Abstract
Postpartum thyroid dysfunction occurs in 5–10% of women within one year after delivery. Women with hypo-thyroidism antedating pregnancy are at high risk for postpartum thyroiditis and should be closely monitored during the first year post-partum. Here, we report a case of recurrent hyperthyroidism between two episodes of postpartum thyroiditis in a woman diagnosed with subclinical hypothyroidism prior to pregnancy. It is of particular interest that spontaneously remitting hyperthyroidism as a sequela of postpartum thyroiditis can occur.
References
1. Ide A, Amino N, Kang S, Yoshioka W, Kudo T, Nishihara E, et al. Differentiation of postpartum Graves' thyrotoxicosis from postpartum destructive thyrotoxicosis using antithyrotropin receptor antibodies and thyroid blood flow. Thyroid. 2014; 24(6):1027–31.

2. Momotani N, Noh J, Ishikawa N, Ito K. Relationship between silent thyroiditis and recurrent Graves' disease in the postpartum period. J Clin Endocrinol Metab. 1994; 79(1):285–9.

3. Amino N, Tanizawa O, Mori H, Iwatani Y, Yamada T, Kurachi K, et al. Aggravation of thyrotoxicosis in early pregnancy and after delivery in Graves' disease. J Clin Endocrinol Metab. 1982; 55(1):108–12.

4. Sartani A, Feigl D, Zaidel L, Ravid M. Painless thyroiditis followed by autoimmune disorders of the thyroid. A case report with biopsy. J Endocrinol Invest. 1980; 3(2):169–72.

5. Sarlis NJ, Brucker-Davis F, Swift JP, Tahara K, Kohn LD. Graves' disease following thyrotoxic painless thyroiditis. Analysis of antibody activities against the thyrotropin receptor in two cases. Thyroid. 1997; 7(6):829–36.

6. Shorey S, Badenhoop K, Walfish PG. Graves' hyperthyroidism after postpartum thyroiditis. Thyroid. 1998; 8(12):1117–22.

7. Papi G, Corrado S, Carapezzi C, Corsello SM. Postpartum thyroiditis presenting as a cold nodule and evolving to Graves' disease. Int J Clin Pract. 2003; 57(6):556–8.
8. Yang JH, Han EJ, Yim CH. Two cases of postpartum thyroiditis followed by Graves' disease. J Korean Thyroid Assoc. 2013; 6(1):75–9.

9. Sergi M, Tomlinson G, Feig DS. Changes suggestive of postpartum thyroiditis in women with established hypothyroidism: incidence and predictors. Clin Endocrinol (Oxf). 2014. [Epub ahead of print].

10. Amino N, Mori H, Iwatani Y, Tanizawa O, Kawashima M, Tsuge I, et al. High prevalence of transient postpartum thyrotoxicosis and hypothyroidism. N Engl J Med. 1982; 306(14):849–52.

11. Codaccioni JL, Orgiazzi J, Blanc P, Pugeat M, Roulier R, Carayon P. Lasting remissions in patients treated for Graves' hyperthyroidism with propranolol alone: a pattern of spontaneous evolution of the disease. J Clin Endocrinol Metab. 1988; 67(4):656–62.

12. Nagai Y, Toya T, Fukuoka K, Tanaka N, Yanagi S, Kobayashi K. Occurrence and spontaneous remission of Graves' hyperthyroidism preceded by painless thyroiditis. Endocr J. 1997; 44(6):881–5.

13. Noh J, Yoshimoto M, Inhikawa N, Momotani N, Hanada N, Morii H, et al. Occurrence of Graves' hyperthyroidism preceded by silent thyroiditis. Nagataki S, Torizuka K, editors. editors.The thyroid 1988. Amsterdam: Elsevier Science Publishers B.V. (Biochemical Devision);1988. p. 249–52.
14. Stagnaro-Green A, Abalovich M, Alexander E, Azizi F, Mestman J, Negro R, et al. Guidelines of the American Thyroid Association for the diagnosis and management of thyroid disease during pregnancy and postpartum. Thyroid. 2011; 21(10):): 1081–125.

15. Vos XG, Smit N, Endert E, Tijssen JG, Wiersinga WM. Frequency and characteristics of TBII-seronegative patients in a population with untreated Graves' hyperthyroidism: a prospective study. Clin Endocrinol (Oxf). 2008; 69(2):311–7.

16. Tamaki H, Amino N, Aozasa M, Mori M, Tanizawa O, Miyai K. Serial changes in thyroid-stimulating antibody and thyrotropin binding inhibitor immunoglobulin at the time of postpartum occurrence of thyrotoxicosis in Graves' disease. J Clin Endocrinol Metab. 1987; 65(2):324–30.

Fig. 1.
Anterior view images of a Tc-99m pertechnetate scan in thyrotoxic phase. (A) Postpartum thyroiditis, (B) The first episode of hyperthyroidism, (C) The second episode of hyperthyroidism, (D) A relapse of postpartum thyroiditis. The normal range of thyroid trapping index in our laboratory is 2.5–6.0. In this case, they were 1.2 (A), 11.3 (B), 3.4 (C), and 1.1 (D).
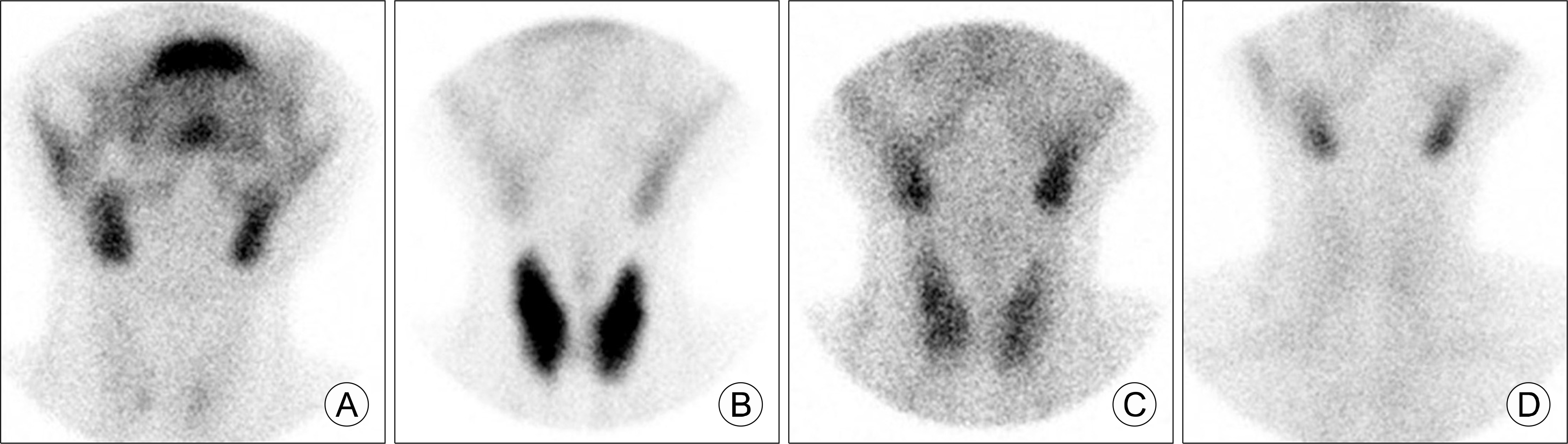
Fig. 2.
Serum free T4 change during pregnancy and after delivery in our patient with Hashimoto's thyroiditis. The dosages of L-T4 (levothyroxine) are shown at the top.
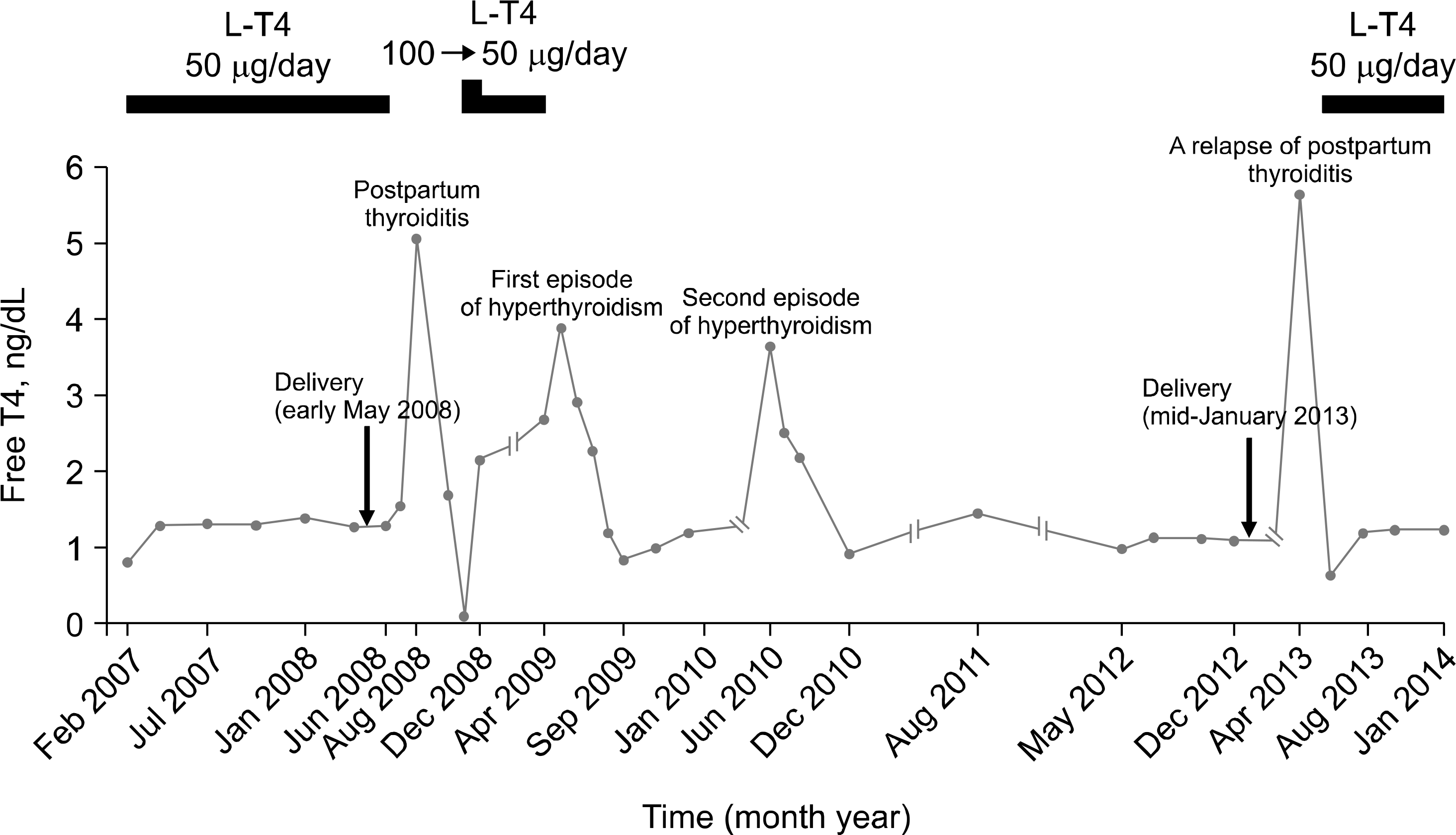
Table 1.
Temporal profile of thyroid function tests and levothyroxine treatment for a patient with recurrent hyperthyroidism following postpartum thyroiditis
Table 2.
Summary of literature describing cases of Graves' disease following postpartum thyroiditis
| Case number (reference) | Age | Family history of thyroid disease | Personal history of pre-pregnancy | PPT (months after delivery) | GD (months after delivery) | Anti-thyroid treatment | Final thyroid outcome |
|---|---|---|---|---|---|---|---|
| 1 (4) | 23 | No | No | 3 | 9 | PTU → Methimazole | Hypothyroidism due to Hashimoto' s thyroiditis |
| 2 (5) | 36 | No | PPT (at age 30) | 4 | 48‡ | Methimazole | N/A |
| 3 (5) | 17 | Yes∗ | Latent lupus | 1 | 5 | No | N/A |
| 4 (6) | 47 | No | No | 4 | 25 | PTU & 131 I | Post-131 I hypothyroidism |
| 5 (7) | 30 | No | No | 5 | 28 | Methimazole | N/A |
| 6 (8) | 31 | Yes† | PPT (3 years ago) | 2.5 | 10.5 | PTU | Euthyroid state |
| 7 (our case) | 29 | No | Hashimoto' s thyroiditis | 3 | 12 (first)25 (second) | No | L-T4 treatment |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download