Abstract
Objective
Promoting axonal regeneration is one of essential goals to be achieved for effective repair from spinal cord injury (SCI). The authors performed an experiment on the effect of Wnt-3a producing fibroblast injection on SCI.
Methods
SCI is performed by New York University (NYU) impactor in 21 rats, and 1 week after injury, the rats were grouped into Wnt-3a producing fibroblast transplantation group (Wnt group, n=7), fibroblast transplantation group (vehicle group, n=7), and no treatment group (sham group, n=7). For the behavioral tests, the Basso, Beattie and Bresnahan (BBB) locomotor rating scale were evaluated. Also, to study radiological evidence of axon regeneration, the rats underwent manganese enhanced MRI (ME-MRI) twice, before treatment and 5 weeks after treatment.
Results
On average, the sham group and vehicle group rats attained BBB score of 10.4 and 12.1, respectively, whereas Wnt group rats achieved a significantly higher BBB score of 14.0 at 5 weeks after treatment. At 5 weeks after treatment, relative signal intensities of ME-MRI was significantly higher in the Wnt group than in the vehicle group and sham group at 5 weeks after injury.
Figures and Tables
FIGURE 1
The spinal cord injury was performed according to the NYU system developed by Gruner 1992. NYU: New York University.

FIGURE 2
ME-MRI of Wnt group. A: At a week after injury, ME-MRI revealed showed manganese uptake was decreased at caudal to the lesion (arrowhead). B: At 5 weeks after treatment, ME-MRI revealed that manganese uptake by active neuronal tissue resulted in signal enhancement below injury level (arrowhead). ME-MRI: manganese enhanced MRI.
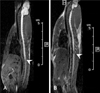
FIGURE 3
Diagram of BBB locomotor scale. At 1 week after injury, the rats were treated (arrow). 5 weeks after treatment, mean values of Wnt group showed significant improvement compared to vehicle group and sham group (*p<0.001). BBB: Basso, Beattie and Bresnahan, AVOVA: analysis of variance.

FIGURE 4
Diagram of the relative signal intensities. A week after injury, the relative signals of all three groups have no significant differences (left histogram of each group). 5 weeks after treatment, mean values of the relative signal intensities showed significant differences between three groups, statistically (right histogram of each group, *p<0.001).

References
1. Arévalo JC, Chao MV. Axonal growth: where neurotrophins meet Wnts. Curr Opin Cell Biol. 2005; 17:112–115.

2. Bilgen M, Dancause N, Al-Hafez B, He YY, Malone TM. Manganese-enhanced MRI of rat spinal cord injury. Magn Reson Imaging. 2005; 23:829–832.

3. Blesch A, Tuszynski MH. Spontaneous and neurotrophin-induced axonal plasticity after spinal cord injury. Prog Brain Res. 2002; 137:415–423.
4. Cheng H, Cao Y, Olson L. Spinal cord repair in adult paraplegic rats: partial restoration of hind limb function. Science. 1996; 273:510–513.

5. Grill RJ, Blesch A, Tuszynski MH. Robust growth of chronically injured spinal cord axons induced by grafts of genetically modified NGF-secreting cells. Exp Neurol. 1997; 148:444–452.

6. Guest JD, Hesse D, Schnell L, Schwab ME, Bunge MB, Bunge RP. Influence of IN-1 antibody and acidic FGF-fibrin glue on the response of injured corticospinal tract axons to human Schwann cell grafts. J Neurosci Res. 1997; 50:888–905.

8. Kalb RG, Strittmatter SM. Neurobiology of spinal cord injury. Totowa, NJ: Humana Press;2000. p. 284.
9. Logan CY, Nusse R. The Wnt signaling pathway in development and disease. Annu Rev Cell Dev Biol. 2004; 20:781–810.

10. Lu W, Yamamoto V, Ortega B, Baltimore D. Mammalian Ryk is a Wnt coreceptor required for stimulation of neurite outgrowth. Cell. 2004; 119:97–108.

11. Schnell L, Schneider R, Kolbeck R, Barde YA, Schwab ME. Neurotrophin-3 enhances sprouting of corticospinal tract during development and after adult spinal cord lesion. Nature. 1994; 367:170–173.

12. Silva AC, Lee JH, Aoki I, Koretsky AP. Manganese-enhanced magnetic resonance imaging (MEMRI): methodological and practical considerations. NMR Biomed. 2004; 17:532–543.

13. Toledo EM, Colombres M, Inestrosa NC. Wnt signaling in neuroprotection and stem cell differentiation. Prog Neurobiol. 2008; 86:281–296.

14. Walder N, Petter-Puchner AH, Brejnikow M, Redl H, Essig M, Stieltjes B. Manganese enhanced magnetic resonance imaging in a contusion model of spinal cord injury in rats: correlation with motor function. Invest Radiol. 2008; 43:277–283.

15. Ye JH, Houle JD. Treatment of the chronically injured spinal cord with neurotrophic factors can promote axonal regeneration from supraspinal neurons. Exp Neurol. 1997; 143:70–81.

16. Yoshikawa S, McKinnon RD, Kokel M, Thomas JB. Wnt-mediated axon guidance via the Drosophila Derailed receptor. Nature. 2003; 422:583–588.

17. Young W. Spinal cord contusion models. Prog Brain Res. 2002; 137:231–255.
18. Zhou FQ, Zhou J, Dedhar S, Wu YH, Snider WD. NGF-induced axon growth is mediated by localized inactivation of GSK-3beta and functions of the microtubule plus end binding protein APC. Neuron. 2004; 42:897–912.
19. Zumbrunn J, Kinoshita K, Hyman AA, Näthke IS. Binding of the adenomatous polyposis coli protein to microtubules increases microtubule stability and is regulated by GSK3 beta phosphorylation. Curr Biol. 2001; 11:44–49.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download