Abstract
Background
The Korea National Health and Nutrition Examination Survey (KNHANES) III (2005) reported that 22.9% of individuals with diabetes have a glycated hemoglobin (HbA1c) <6.5% and that 43.5% have an HbA1c <7%. We investigated the levels of glycemic control and the factors associated with glycemic control using data from the KNHANES V (2010 to 2012).
Methods
Subjects with diabetes diagnosed by a physician or those taking antidiabetic medications were classified as individuals with known diabetes. Of 1,498 subjects aged ≥30 years with diabetes, we excluded 157 individuals who were missing HbA1c data. A total of 1,341 subjects were included in the final analysis.
Results
The prevalence of known diabetes was 7.7% (n=1,498, estimated to be 2.32 million people). The proportions of well-controlled diabetes meeting a HbA1c goal of <6.5% and <7% were 27% and 45.6%, respectively. HbA1c increased as the duration of diabetes increased. HbA1c in subjects with a duration of diabetes ≤5 years was lower than in subjects with a duration >5 years. HbA1c in the group taking only oral hypoglycemic agents (OHAs) was significantly lower than that in the group administered only insulin or OHA and insulin in combination. In logistic regression analysis, a longer duration of diabetes, insulin use and the absence of chronic renal failure were associated with HbA1c levels >6.5%.
Diabetes, which is characterized by persistent hyperglycemia and various consequent complications, is the 5th leading cause of death in Korea [1]. More than 50% of cases of acquired blindness, chronic renal failure requiring renal replacement therapy and nontraumatic limb loss are related to microvascular complications of diabetes [2]. Diabetes is closely related to various cardiovascular diseases, including coronary arterial disease, stroke, peripheral arterial disease, and cardiomyopathy; these comorbidities occur in 70% to 80% of patients with type 2 diabetes mellitus, ultimately leading to death [3]. Westernized lifestyles and the persistent increase in obesity in Korea have led to the present situation; 12.4% of adults over 30 years of age are diagnosed with diabetes [4]. Furthermore, 38.3% of adult subjects are prediabetic and are very likely to progress to diabetes, and these increasing trends are expected to continue [4]. The increase in diabetes will consequently result in an increase in diabetes-related morbidity, thus becoming a socioeconomic burden.
Previous studies revealed that thorough glycemic control is essential for preventing the microvascular and macrovascular complications of diabetes. Based on these results, many organizations, including the Korean Diabetes Association (KDA), recommend maintaining a glycemic level below 6.5% to 7.0%. However, the various causes of diabetes, the continuously progressing characteristic, the importance of changing lifestyles, etc., make it difficult in practice to control blood glucose levels. From the Korea National Health and Nutrition Examination Survey (KNHANES) III (2005), adequate glycemic control was achieved in 43.5% and 22.9% of diabetic patients, with A1c levels <7.0% and <6.5%, respectively [5]. As is shown by similar results from a study using the health insurance data from the Health Insurance Review & Assessment Service, only 40.3% of patients managed to maintain their blood glucose level below 7.0% [6]. Thus, more than half of diabetic patients are exposed to the threat of chronic complications secondary to persistent hyperglycemia. Therefore, recognizing the problems in controlling the glycemic level at present and seeking a solution will be crucial in reducing the morbidity and mortality caused by diabetes [6].
We investigated the levels of glycemic control and the factors associated with glycemic control using data from the KNHANES V (2010 to 2012).
We analyzed data from the 5th KNHANES V (2010 to 2012). The KNHANES is a nationally representative, cross-sectional survey designed to estimate the health and nutritional status of the Korean population and consists of three distinct surveys: a health interview survey, health examination survey, and nutrition survey. A total of 25,534 individuals participated in the survey, and 9,103 subjects aged <30 years were excluded. Of 16,431 subjects aged ≥30 years, we excluded 631 subjects as nonrespondents on a previous diagnosis of diabetes made by physicians and/or current use of antidiabetic medications including insulin and oral hypoglycemic agents (OHAs). Subjects with diabetes diagnosed by physician or those taking anti-diabetic medications were classified as patients with known diabetes. Among subjects with known diabetes (n=1,498), 157 individuals without glycated hemoglobin (HbA1c) data were excluded. A final total of 1,341 subjects were included in this analysis (Fig. 1).
The presence of smoking history is defined more than 5 pack-years of smoking; the presence of alcohol history is defined as ever having consumed alcohol. A family history of diabetes refers to individuals who have a first degree family member with a diagnosis of diabetes made by a physician.
After fasting for 8 hours or more, blood was drawn from the antecubital vein of each participant. The samples were properly processed, refrigerated at 2℃ to 8℃, and transported to the Central Testing Institute in Seoul, Korea. Blood samples were analyzed within 24 hours of transportation. Fasting glucose was measured using a Hitachi Automatic Analyzer 7600 (Hitachi, Tokyo, Japan). High performance liquid chromatography-723G7 (Tosoh, Tokyo, Japan) was used to check HbA1c. We defined chronic kidney disease (CKD) as estimated glomerular filtration rates of less than 60 mL/min/1.73 m2, as calculated by the Cockcroft-Gault formula.
All analyses were conducted using SPSS version 19.0 (IBM Co., Armonk, NY, USA). We used the KNHANES sampling weight variables with stratification and clustering variables to incorporate sample weights and adjust the analysis for the complex sample design of the survey. Nominal variables are presented as the number of cases, and percentage, and continuous variables are presented as the mean±standard deviation. To identify the factors involved in controlling glycemic levels, logistic-regression models adjusted for age, sex, body mass index, diabetes duration, use or nonuse of insulin, family history of diabetes, smoking status, alcohol consumption, renal function, house income, and educational level were used. Statistical significance was defined as a 2-tailed P value less than 0.05.
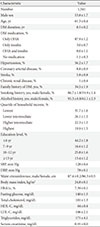
We first observed the baseline characteristics of the 1,341 diabetic patients. Approximately 53.8% were male, the average age was 61.3 years old, and the average diabetes duration was 8.5 years. The average waist size was 87.4 cm for males and 86.3 cm for females, and the average body mass index was 24.8 kg/m2. Approximately 87.9% of the patients were treated with OHA, 11% were treated with insulin, and 1.1% were treated without any medication. Approximately 56.2% of the patients were also diagnosed with hypertension, and the proportion of patients diagnosed with coronary artery diseases or stroke was 8.8% and 5.8%, respectively. The average HbA1c was 7.39% (7.34% for males, 7.46% for females), and the average fasting glucose concentration was 140 mg/dL (Table 1). The prevalence of known diabetes was 7.7%, estimated to be 2.32 million people.
With respect to the overall glycemic control, 45.6% reached the target of <7.0% HbA1c, according to the American Diabetes Association standard, whereas 27% reached the target when the standard was set to <6.5% HbA1c, according to the International Diabetes Federation and KDA standard (Fig. 2). According to the treatment method, the HbA1c level was 7.27% for the group treated with OHA, which was the lowest, and was 8.68% for the group treated concurrently with insulin and OHA, which was the highest (Fig. 3). The average HbA1c level also varied according to the diabetes duration, with levels of 7.15%, 7.49%, 7.75%, and 7.79% for the ≤5, 6 to 10, 11 to 20, and >20 years groups, respectively, indicating that A1c level increases proportionally to the diabetes duration (Fig. 4A). However, this result was only distinct in the group treated only with OHA, and we observed no differences in the average A1c level according to the duration in groups that are treated with insulin (Fig. 4B).
To uncover the variables related to the diabetic control state, we performed a multiple logistic regression analysis and discovered that diabetes duration, insulin use and the absence of chronic renal failure were statistically significantly correlated to poor glycemic control (Table 2).
Glycemic control is an essential component for the prevention of chronic complications in diabetic patients. However, the various causes of diabetes, the treatment medications, the lifestyle modifications, and the need for a team approach including doctors, nurses, nutritionists, and physical therapists only achieve glycemic control in approximately 50% of patients reaching their goal of glycemic levels [5,7]. In other words, half of patients are constantly exposed to hyperglycemia, with increased risks of developing complications and ultimately leading to elevated morbidity and mortality.
Our data from KNHANES V (2010 to 2012) also revealed that only 27% reached the HbA1c goal of 6.5%, and only 45.6% succeeded even if the goal was set to 7%. These proportions are not very different from the previous results from KNHANES 2005, which demonstrated that only 24% were below 6.5% and that only 42.5% were below 7.0% [5]. If newly diagnosed diabetes refers to subjects with fasting plasma glucose (FPG) ≥126 mg/dL and/or HbA1c ≥6.5% in the absence of known diabetes, the prevalence of newly diagnosed diabetes is 4%. The mean HbA1c and FPG among the newly diagnosed diabetic subjects is 7.2% and 143 mg/dL, respectively. Approximately 63.4% of newly diagnosed diabetics reached the target of <7.0% HbA1c. Therefore, when we include the cases of newly diagnosed diabetes in the analysis, the proportion of patients reaching the HbA1c goal would be greater. Between 2005 and 2012, a new proposal recommended that patients begin diet and exercise plans as well as take oral medication, including metformin, immediately after they had been diagnosed with diabetes to reach the target early for diabetes treatment, and incretin-based therapy was introduced to the clinics for patient treatment and has since been used actively [8]. Thus, our data suggest that despite the medical advancements in guidelines and treatment methods, the target success rate remains unchanged. Such a phenomenon, however, is not only limited to Korea; data from a similar period in America were similar, revealing a success rate of 56.8% in reaching the HbA1c level of 7.0% from 2003 to 2006 and a success rate of 52.5% from 2007 to 2010 [7]. Thus, now is the time when ground-breaking action is required to exhaustively analyze the reasons behind the dismal success rate of diabetes control and to thereby develop a new approach to intensively control glycemic levels. A study from America that was conducted over a similar time period, from 2007 to 2010, reported that only 52.5% of patients maintained an HbA1c level below 7.0%, which is 6.9% higher than our results of 45.6% [7]. Although the differences in lifestyles, socioeconomic status, medical treatment system, and research methods may be the reason for the difference, one of the biggest differences between Korea and America was the proportion of people using insulin between groups. Although only 11% of patients with diabetes used insulin in Korea, 27% to 30% of American patients received insulin treatment, nearly 3-fold higher than in Korea. This discrepancy may indicate that Korea does not begin insulin treatment sufficient early or at an appropriate time, and this delay in the start of insulin treatment may be the reason for the poor glycemic control compared to America. However, 27.3% of the patients were treated with insulin, and yet only 34.5% exhibited HbA1c levels below 7.0% in Taiwan, which is lower than in America or in Korea, thereby making it difficult to explain the differences solely based on the portion of insulin usage [9]. There is also a difference in the implementation of education for diabetes. Although 54.6% received education in America, only 39.4% received education in Korea, which allows us to deduce that understanding the disease, recognizing the importance of diet and exercise, and motivating self-care through diabetes education has led to the superior glycemic control in America [6,7].
Our inspection of the factors involved in glycemic control revealed that longer durations of diabetes, insulin use, and the absence of CKD were all related to poorer glycemic control. Given that more cases that require insulin arose as the duration of diabetes increased (3.1%, 9.2%, 22.4%, and 35.4% of the patients used insulin after being diagnosed with diabetes for ≤5, 6 to 10, 11 to 20, and >20 years, respectively), we suggest that the duration of diabetes is the most important parameter affecting glycemic control. It is well-known that β-cell function deteriorates as the duration of diabetes lengthens and therefore results in poor glycemic control [10,11]. These results also indicate that even if the duration of diabetes is prolonged, good glycemic control can be maintained if the functions of β-cells are well-preserved. Therefore, choosing treatment methods that aim to maximize the preservation of β-cell function is beneficial in glycemic control in the long term. Therefore, drugs such as thiazolidinedione that have indirect effects of reducing lipotoxicity and glucotoxicity as well as direct effects on activating peroxisome proliferator-activated receptor γ of β-cells to augment their function have been a mainstay to increase the durability of β-cells, but side-effects such as heart failure, weight gain, and bladder cancer have prevented the drug's routine usage [12,13]. The recently popular incretin-based therapy also theoretically stimulates β-cell regeneration and hypertrophy to enhance its function and rejuvenation, but actual clinical results proving that β-cell function improves remain scarce, and more long-term research is needed [14,15].
At the current point in time, we need to devise plans to transfer the 28.6% of the patients with 7.0% to 7.9% HbA1c levels, i.e., those patients within 1% of the HbA1c target range, to the ≤7.0% group to alleviate the general glycemic control state. First, diabetes education, including education for lifestyle modifications that amplify the ability for self-care must be strengthened. The education rate, which remains below 40%, must be improved, and doctors, nurses, nutritionists, and physical therapists must form a team to provide systematic support for efficient education. Furthermore, physicians must devote increased efforts to allow patients who have difficulty in controlling their glycemic level to progress to the next stages of treatment, including early insulin usage. The longer patients are exposed to hyperglycemia, the more β-cell function deteriorates, which in turn further worsens hyperglycemia, resulting in a vicious feedback circle. Thus, this feedback circle must be put to a stop early to stabilize glycemic levels and normalize β-cell function so that the long-term target glycemic level can be maintained. To achieve this goal, it is important to quickly progress to the next plan of action as soon as the glycemic level jumps outside its target range. Finally, the glycemic level must be controlled in insulin users. Despite using insulin, which is the strongest hypoglycemic agent, the average glycemic level was higher than in the OHA group, and we speculate that the main cause for this difference is that insulin treatment is started after the point at which β-cell function is depleted, thus beyond the point at which the glycemic level can be controlled by OHA. By this time, patients are struck with a sense of shame in using insulin, despair in using the last possible means of cure, fear of hypoglycemic phenomena, etc., which makes it increasingly more difficult to definitively control glycemic levels. Therefore, diabetes education that corrects the wrongly understood perception of insulin, reminds patients of the importance of diet and exercise, trains the patients to control their own insulin usage volume and frequency, etc., are essential.
This study has some limitations. We cannot define diabetes subtypes in these KNHANES data. The diabetic patients aged 30 to 39 represented 3.4% of the total number of diabetics, and insulin-only treated patients represented 35.5% of patients aged 30 to 39. Therefore, 1.2% of all diabetic patients may be regarded as type 1 diabetes patients. This small percent had little impact on these analyses. Furthermore, the actual duration of diabetes is difficult to determine. Because many patients with type 2 diabetes have chronic diabetic complications at the time of diagnosis, many patients likely experience a long asymptomatic period of hyperglycemia. This measurement error may affect the analysis of the duration of diabetes.
In conclusion, the level of adequate glycemic control was similar but slightly improved relative to previous data. The glycemic control of patients with long-standing diabetes can be harder even though they receive insulin treatment. Various actions to strengthen diabetes education, to use insulin at appropriate points in time, etc., are required to improve glycemic control.
Figures and Tables
Fig. 1
Study population framework. KNHANES, Korea National Health and Nutrition Examination Survey; HbA1c, glycated hemoglobin.

Fig. 2
The distribution of diabetic patients according to glycated hemoglobin (HbA1c, %) from the Korea National Health and Nutrition Examination Survey (KNHANES) III (2005) and KNHANES V (2012).

Fig. 3
The distribution of average glycated hemoglobin (HbA1c) according to the modality of diabetes treatment. OHA, oral hypoglycemic agent; DM, diabetes mellitus. aP<0.05 compared with the only OHA group.

Fig. 4
(A) The distribution of average glycated hemoglobin (HbA1c) according to the duration of diabetes. (B) The distribution of average HbA1c according to the duration of diabetes and the modality of its treatment. OHA, oral hypoglycemic agent; DM, diabetes mellitus. aP<0.05 compared with the ≤5 years group, bP<0.05 compared with the same duration OHA group.

References
1. Statistics Korea: Causes of death statistics 2012. updated 2013 Sep 25. Available from: http://kostat.go.kr.
2. Nolan CJ, Damm P, Prentki M. Type 2 diabetes across generations: from pathophysiology to prevention and management. Lancet. 2011; 378:169–181.
3. Beckman JA, Creager MA, Libby P. Diabetes and atherosclerosis: epidemiology, pathophysiology, and management. JAMA. 2002; 287:2570–2581.
4. Jeon JY, Ko SH, Kwon HS, Kim NH, Kim JH, Kim CS, Song KH, Won JC, Lim S, Choi SH, Jang MJ, Kim Y, Oh K, Kim DJ, Cha BY. Taskforce Team of Diabetes Fact Sheet of the Korean Diabetes Association. Prevalence of diabetes and prediabetes according to fasting plasma glucose and HbA1c. Diabetes Metab J. 2013; 37:349–357.
5. Choi YJ, Kim HC, Kim HM, Park SW, Kim J, Kim DJ. Prevalence and management of diabetes in Korean adults: Korea National Health and Nutrition Examination Surveys 1998-2005. Diabetes Care. 2009; 32:2016–2020.
6. Task Force Team for Basic Statistical Study of Korean Diabetes Mellitus of Korean Diabetes Association. Park IB, Kim J, Kim DJ, Chung CH, Oh JY, Park SW, Lee J, Choi KM, Min KW, Park JH, Son HS, Ahn CW, Kim H, Lee S, Lee IB, Choi I, Baik SH. Diabetes epidemics in Korea: reappraise nationwide survey of diabetes "diabetes in Korea 2007". Diabetes Metab J. 2013; 37:233–239.
7. Ali MK, Bullard KM, Saaddine JB, Cowie CC, Imperatore G, Gregg EW. Achievement of goals in U.S. diabetes care, 1999-2010. N Engl J Med. 2013; 368:1613–1624.
8. Nathan DM, Buse JB, Davidson MB, Heine RJ, Holman RR, Sherwin R, Zinman B. Professional Practice Committee American Diabetes Association. European Association for the Study of Diabetes. Management of hyperglycaemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy. A consensus statement from the American Diabetes Association and the European Association for the Study of Diabetes. Diabetologia. 2006; 49:1711–1721.
9. Yu NC, Su HY, Chiou ST, Yeh MC, Yeh SW, Tzeng MS, Sheu WH. Trends of ABC control 2006-2011: a National Survey of Diabetes Health Promotion Institutes in Taiwan. Diabetes Res Clin Pract. 2013; 99:112–119.
10. UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998; 352:837–853.
11. Wajchenberg BL. beta-cell failure in diabetes and preservation by clinical treatment. Endocr Rev. 2007; 28:187–218.
12. Walter H, Lubben G. Potential role of oral thiazolidinedione therapy in preserving beta-cell function in type 2 diabetes mellitus. Drugs. 2005; 65:1–13.
13. Cariou B, Charbonnel B, Staels B. Thiazolidinediones and PPARgamma agonists: time for a reassessment. Trends Endocrinol Metab. 2012; 23:205–215.
14. Campbell JE, Drucker DJ. Pharmacology, physiology, and mechanisms of incretin hormone action. Cell Metab. 2013; 17:819–837.
15. Drucker DJ, Sherman SI, Gorelick FS, Bergenstal RM, Sherwin RS, Buse JB. Incretin-based therapies for the treatment of type 2 diabetes: evaluation of the risks and benefits. Diabetes Care. 2010; 33:428–433.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download