,4,5]. In this study, not only the ambulatory activities but also the distance travelled during the activity test were suppressed significantly in MS rats fed with chow only compared with NH rats, suggesting increased anxiety and/or depression by experience of our MS protocol. Defecation scores during the activity test were increased in MS rats fed with chow only relative to NH rats in this study. Low locomotor activity and high defecation rates are often regarded as central emotional responses of rats exposed to novelty [22]. Grooming is often seen in animal models of stress and anxiety [23,24], leading to a long-standing view of grooming as an anxiogenic response [25,26]. It has been reported that highly stressed mice spend significantly more time grooming rostral than caudal areas [20,27]. In this study, MS rats fed with chow only spent more time in rostral grooming and less time in caudal grooming, compared to NH rats. That is, increased defecation scores and rostral grooming, and decreased caudal grooming in MS rats fed with chow only further supports the anxiogenic effect of MS in our protocol. It has been shown that alterations in the depression-like states in rodents by chronic stress affect grooming behaviors [28,29,30]. Thus, it is likely that altered grooming behaviors in our MS rats fed with chow only may also reveal increased depression-like states. Indeed, MS rats fed with chow only showed behavioral depression in the swim test with increased immobility duration, and increased anxiety in the elevated plus maze test, compared to NH rats. Together, it is concluded that the rats subjected to our MS protocol show anxiety- and depression-like behaviors in later life, in accordance with our previous reports [17,19].
It has been reported that MS experiences during neonatal period results in permanent alterations in the characteristics of the HPA response to stress [5,6,7]. In this study, MS rats fed with chow only showed blunted corticosterone response to restraint stress with increased basal levels of corticosterone compared with NH control rats. This result concurs with our previous report showing altered HPA axis activities in our MS rats responding to metabolic stresses [16,18], and further supports the HPA axis dysfunction by experience of our MS protocol. It is likely that increased basal corticosterone levels in MS rats fed with chow only may reveal increased HPA axis activity responding to daily stressors by MS experience and result in a blunted response to acute restraint stress. Studies have suggested that the HPA axis dysfunction is implicated in the pathophysiology of anxiety [14] and depression [15]. Elevated basal cortisol levels were associated with anxiety and depressive symptoms in young subjects [31,32,33]. Thus, it is plausible that the HPA axis dysfunction in our young MS rats; i.e., blunted stress-induced corticosterone increase with elevated basal levels, is implicated in their anxiety- and depression-like behaviors observed.
In this study, MS rats, that had free access to chocolate cookies in addition to the chow access (MS+HPF group), showed a significant increase in the plasma corticosterone levels responding to restraint stress, unlike the blunted response in MS rats fed with chow only (MS group). Furthermore, the basal corticosterone levels of MS+HPF rats did not differ from NH rats. These results suggest that free access to HPF during adolescence and youth might have improved the HPA axis dysfunction by MS experience. Puberty is a crucial developmental period characterized by increased endocrine plasticity and changes in stress responsiveness [34]. Studies comparing HPA axis activity in prepubertal and adult rats show that there is a significantly persistent hormonal stress response in prepubertal animals compared to adults [35,36,37]. Previous study suggested that a postweaning highly preferred diet can modify the basal HPA axis activity and the endocrine responses to an acute stress by affecting both stress and metabolic mediators [38,39]. Exposure to a highly preferred diet high in fat was suggested to reduce stress sensitivity [10]. Earlier studies reported that sympathetic responses to psychological stress are decreased in rats consuming a high fat diet for a long-time period [11], and that consumption of palatable food improves the stress response in rats subjected to restraint [12]. Also, exaggerated corticosterone release following acute stress was observed in a study using a 7-day moderate high fat diet (20% fat content) protocol to dissociate the impact of obesity from that of fat [40]. The chocolate cookies used in this study is a moderate high fat diet (~21% fat content), and caloric intake and body weight gain of MS rats was not affected by cookie access. Altogether, it is concluded that free access to a highly preferred diet during adolescence and youth may reduce the stress sensitivity of MS rats to daily routine stressors and normalize the basal corticosterone levels which were increased by MS experience. Consequently, MS rats fed with HPF were likely able to show a normal stress response; i.e., a significant increase of corticosterone levels in response to an acute restraint stress.
In this study, ambulatory activities and the distance travelled during the activity test were significantly increased, and defecation activity decreased, in the MS+HPF group compared to the MS group. Also, grooming behaviors of MS+HPF rats did not differ from NH rats, while rostral grooming was increased and caudal grooming decreased in MS rats compared with NH rats. Previous studies showed that prolonged fat intake decreased anxiety-like behaviors with reduced sympathetic response to stressors [11] and short-term exposure to a high fat diet also reduced anxiety on the elevated plus maze in rats [13]. Together, it is suggested that prolonged access to chocolate cookies, a moderate high fat diet, during adolescence and youth might have improved anxiety-related behaviors of MS rats in this study. As mentioned above, HPA axis dysfunction has been implicated in the pathophysiology of anxiety and depression [14,15], and effective treatments of affective disorders with antidepressants or anxiolytic agents also modulate the HPA axis function [41,42]. In this study, although some anxiety-related behaviors of MS rats and their HPA axis functions were improved together by prolonged cookie access, anxiety on the elevated plus maze and depressive behavior during the swim test were not improved. Thus, it is concluded that prolonged cookie access during adolescence and youth partly improves anxiety-related, but not depressive, symptoms in rats that experienced our MS protocol, possibly in relation with improved HPA axis function.
The nucleus accumbens (NAc), a basal forebrain structure constituting a mesolimbic dopaminergic pathway, has a role in reward, motivation, and reinforcement [43]. It has been reported that reduced dopaminergic function within the NAc may cause depression-like behaviors in rodents [44], and the striatal dopaminergic activity was suggested to be associated with the severity of anhedonia in depressed patients [45]. The NAc is activated in response to behavioral stress paradigm [46,47], suggesting its implication in the stress-responsive HPA axis regulation. Rats that experienced our MS protocol showed not only HPA axis dysfunction but also reduced dopaminergic activity in the NAc [48]. Consumption of palatable foods elevates extracellular dopamine concentrations in the NAc of the ventral striatum [49], and the striatal dopamine release reflects the perceived pleasantness of a meal in humans [50]. However, we have found that limited cookie access during adolescent period did not affect the NAc dopamine levels in MS rats, but normalized the amygdala dopamine levels which was increased by MS experience (our unpublished observation). Mesolimbic dopamine neurons originating in the ventral tegmental area (VTA) innervate limbic sites including the NAc and amygdala. The VTA, Nac, and amygdala are posited to form an interconnected network important for reward processing [51], while the amygdala also has a fundamental role in the control of anxiety [52,53]. The NAc has been viewed as a transition area between the ventral striatum and the extended amygdala including the central nucleus of the amygdala (CeA) [54]. Studies have suggested that enhanced dopamine signaling within the CeA is associated with reward-seeking [55,56] and anxiety-like behaviors [57,58]. The CeA has been shown to respond to hormonal changes associated with stress and fear, the core symptoms of anxiety disorders [59], and interact with the HPA axis [60,61]. Taken all together, it is likely that altered dopaminergic activity in the NAc and CeA with the HPA axis dysfunction may be implicated in MS-induced depression- and anxiety-like behaviors, and speculated that the partial improvement of anxiety-related behaviors in MS rats by prolonged palatable food consumption may be due to a region specific effect of it on brain dopamine neurotransmission. Further studies are warranted to define its underlying mechanism.
Figures and Tables
 | Fig. 1(A) Daily chow and caloric intake and (B) body weight gain. Values are expressed as means±SEM. NH, nonhandled fed with chow only; MS, maternal separation fed with chow only; MS+HPF, maternal separation fed with chow and cookies; HPF, highly palatable food. aP<0.05; bP<0.01; cP<0.001 vs. MS, n=10 to 12 in each group. |
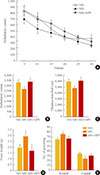 | Fig. 2Ambulatory activity test. (A) Ambulatory counts scored consecutively at every 5 minutes session, (B) total ambulatory counts, and (C) travelled distance during 30 minutes of the ambulatory activity test. (D) Defecation activity and (E) stereotyped behavior, number of groomings, were scored during 30 minutes of the activity test. Values are expressed as means±SEM. NH, nonhandled fed with chow only; MS, maternal separation fed with chow only; MS+HPF, maternal separation fed with chow and cookies; HPF, highly palatable food. aP<0.05 vs. NH; bP<0.05 vs. MS, n=10 to 12 in each group. |
 | Fig. 3(A) Time spent in each arm during the elevated plus maze test, and (B) scores during the forced swim test. Values are expressed as means±SEM. NH, nonhandled fed with chow only; MS, maternal separation fed with chow only; MS+HPF, maternal separation fed with chow and cookies; HPF, highly palatable food. aP<0.05; bP<0.01 vs. NH, n=8 in each group. |
 | Fig. 4Plasma corticosterone levels during 2 hours of restraint session. Rats were subjected to restraint stress following a week of recovery from the forced swim test. Feeding conditions continued during the recovery period. Rats were placed in the restraint box and tail blood was collected at each time point. Values are expressed as means±SEM. NH, nonhandled fed with chow only; MS, maternal separation fed with chow only; MS+HPF, maternal separation fed with chow and cookies; HPF, highly palatable food. aP<0.05 vs. 0 time point of NH; bP<0.05 vs. 0 time point of NH; cP<0.05 vs. 0 time point of MS+HPF, n=8 in each group. |
ACKNOWLEDGMENTS
This study was supported by grants from the National Research Foundation (2013R1A1A3A04) and the Oromaxillofacial Dysfunction Research Center for the Elderly at Seoul National University in Korea (2013070465) funded by the Ministry of Science, ICT, and Future Planning.
References
1. Ladd CO, Huot RL, Thrivikraman KV, Nemeroff CB, Meaney MJ, Plotsky PM. Long-term behavioral and neuroendocrine adaptations to adverse early experience. Prog Brain Res. 2000; 122:81–103.
2. El Khoury A, Gruber SH, Mork A, Mathe AA. Adult life behavioral consequences of early maternal separation are alleviated by escitalopram treatment in a rat model of depression. Prog Neuropsychopharmacol Biol Psychiatry. 2006; 30:535–540.
3. Kalinichev M, Easterling KW, Plotsky PM, Holtzman SG. Long-lasting changes in stress-induced corticosterone response and anxiety-like behaviors as a consequence of neonatal maternal separation in Long-Evans rats. Pharmacol Biochem Behav. 2002; 73:131–140.
4. Daniels WM, Pietersen CY, Carstens ME, Stein DJ. Maternal separation in rats leads to anxiety-like behavior and a blunted ACTH response and altered neurotransmitter levels in response to a subsequent stressor. Metab Brain Dis. 2004; 19:3–14.
5. Ladd CO, Owens MJ, Nemeroff CB. Persistent changes in corticotropin-releasing factor neuronal systems induced by maternal deprivation. Endocrinology. 1996; 137:1212–1218.
6. van Oers HJ, de Kloet ER, Levine S. Early vs. late maternal deprivation differentially alters the endocrine and hypothalamic responses to stress. Brain Res Dev Brain Res. 1998; 111:245–252.
7. Vazquez DM, Lopez JF, Van Hoers H, Watson SJ, Levine S. Maternal deprivation regulates serotonin 1A and 2A receptors in the infant rat. Brain Res. 2000; 855:76–82.
8. Jahng JW. An animal model of eating disorders associated with stressful experience in early life. Horm Behav. 2011; 59:213–220.
9. Desmet PM, Schifferstein HN. Sources of positive and negative emotions in food experience. Appetite. 2008; 50:290–301.
10. Teegarden SL, Bale TL. Decreases in dietary preference produce increased emotionality and risk for dietary relapse. Biol Psychiatry. 2007; 61:1021–1029.
11. Buwalda B, Blom WA, Koolhaas JM, van Dijk G. Behavioral and physiological responses to stress are affected by high-fat feeding in male rats. Physiol Behav. 2001; 73:371–377.
12. Pecoraro N, Reyes F, Gomez F, Bhargava A, Dallman MF. Chronic stress promotes palatable feeding, which reduces signs of stress: feedforward and feedback effects of chronic stress. Endocrinology. 2004; 145:3754–3762.
13. Prasad A, Prasad C. Short-term consumption of a diet rich in fat decreases anxiety response in adult male rats. Physiol Behav. 1996; 60:1039–1042.
14. Albanidou-Farmaki E, Poulopoulos AK, Epivatianos A, Farmakis K, Karamouzis M, Antoniades D. Increased anxiety level and high salivary and serum cortisol concentrations in patients with recurrent aphthous stomatitis. Tohoku J Exp Med. 2008; 214:291–296.
15. Heim C, Newport DJ, Heit S, Graham YP, Wilcox M, Bonsall R, Miller AH, Nemeroff CB. Pituitary-adrenal and autonomic responses to stress in women after sexual and physical abuse in childhood. JAMA. 2000; 284:592–597.
16. Kim HJ, Lee JH, Choi SH, Lee YS, Jahng JW. Fasting-induced increases of arcuate NPY mRNA and plasma corticosterone are blunted in the rat experienced neonatal maternal separation. Neuropeptides. 2005; 39:587–594.
17. Lee JH, Kim HJ, Kim JG, Ryu V, Kim BT, Kang DW, Jahng JW. Depressive behaviors and decreased expression of serotonin reuptake transporter in rats that experienced neonatal maternal separation. Neurosci Res. 2007; 58:32–39.
18. Ryu V, Lee JH, Yoo SB, Gu XF, Moon YW, Jahng JW. Sustained hyperphagia in adolescent rats that experienced neonatal maternal separation. Int J Obes (Lond). 2008; 32:1355–1362.
19. Ryu V, Yoo SB, Kang DW, Lee JH, Jahng JW. Post-weaning isolation promotes food intake and body weight gain in rats that experienced neonatal maternal separation. Brain Res. 2009; 1295:127–134.
20. Kalueff AV, Aldridge JW, LaPorte JL, Murphy DL, Tuohimaa P. Analyzing grooming microstructure in neurobehavioral experiments. Nat Protoc. 2007; 2:2538–2544.
21. Porsolt RD, Le Pichon M, Jalfre M. Depression: a new animal model sensitive to antidepressant treatments. Nature. 1977; 266:730–732.
22. Ramos A, Mormede P. Stress and emotionality: a multidimensional and genetic approach. Neurosci Biobehav Rev. 1998; 22:33–57.
23. Barros HM, Tannhauser SL, Tannhauser MA, Tannhauser M. The effects of GABAergic drugs on grooming behaviour in the open field. Pharmacol Toxicol. 1994; 74:339–344.
24. Kalueff AV, Tuohimaa P. Contrasting grooming phenotypes in three mouse strains markedly different in anxiety and activity (129S1, BALB/c and NMRI). Behav Brain Res. 2005; 160:1–10.
25. Ferre P, Fernandez-Teruel A, Escorihuela RM, Driscoll P, Corda MG, Giorgi O, Tobena A. Behavior of the Roman/Verh high- and low-avoidance rat lines in anxiety tests: relationship with defecation and self-grooming. Physiol Behav. 1995; 58:1209–1213.
26. Nosek K, Dennis K, Andrus BM, Ahmadiyeh N, Baum AE, Solberg Woods LC, Redei EE. Context and strain-dependent behavioral response to stress. Behav Brain Funct. 2008; 4:23.
27. Kalueff AV, Tuohimaa P. Grooming analysis algorithm for neurobehavioural stress research. Brain Res Brain Res Protoc. 2004; 13:151–158.
28. Kompagne H, Bardos G, Szenasi G, Gacsalyi I, Harsing LG, Levay G. Chronic mild stress generates clear depressive but ambiguous anxiety-like behaviour in rats. Behav Brain Res. 2008; 193:311–314.
29. Piato AL, Detanico BC, Jesus JF, Lhullier FL, Nunes DS, Elisabetsky E. Effects of Marapuama in the chronic mild stress model: further indication of antidepressant properties. J Ethnopharmacol. 2008; 118:300–304.
30. Yalcin I, Aksu F, Bodard S, Chalon S, Belzung C. Antidepressant-like effect of tramadol in the unpredictable chronic mild stress procedure: possible involvement of the noradrenergic system. Behav Pharmacol. 2007; 18:623–631.
31. Stetler C, Miller GE. Depression and hypothalamic-pituitary-adrenal activation: a quantitative summary of four decades of research. Psychosom Med. 2011; 73:114–126.
32. Garcia de Miguel B, Nutt DJ, Hood SD, Davies SJ. Elucidation of neurobiology of anxiety disorders in children through pharmacological challenge tests and cortisol measurements: a systematic review. J Psychopharmacol. 2012; 26:431–442.
33. Dietrich A, Ormel J, Buitelaar JK, Verhulst FC, Hoekstra PJ, Hartman CA. Cortisol in the morning and dimensions of anxiety, depression, and aggression in children from a general population and clinic-referred cohort: an integrated analysis. The TRAILS study. Psychoneuroendocrinology. 2013; 38:1281–1298.
34. Romeo RD, McEwen BS. Stress and the adolescent brain. Ann N Y Acad Sci. 2006; 1094:202–214.
35. Romeo RD, Waters EM, McEwen BS. Steroid-induced hippocampal synaptic plasticity: sex differences and similarities. Neuron Glia Biol. 2004; 1:219–229.
36. Goldman L, Winget C, Hollingshead GW, Levine S. Postweaning development of negative feedback in the pituitary-adrenal system of the rat. Neuroendocrinology. 1973; 12:199–211.
37. Vazquez DM, Morano MI, Lopez JF, Watson SJ, Akil H. Short-term adrenalectomy increases glucocorticoid and mineralocorticoid receptor mRNA in selective areas of the developing hippocampus. Mol Cell Neurosci. 1993; 4:455–471.
38. Boukouvalas G, Antoniou K, Papalexi E, Kitraki E. Post weaning high fat feeding affects rats' behavior and hypothalamic pituitary adrenal axis at the onset of puberty in a sexually dimorphic manner. Neuroscience. 2008; 153:373–382.
39. Boukouvalas G, Gerozissis K, Markaki E, Kitraki E. High-fat feeding influences the endocrine responses of pubertal rats to an acute stress. Neuroendocrinology. 2010; 92:235–245.
40. Soulis G, Papalexi E, Kittas C, Kitraki E. Early impact of a fat-enriched diet on behavioral responses of male and female rats. Behav Neurosci. 2007; 121:483–490.
41. Saiyudthong S, Marsden CA. Acute effects of bergamot oil on anxiety-related behaviour and corticosterone level in rats. Phytother Res. 2011; 25:858–862.
42. Scharf SH, Sterlemann V, Liebl C, Muller MB, Schmidt MV. Chronic social stress during adolescence: interplay of paroxetine treatment and ageing. Neuropharmacology. 2013; 72:38–46.
43. Salamone JD, Correa M. Motivational views of reinforcement: implications for understanding the behavioral functions of nucleus accumbens dopamine. Behav Brain Res. 2002; 137:3–25.
44. Di Chiara G, Loddo P, Tanda G. Reciprocal changes in prefrontal and limbic dopamine responsiveness to aversive and rewarding stimuli after chronic mild stress: implications for the psychobiology of depression. Biol Psychiatry. 1999; 46:1624–1633.
45. Gorwood P. Neurobiological mechanisms of anhedonia. Dialogues Clin Neurosci. 2008; 10:291–299.
46. Imperato A, Angelucci L, Casolini P, Zocchi A, Puglisi-Allegra S. Repeated stressful experiences differently affect limbic dopamine release during and following stress. Brain Res. 1992; 577:194–199.
47. Saal D, Dong Y, Bonci A, Malenka RC. Drugs of abuse and stress trigger a common synaptic adaptation in dopamine neurons. Neuron. 2003; 37:577–582.
48. Jahng JW, Ryu V, Yoo SB, Noh SJ, Kim JY, Lee JH. Mesolimbic dopaminergic activity responding to acute stress is blunted in adolescent rats that experienced neonatal maternal separation. Neuroscience. 2010; 171:144–152.
49. Hernandez L, Hoebel BG. Food reward and cocaine increase extracellular dopamine in the nucleus accumbens as measured by microdialysis. Life Sci. 1988; 42:1705–1712.
50. Small DM, Jones-Gotman M, Dagher A. Feeding-induced dopamine release in dorsal striatum correlates with meal pleasantness ratings in healthy human volunteers. Neuroimage. 2003; 19:1709–1715.
51. Carlezon WA Jr, Thomas MJ. Biological substrates of reward and aversion: a nucleus accumbens activity hypothesis. Neuropharmacology. 2009; 56:Suppl 1. 122–132.
52. Etkin A, Prater KE, Schatzberg AF, Menon V, Greicius MD. Disrupted amygdalar subregion functional connectivity and evidence of a compensatory network in generalized anxiety disorder. Arch Gen Psychiatry. 2009; 66:1361–1372.
53. Tye KM, Prakash R, Kim SY, Fenno LE, Grosenick L, Zarabi H, Thompson KR, Gradinaru V, Ramakrishnan C, Deisseroth K. Amygdala circuitry mediating reversible and bidirectional control of anxiety. Nature. 2011; 471:358–362.
54. Cardinal RN, Parkinson JA, Hall J, Everitt BJ. Emotion and motivation: the role of the amygdala, ventral striatum, and prefrontal cortex. Neurosci Biobehav Rev. 2002; 26:321–352.
55. Rezayof A, Zarrindast MR, Sahraei H, Haeri-Rohani AH. Involvement of dopamine D2 receptors of the central amygdala on the acquisition and expression of morphine-induced place preference in rat. Pharmacol Biochem Behav. 2002; 74:187–197.
56. Thiel KJ, Wenzel JM, Pentkowski NS, Hobbs RJ, Alleweireldt AT, Neisewander JL. Stimulation of dopamine D2/D3 but not D1 receptors in the central amygdala decreases cocaine-seeking behavior. Behav Brain Res. 2010; 214:386–394.
57. Guarraci FA, Frohardt RJ, Young SL, Kapp BS. A functional role for dopamine transmission in the amygdala during conditioned fear. Ann N Y Acad Sci. 1999; 877:732–736.
58. Silberman Y, Winder DG. Corticotropin releasing factor and catecholamines enhance glutamatergic neurotransmission in the lateral subdivision of the central amygdala. Neuropharmacology. 2013; 70:316–323.
59. Hui GK, Figueroa IR, Poytress BS, Roozendaal B, McGaugh JL, Weinberger NM. Memory enhancement of classical fear conditioning by post-training injections of corticosterone in rats. Neurobiol Learn Mem. 2004; 81:67–74.
60. Shepard JD, Barron KW, Myers DA. Corticosterone delivery to the amygdala increases corticotropin-releasing factor mRNA in the central amygdaloid nucleus and anxiety-like behavior. Brain Res. 2000; 861:288–295.
61. Shepard JD, Barron KW, Myers DA. Stereotaxic localization of corticosterone to the amygdala enhances hypothalamo-pituitary-adrenal responses to behavioral stress. Brain Res. 2003; 963:203–213.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download