Abstract
Background
The ratio of glycated albumin to glycated hemoglobin (GA/A1c) is known to be elevated in subjects with type 2 diabetes mellitus (T2DM) who had decreased insulin secretion. Additionally, the carotid intima media thickness (IMT) is greater in T2DM patients with higher GA/A1c ratios. We investigated whether increased GA/A1c ratio and IMT are also associated in type 1 diabetes mellitus (T1DM), which is characterized by lack of insulin secretory capacity.
Methods
In this cross-sectional study, we recruited 81 T1DM patients (33 men, 48 women; mean age 44.1±13.0 years) who underwent carotid IMT, GA, and HbA1c measurements.
Results
The mean GA/A1c ratio was 2.90. Based on these results, we classified the subjects into two groups: group I (GA/A1c ratio <2.90, n=36) and group II (GA/A1c ratio ≥2.90, n=45). Compared with group I, the body mass indexes (BMIs), waist circumferences, and IMTs were lower in group II. GA/A1c ratio was negatively correlated with BMI, urine albumin to creatinine ratio (P<0.001 for both), and both the mean and maximal IMT (P=0.001, both). However, after adjusting the confounding factors, we observed that IMT was no longer associated with GA/A1c ratio.
Conclusion
In contrast to T2DM, IMT was not significantly related to GA/A1c ratio in the subjects with T1DM. This suggests that the correlations between GA/A1c ratio and the parameters known to be associated with atherosclerosis in T2DM could be manifested differently in T1DM. Further studies are needed to investigate these relationships in T1DM.
Although glycated albumin (GA) was initially viewed as an adjunct to glycated hemoglobin (HbA1c) in initial assessments and then as a part of continuing diabetes care, recently mounting evidence has demonstrated the contributing roles of GA to the pathogenesis of atherosclerosis [1,2,3]. In accordance with these reports, we recently demonstrated that GA has atherogenic effects that lead to the development of atherosclerosis in subjects with type 2 diabetes mellitus (T2DM) based on cross-sectional and longitudinal analyses [4,5,6]. In those studies, the serum GA level and the ratio of GA to HbA1c (GA/A1c) were identified as potential surrogate parameters that are associated with or predict the progression of atherosclerosis in T2DM subjects [5,6]. It has been demonstrated that the GA/A1c ratio of T2DM increases in parallel with decreasing insulin secretory function [7], which results in elevated postprandial blood glucose and glucose variability [8]. Considering that insulin resistance and its related inflammatory biomarkers are associated with the risk factors of atherosclerosis and/or atherosclerosis itself, the GA/A1c ratio of subjects with T2DM might be associated with intima media thickness (IMT) as an additional mechanism.
In contrast to the results of the study of T2DM, the recently published Diabetes Control and Complications Trial (DCCT)/Epidemiology of Diabetes Interventions and Complications study found no association between GA and cardiovascular events in subjects with type 1 diabetes mellitus (T1DM) [9]. While the reason for this finding has not yet been elucidated, it may likely be due to the differences in pathologic mechanisms between T2DM and T1DM. Unlike T2DM, there is a dearth of insulin secretion in T1DM, which might result in different correlations between GA/A1c ratio and various biomarkers as compared with T2DM. Therefore, to confirm this hypothesis, we attempted to identify the associations between GA/A1c ratio and various other known risk factors for cardiovascular diseases in subjects with T1DM.
This study was a retrospective, cross-sectional investigation of T1DM patients. We included T1DM patients who were registered in the Severance Hospital Diabetes Registry from August 2011 to January 2013. T1DM was defined by a glucose level greater than 200 mg/dL, clinical characteristics, positivity for antiglutamic acid decarboxylase, and a decreased C-peptide level (below 0.6 ng/mL) at the initial visit. T1DM patients were included in the study if they satisfied the following criteria: (1) aged 20 to 75 years; (2) had undergone carotid IMT measurements; (3) had been tested for both HbA1c and GA every 3 or 6 months; and (4) no documented symptoms, signs, or history of atherosclerotic vascular disease (e.g., cerebrovascular accidents, angina, myocardial infarction, or peripheral vascular disease). The exclusion criteria were as follows: (1) hematological and/or malignant disease; (2) chronic kidney disease (creatinine >1.5 mg/dL); and (3) plasma aspartate transaminase (AST) and/or alanine transaminase (ALT) levels 2-fold higher than the upper limit of the normal range. Hypertension was defined as two documented measurements of systolic blood pressure above 140 mm Hg and/or diastolic blood pressure above 90 mm Hg according to the JNC 7 Report [10] or present treatment with antihypertensive drugs. Dyslipidemia was defined by serum concentrations of total cholesterol ≥240 mg/dL [11], triglyceride concentrations exceeding 150 mg/dL, or current treatment with lipid-lowering agents.
We performed routine screening laboratory tests and IMT measurements for 86 patients, of whom we excluded five patients for age differences (n=2) and chronic kidney disease (n=3). We calculated the GA/A1c ratios and classified the remaining patients into two groups based on their mean value of this measurement (group I, GA/A1c <2.90; and group II, GA/A1c ≥2.90). This study protocol was approved by the Institutional Review Board of Severance Hospital (No. 2013-0917-001), which waived the requirement of informed consent.
Blood samples were collected from subjects after overnight fasts. The fasting glucose and 2-hour postprandial glucose after a conventional meal were measured. Plasma glucose was measured using the glucose oxidase method. Plasma total cholesterol, triglyceride, high density lipoprotein cholesterol, blood urea nitrogen, creatinine, AST, and ALT levels were assayed using routine procedures with a Hitachi 7600 auto analyzer (Hitachi Instruments Service, Tokyo, Japan). Low density lipoprotein cholesterol was calculated using the Friedewald equation. Serum C-peptide levels were measured in duplicate with immunoradiometric assays (Beckman Coulter, Fullerton, CA, USA). Urine albumin to creatinine ratio (ACR) was calculated using the results of urine microanalysis (Hitachi 7180; Hitachi Instruments Service). Serum GA was determined by an enzymatic method utilizing an albumin-specific proteinase, ketamine oxidase, albumin assay reagents (LUCICA GA-L; Asahi Kasei Pharma Co., Tokyo, Japan), and a Hitachi 7699 P module auto-analyzer; the coefficient of variation (CV) for these measurements was 1.43%. Serum HbA1c was measured via high-performance liquid chromatography using a Variant II Turbo system (BioRad Laboratories, Hercules, CA, USA). HbA1c measurements were standardized to the reference method according to the DCCT and the National Glycohemoglobin Standardization Program standards [12]. The reference interval was 20 to 40 mmol/mol (4.0% and 6.0%) for HbA1c; for GA, this interval was 11.0% to 16.0%.
Ultrasound measurements of the common carotid arteries were obtained with two sonographers using an Aloka ProSound ALPHA 10 (Hitachi) with a 13 MHz linear probe. All measurements were collected with the patients in the supine position with their heads elevated to 45° and tilted to either side at 30° depending on the side being examined. We also performed bilateral B-mode examinations 1.5 cm proximal to the carotid bifurcation on the far wall of the common carotid artery. IMT was defined as the distance between the media-adventitia interface and the lumen-intima interface. The average IMT was defined as the mean of the computer-based points in the region, and the maximum IMT was defined as the IMT at the maximum point of the region [13]. We assessed the reproducibility of the carotid IMT measurements and found an interobserver CV of 2.11% and a day-to-day CV of 2.06%. Plaques were defined according to the Mannheim consensus [14] and were diagnosed when the vessel wall thickness was greater than 1.5 mm or when it was at least 0.5 mm or 50% thicker than the surrounding wall.
All statistical analyses were performed with PASW version 18.0 (SPSS, Chicago, IL, USA). The Kolmogorov-Smirnov test was performed to determine whether the data were normally distributed, and this test confirmed that the GA/A1c ratio data were normally distributed. Continuous variables with normal distributions are expressed as means±standard deviation, and variables with non-normal distributions are expressed as the medians (with tertile ranges). Discrete variables are expressed as percentages. Statistical comparisons between groups that were divided according to the GA/A1c ratios were performed using Student t-tests or Mann-Whitney U tests. Spearman correlations were used to determine the relationships between GA/A1c ratio and the continuous variables. Multiple regression analysis was used to estimate multiple correlations of GA/A1c ratio with IMT and other clinical and laboratory risk factors for atherosclerosis. Comparisons yielding P<0.05 were considered significant.
A total of 81 subjects (33 men and 48 women; mean age 44.1±13.0 years) participated in this study. Table 1 shows the baseline clinical and laboratory characteristics of patients. The mean durations of diabetes and body mass index (BMI) were 13.0±7.5 years and 22.6±3.0 kg/m2, respectively. The fasting C-peptide level was 0.16±0.2 ng/mL. The serum GA and HbA1c levels were 26.2%±9.9%, and 8.7%±2.0%, respectively. The mean GA/HbA1c ratio was 2.90±0.8. Among these patients, 46 had been diagnosed with hypertension, and 51 had dyslipidemia. Antiplatelet agents had been prescribed to 31 patients, and all patients were dependent on insulin. The daily dose of insulin was 53.9±41.2 units/day. Twenty-three patients were being cotreated with oral hypoglycemic agents.
Given that GA/A1c ratio is associated with glucose fluctuations, we divided the patients into two groups according to the mean GA/A1c ratio (group I, GA/A1c <2.90; group II, GA/A1c ≥2.90). The BMIs and waist circumferences of group I were significantly higher than those of group II (23.9±2.8 kg/m2 vs. 21.4±2.7 kg/m2 and 83.7±9.3 cm vs. 77.2±8.4 cm; P<0.001, P=0.003, respectively). We noted that the serum HbA1c level was higher in group II, but this difference was not statistically significant (8.5%±1.8% vs. 8.9%±2.2%; P=0.458). However, the serum GA levels were significantly different between group I and II (21.0%±5.0% vs. 30.0%±10.4%, respectively; P<0.001), explaining the much higher GA/A1c ratio in group II (3.34±0.4) compared to group I (2.47±0.4, P<0.001). Furthermore, group II exhibited significantly lower mean and maximum IMT values compared to group I. These results suggest that T1DM patients with greater glucose fluctuations had lower IMTs, which contrasts to the results that have been observed in T2DM patients. The numbers of patients who were taking antihypertensive medications were 18 (50.0%) in group I and 17 (37.8%) in group II (chi-square, P=0.270). Statins had been prescribed for 18 patients (50.0%) in group I and 12 patients (26.7%) in group II (chi-square, P=0.031). Half of the patients (18, 50.0%) in group I and 13 (28.9%) patients in group II were being treated with antiplatelet agents (chi-square, P=0.052).
Table 2 illustrates the relationships of GA/A1c ratio with cardiovascular risk factors and glycemic indices. In all subjects, higher GA/A1c ratios were positively associated with higher postprandial glucose levels (P=0.008). In contrast, this ratio was negatively correlated with fasting C-peptide (P<0.001), BMI (P<0.001), waist circumference (P=0.001), urine ACR (P<0.001), and both mean and maximal IMTs (P=0.001 for both). However, the negative relationship between GA/A1c ratio and IMT was only statistically significant in group II (GA/A1c ≥2.90). We also analyzed the relationships of BMI with GA/A1c ratio and IMT; BMI was positively associated with IMT and negatively associated with GA/A1c ratio (Fig. 1).
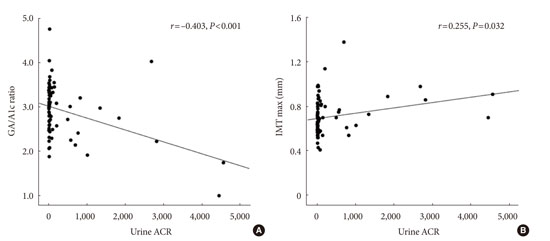
The patients were further segregated into the following three categories according to urine ACR levels: category I, normal (<30.0 µg/mg); category II, microalbuminuria (30.0 to 299.9 µg/mg); and category III, macroalbuminuria (≥300.0 µg/mg). Consequently, we observed that the GA/A1c ratio of the category II patients was significantly higher than those of the other groups, but the mean and maximum IMTs were similar across all groups (Appendix Table 1). Appendix Fig. 1 illustrates the associations of urine ACR with GA/A1c ratio and IMT; in this figure, the negative correlation between GA/A1c ratio and urine ACR (r=-0.403, P<0.001) and the positive correlation between IMT and urine ACR (r=0.255, P=0.032) can be observed.
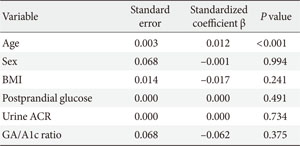
A multiple regression analysis was used to investigate the relationships between GA/A1c ratio and the other significant variables as selected from Table 2; these variables included well-known cardiovascular risk factors (i.e., BMI and urine ACR), postprandial glucose, and IMT. Due to redundancy, we omitted waist circumference from the obesity-related variables. In all patients, BMI, postprandial glucose, and urine ACR were significantly related to GA/A1c ratio (Table 3). However, IMT did not exhibit a significant relationship with GA/A1c ratio in the subjects with T1DM. To assess the associations between carotid IMT and the other risk variables (i.e., age, sex, BMI, postprandial glucose, urine ACR, and GA/A1c ratio), another multiple regression analysis was performed and indicated that age was the only factor that was statistically associated with IMT in the subjects with T1DM (Appendix Table 2).
Previous studies have demonstrated that elevated GA and GA/A1c ratios in T2DM are implicated not only in increased glycemic variability [8,15,16,17], but also in the development of atherosclerosis [2,5,6,18]. However, the associations of increased GA and GA/A1c ratio with atherosclerosis are not well understood in T1DM, in which not only insulin resistance but also absolute insulin deficits are the primary mechanisms of disease pathology [19,20,21,22]. Many questions remain to be answered regarding whether a simple and accurate glycemic index that covers glycemic variability exists and if so, whether such an index could successfully reflect glycemic variability in clinical practice. Based on the hypothesis that the GA/A1c ratio is a simple tool for the evaluation of glucose fluctuation regardless of the type of diabetes, we investigated the relationship between GA/A1c ratio and carotid atherosclerosis in subjects with T1DM.
This study has three main findings. Firstly, those with higher GA/A1c ratios had lower BMIs. This inverse correlation between glycemic variability and BMI has previously been reported in T1DM and insulin-treated T2DM patients [23,24]. Due to the retrospective nature of this study, we cannot completely explain the negative association between high GA/A1c ratios (including poor glycemic control) and low BMIs. However, considering the distinct pathophysiologic mechanisms of T1DM compared to those of T2DM (i.e., relative insulin secretory dysfunction in conjunction with increased insulin resistance plays a major role in the development of T2DM [22]), we additionally showed that increased BMI does not affect the onset and development of T1DM. One plausible explanation for the negative association between GA/A1c ratio and BMI might be the limited body reservoir for exogenous insulin-mediated glucose uptake [24]. In this regard, we postulated that decreased body mass might be the limiting factor in the facilitation of glucose uptake inside of cells in the setting of external insulin injection.
Second, only age was found to predict the development of carotid IMT in the multiple regression analysis; i.e., in this study, carotid IMT was not statistically influenced by traditionally important risk factors for cardiovascular diseases, such as high BMI and urine ACR (Appendix Table 2). Although it is possible that the association between high BMI with atherosclerosis was mediated by unknown pathophysiologic mechanisms in this T1DM population, low mean BMI (22.6±3.0 kg/m2) and relatively young age (44.1±13.0 years) might account for this result. These findings suggest that the development of atherosclerosis in T1DM might be primarily caused not by insulin resistance (i.e., high BMI) but by the aging process. In this regard, the positive association between BMI and carotid IMT in our study might not be of the clinical relevance.
Finally, GA/A1c ratio was not related to carotid IMT in T1DM. In previous studies conducted with subjects with T2DM, we have demonstrated that GA/A1c ratio, which reflects glycemic variability, positively, and significantly affects the development and progression of atherosclerosis in T2DM in which the underlying pathophysiologic mechanism of insulin resistance plays a major role in the development and aggravation of both T2DM and atherosclerosis [5,6]. Based on these findings, we hypothesized that glycemic variability might play a different and important role in the development of atherosclerosis in T1DM in which absolute insulin deficiency is the central mechanism of hyperglycemia. Indeed, our findings emphasize insulin resistance is an important factor in the development of atherosclerosis in T2DM but not in T1DM.
This study had the following limitations. First, this was a cross-sectional, retrospective study. Thus, this study failed to examine insulin resistant markers and utilize the gold standard tool of continuous glucose monitoring for evaluating glucose variability to determine the relationship between glucose fluctuation and carotid atherosclerosis in T1DM subjects. Furthermore, the cross-sectional nature of this study precluded the definite establishment of the statistical correlation between GA/A1c ratio and BMI in subjects with T1DM. Second, as discussed above, we were unable to provide a clear explanation of the association between high GA/A1c ratio and low BMI. Lastly, GA, and GA/A1c ratio might not have accurately reflected the actual glucose variability.
In this study, we evaluated the relationship between GA/A1c ratio, which reflects glucose fluctuation, and carotid atherosclerosis in Korean T1DM subjects. IMT was not significantly related to GA/A1c ratio in subjects with T1DM. Given the different pathophysiologic mechanisms of T1DM and T2DM, we suspect that glycemic variability plays a role in the development of carotid atherosclerosis only in the setting of insulin resistance. However, larger, prospective, longitudinal, and controlled studies are needed to investigate the relationship between glycemic variability as reflected by GA/A1c ratio and the progression of atherosclerosis in subjects with T1DM and insulin resistance.
Figures and Tables
Fig. 1
(A, B) Relationships of body mass index (BMI) with glycated albumin to glycated hemoglobin (GA/A1c) ratio and intima media thickness (IMT) in type 1 diabetes mellitus patients. (A) There was a positive association between BMI and IMT (r=0.331, P=0.005). (B) In contrast, BMI and GA/A1c were negatively related (r=-0.448, P<0.001).

Table 1
Baseline characteristics of the study population

Values are presented as mean±standard deviation or number (%).
GA/A1c, glycated albumin to glycated hemoglobin ratio; DM, diabetes mellitus; BMI, body mass index; WC, waist circumference; SBP, systolic blood pressure; DBP, diastolic blood pressure; TG, triglyceride; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; ACR, albumin to creatinine ratio; IMT, intima media thickness.
aP value of the comparisons between group I and II.
ACKNOWLEDGMENTS
The authors are grateful to Jae-Hyeon Kim, M.D., Ph.D. in the Samsung Medical Center for communicating with the authors and reviewing this article.
Appendices
Appendix Table 1
GA/A1c ratio and IMT according to urine ACR category
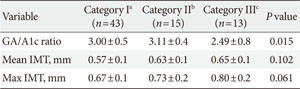
Values are presented as mean±standard deviation.
GA/A1c, glycated albumin to glycated hemoglobin ratio; IMT, intima media thickness; ACR, albumin to creatinine ratio.
aCategory I, normal (urine ACR <30.0 µg/mg), bCategory II, microalbuminuria (urine ACR 30.0 to 299.9 µg/mg), cCategory III, macroalbuminuria (urine ACR ≥300.0 µg/mg).
Appendix Fig. 1
(A, B) Correlations of urine albumin to creatinine ratio (ACR) with glycated albumin to glycated hemoglobin (GA/A1c) ratio and intima media thickness (IMT). There was a negative correlation between urine ACR and GA/A1c ratio (A, r=-0.403, P<0.001), while urine ACR and IMT were positively related (B, r=0.255, P=0.032).
References
1. Kim KJ, Lee BW. The roles of glycated albumin as intermediate glycation index and pathogenic protein. Diabetes Metab J. 2012; 36:98–107.
2. Machado-Lima A, Iborra RT, Pinto RS, Sartori CH, Oliveira ER, Nakandakare ER, Stefano JT, Giannella-Neto D, Correa-Giannella ML, Passarelli M. Advanced glycated albumin isolated from poorly controlled type 1 diabetes mellitus patients alters macrophage gene expression impairing ABCA-1-mediated reverse cholesterol transport. Diabetes Metab Res Rev. 2013; 29:66–76.
3. Okuda LS, Castilho G, Rocco DD, Nakandakare ER, Catanozi S, Passarelli M. Advanced glycated albumin impairs HDL anti-inflammatory activity and primes macrophages for inflammatory response that reduces reverse cholesterol transport. Biochim Biophys Acta. 2012; 1821:1485–1492.
4. Kim HM, Lee BW, Song YM, Kim WJ, Chang HJ, Choi DH, Yu HT, Kang E, Cha BS, Lee HC. Potential association between coronary artery disease and the inflammatory biomarker YKL-40 in asymptomatic patients with type 2 diabetes mellitus. Cardiovasc Diabetol. 2012; 11:84.
5. Moon JH, Chae MK, Kim KJ, Kim HM, Cha BS, Lee HC, Kim YJ, Lee BW. Decreased endothelial progenitor cells and increased serum glycated albumin are independently correlated with plaque-forming carotid artery atherosclerosis in type 2 diabetes patients without documented ischemic disease. Circ J. 2012; 76:2273–2279.
6. Song SO, Kim KJ, Lee BW, Kang ES, Cha BS, Lee HC. Serum glycated albumin predicts the progression of carotid arterial atherosclerosis. Atherosclerosis. 2012; 225:450–455.
7. Kim D, Kim KJ, Huh JH, Lee BW, Kang ES, Cha BS, Lee HC. The ratio of glycated albumin to glycated haemoglobin correlates with insulin secretory function. Clin Endocrinol (Oxf). 2012; 77:679–683.
8. Lee EY, Lee BW, Kim D, Lee YH, Kim KJ, Kang ES, Cha BS, Lee EJ, Lee HC. Glycated albumin is a useful glycation index for monitoring fluctuating and poorly controlled type 2 diabetic patients. Acta Diabetol. 2011; 48:167–172.
9. Nathan DM, McGee P, Steffes MW, Lachin JM. DCCT/EDIC Research Group. Relationship of glycated albumin to blood glucose and HbA1c values and to retinopathy, nephropathy, and cardiovascular outcomes in the DCCT/EDIC study. Diabetes. 2014; 63:282–290.
10. Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL Jr, Jones DW, Materson BJ, Oparil S, Wright JT Jr, Roccella EJ. National Heart, Lung, and Blood Institute Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. National High Blood Pressure Education Program Coordinating. The seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure: the JNC 7 report. JAMA. 2003; 289:2560–2572.
11. Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive summary of the third report of the National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). JAMA. 2001; 285:2486–2497.
12. Little RR. Glycated hemoglobin standardization: National Glycohemoglobin Standardization Program (NGSP) perspective. Clin Chem Lab Med. 2003; 41:1191–1198.
13. Kim SK, Park SW, Kim SH, Cha BS, Lee HC, Cho YW. Visceral fat amount is associated with carotid atherosclerosis even in type 2 diabetic men with a normal waist circumference. Int J Obes (Lond). 2009; 33:131–135.
14. Touboul PJ, Hennerici MG, Meairs S, Adams H, Amarenco P, Bornstein N, Csiba L, Desvarieux M, Ebrahim S, Fatar M, Hernandez Hernandez R, Jaff M, Kownator S, Prati P, Rundek T, Sitzer M, Schminke U, Tardif JC, Taylor A, Vicaut E, Woo KS, Zannad F, Zureik M. Mannheim carotid intima-media thickness consensus (2004-2006). An update on behalf of the Advisory Board of the 3rd and 4th Watching the Risk Symposium, 13th and 15th European Stroke Conferences, Mannheim, Germany, 2004, and Brussels, Belgium, 2006. Cerebrovasc Dis. 2007; 23:75–80.
15. Ogawa A, Hayashi A, Kishihara E, Yoshino S, Takeuchi A, Shichiri M. New indices for predicting glycaemic variability. PLoS One. 2012; 7:e46517.
16. Vos FE, Schollum JB, Coulter CV, Manning PJ, Duffull SB, Walker RJ. Assessment of markers of glycaemic control in diabetic patients with chronic kidney disease using continuous glucose monitoring. Nephrology (Carlton). 2012; 17:182–188.
17. Matsumoto H, Murase-Mishiba Y, Yamamoto N, Sugitatsu-Nakatsukasa S, Shibasaki S, Sano H, Terasaki J, Imagawa A, Hanafusa T. Glycated albumin to glycated hemoglobin ratio is a sensitive indicator of blood glucose variability in patients with fulminant type 1 diabetes. Intern Med. 2012; 51:1315–1321.
18. Lu L, Pu LJ, Zhang Q, Wang LJ, Kang S, Zhang RY, Chen QJ, Wang JG, De Caterina R, Shen WF. Increased glycated albumin and decreased esRAGE levels are related to angiographic severity and extent of coronary artery disease in patients with type 2 diabetes. Atherosclerosis. 2009; 206:540–545.
19. Rewers M. Challenges in diagnosing type 1 diabetes in different populations. Diabetes Metab J. 2012; 36:90–97.
20. Lee HK. Mitochondrial dysfunction and insulin resistance: the contribution of dioxin-like substances. Diabetes Metab J. 2011; 35:207–215.
21. Gonzalez-Chavez A, Elizondo-Argueta S, Gutierrez-Reyes G, Leon-Pedroza JI. Pathophysiological implications between chronic inflammation and the development of diabetes and obesity. Cir Cir. 2011; 79:209–216.
22. Rhee SY, Woo JT. The prediabetic period: review of clinical aspects. Diabetes Metab J. 2011; 35:107–116.
23. Greven WL, Beulens JW, Biesma DH, Faiz S, de Valk HW. Glycemic variability in inadequately controlled type 1 diabetes and type 2 diabetes on intensive insulin therapy: a cross-sectional, observational study. Diabetes Technol Ther. 2010; 12:695–699.
24. Murata GH, Duckworth WC, Shah JH, Wendel CS, Hoffman RM. Sources of glucose variability in insulin-treated type 2 diabetes: the Diabetes Outcomes in Veterans Study (DOVES). Clin Endocrinol (Oxf). 2004; 60:451–456.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download