Abstract
Background
Bile acids were important for the regulation of cholesterol homeostasis. Thyroid hormone increased the expression of CYP7A1 (cholesterol 7α-hydroxylase), catalyzing the first step in the biosynthesis of bile acids. However, the effect of thyroid hormone on bile acid export has not been previously assessed. The principal objective of this study is to evaluate the effects of thyroid hormone on the bile salt export pump (BSEP).
Methods
Thyroid hormone, T3 (1 mg/g) was administered to male mice via intraperitoneal injection. After 6 hours and 5 days of T3 treatment, we measured serum total and LDL cholesterol and hepatobiliary bile acid concentrations. We assessed the changes associated with bile acid synthesis and transport. In order to evaluate the direct effect of thyroid hormone, we assessed the changes in the levels of BSEP protein after T3 administration in human hepatoma cells.
Results
Serum total and LDL cholesterol were reduced and hepatobiliary bile acid concentrations were increased following T3 treatment. Expressions of Cyp7a1 and BSEP mRNA were increased following T3 treatment. The levels of the BSEP protein in the mouse liver as well as in the human hepatoma cells were increased after T3 treatment.
Figures and Tables
Fig. 1
Genes related to bile acid synthesis and transport in hepatocyte. BSEP, bile salt export pump; CYP7A1, cholesterol 7α hydroxylase; CYP27A1, sterol 27 hydroxylase; CYP8B1, sterol 12α hydroxylase; MRP, multidrug-resistance associated protein; NTCP, Na-dependent taurocholate cotransport peptide; OATP, organic anion transporting polypeptide.
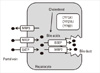
Fig. 2
Effect of thyroid hormone to serum cholesterol (A), LDL cholesterol (B) and hepatobiliary bile acid (C) level. LDL, low density lipoprotein; T3, 3,5,3'-triiodo-L-thyronine (thyroid hormone).

Fig. 3
Effect of thyroid hormone to Cyp7a1, BSEP and MRP2 expression. Cyp7a1 and BSEP mRNA expression of mouse liver were increased after T3 treatment in northern blot analysis (A), MRP2 mRNA expression of mouse liver after 5 days of T3 treatment did not change in RT-PCR analysis (B), protein level of BSEP in mouse liver was also increased after 5 days of T3 treatment in western blot analysis (C). Cyp7a1, cholesterol 7α hydroxylase; BSEP, bile salt export pump; MRP2, -resistance associated protein 2; RT-PCR, reverse transcriptase polymerase chain reaction; T3, 3,5,3'-triiodo-L-thyronine (thyroid hormone).

References
1. Chiang JY. Regulation of bile acid synthesis: pathways, nuclear receptors, and mechanisms. J Hepatol. 2004. 40:539–551.
2. Chiang JY. Bile acids: regulation of synthesis. J Lipid Res. 2009. 50:1955–1966.
3. Russell DW, Setchell KD. Bile acid biosynthesis. Biochemistry. 1992. 31:4737–4749.
4. Jelinek DF, Andersson S, Slaughter CA, Russell DW. Cloning and regulation of cholesterol 7 alpha-hydroxylase, the rate-limiting enzyme in bile acid biosynthesis. J Biol Chem. 1990. 265:8190–8197.
5. Goodwin B, Jones SA, Price RR, Watson MA, McKee DD, Moore LB, Galardi C, Wilson JG, Lewis MC, Roth ME, Maloney PR, Willson TM, Kliewer SA. A regulatory cascade of the nuclear receptors FXR, SHP-1, and LRH-1 represses bile acid biosynthesis. Mol Cell. 2000. 6:517–526.
6. Lu TT, Makishima M, Repa JJ, Schoonjans K, Kerr TA, Auwerx J, Mangelsdorf DJ. Molecular basis for feedback regulation of bile acid synthesis by nuclear receptors. Mol Cell. 2000. 6:507–515.
7. Trauner M, Boyer JL. Bile salt transporters: molecular characterization, function, and regulation. Physiol Rev. 2003. 83:633–671.
8. Stieger B, Meier Y, Meier PJ. The bile salt export pump. Pflugers Arch. 2007. 453:611–620.
9. Akita H, Suzuki H, Ito K, Kinoshita S, Sato N, Takikawa H, Sugiyama Y. Characterization of bile acid transport mediated by multidrug resistance associated protein 2 and bile salt export pump. Biochim Biophys Acta. 2001. 1511:7–16.
10. Lam P, Wang R, Ling V. Bile acid transport in sister of P-glycoprotein (ABCB11) knockout mice. Biochemistry. 2005. 44:12598–12605.
11. Hashimoto K, Cohen RN, Yamada M, Markan KR, Monden T, Satoh T, Mori M, Wondisford FE. Cross-talk between thyroid hormone receptor and liver X receptor regulatory pathways is revealed in a thyroid hormone resistance mouse model. J Biol Chem. 2006. 281:295–302.
12. Ness GC, Pendleton LC, Li YC, Chiang JY. Effect of thyroid hormone on hepatic cholesterol 7 alpha hydroxylase, LDL receptor, HMG-CoA reductase, farnesyl pyrophosphate synthetase and apolipoprotein A-I mRNA levels in hypophysectomized rats. Biochem Biophys Res Commun. 1990. 172:1150–1156.
13. Ness GC, Pendelton LC, Zhao Z. Thyroid hormone rapidly increases cholesterol 7 alpha-hydroxylase mRNA levels in hypophysectomized rats. Biochim Biophys Acta. 1994. 1214:229–233.
14. Petty KJ, Desvergne B, Mitsuhashi T, Nikodem VM. Identification of a thyroid hormone response element in the malic enzyme gene. J Biol Chem. 1990. 265:7395–7400.
15. Shin DJ, Plateroti M, Samarut J, Osborne TF. Two uniquely arranged thyroid hormone response elements in the far upstream 5' flanking region confer direct thyroid hormone regulation to the murine cholesterol 7alpha hydroxylase gene. Nucleic Acids Res. 2006. 34:3853–3861.
16. Kullak-Ublick GA, Stieger B, Meier PJ. Enterohepatic bile salt transporters in normal physiology and liver disease. Gastroenterology. 2004. 126:322–342.
17. Suchy FJ, Ananthanarayanan M. Bile salt excretory pump: biology and pathobiology. J Pediatr Gastroenterol Nutr. 2006. 43:Suppl 1. S10–S16.
18. Ananthanarayanan M, Balasubramanian N, Makishima M, Mangelsdorf DJ, Suchy FJ. Human bile salt export pump promoter is transactivated by the farnesoid X receptor/bile acid receptor. J Biol Chem. 2001. 276:28857–28865.
19. Plass JR, Mol O, Heegsma J, Geuken M, Faber KN, Jansen PL, Muller M. Farnesoid X receptor and bile salts are involved in transcriptional regulation of the gene encoding the human bile salt export pump. Hepatology. 2002. 35:589–596.
20. Sinal CJ, Tohkin M, Miyata M, Ward JM, Lambert G, Gonzalez FJ. Targeted disruption of the nuclear receptor FXR/BAR impairs bile acid and lipid homeostasis. Cell. 2000. 102:731–744.
21. Song X, Kaimal R, Yan B, Deng R. Liver receptor homolog 1 transcriptionally regulates human bile salt export pump expression. J Lipid Res. 2008. 49:973–984.
22. Mataki C, Magnier BC, Houten SM, Annicotte JS, Argmann C, Thomas C, Overmars H, Kulik W, Metzger D, Auwerx J, Schoonjans K. Compromised intestinal lipid absorption in mice with a liver-specific deficiency of liver receptor homolog 1. Mol Cell Biol. 2007. 27:8330–8339.
23. Honjo Y, Sasaki S, Kobayashi Y, Misawa H, Nakamura H. 1,25-dihydroxyvitamin D3 and its receptor inhibit the chenodeoxycholic acid-dependent transactivation by farnesoid X receptor. J Endocrinol. 2006. 188:635–643.
24. Gerloff T, Geier A, Roots I, Meier PJ, Gartung C. Functional analysis of the rat bile salt export pump gene promoter. Eur J Biochem. 2002. 269:3495–3503.
25. Scotto KW. Transcriptional regulation of ABC drug transporters. Oncogene. 2003. 22:7496–7511.
26. Kast HR, Goodwin B, Tarr PT, Jones SA, Anisfeld AM, Stoltz CM, Tontonoz P, Kliewer S, Willson TM, Edwards PA. Regulation of multidrug resistance-associated protein 2 (ABCC2) by the nuclear receptors pregnane X receptor, farnesoid X-activated receptor, and constitutive androstane receptor. J Biol Chem. 2002. 277:2908–2915.
27. Simon FR, Iwahashi M, Hu LJ, Qadri I, Arias IM, Ortiz D, Dahl R, Sutherland E. Hormonal regulation of hepatic multidrug resistance-associated protein 2 (Abcc2) primarily involves the pattern of growth hormone secretion. Am J Physiol Gastrointest Liver Physiol. 2006. 290:G595–G608.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download