Abstract
Purpose
For periodontal tissue engineering, it is a primary requisite and a challenge to select the optimum types of cells, properties of scaffold, and growth factor combination to reconstruct a specific tissue in its natural form and with the appropriate function. Owing to fundamental disadvantages associated with using a two-dimensional substrate, several methods of seeding cells into three-dimensional scaffolds have been reported and the authors have asserted its usefulness and effectiveness. In this study, we explore the cell attachment of periodontal ligament fibroblasts on nanohydroxyapatite (n-HA) scaffold using avidin biotin binding system (ABBS).
Methods
Human periodontal ligament fibroblasts were isolated from the health tooth extracted for the purpose of orthodontic procedure. HA nanoparticles were prepared and Ca(NO3)2-4H2O and (OC2H5)3P were selected as precursors of HA sol. The final scaffold was 8 mm in diameter and 3 mm in height disk with porosity value of 81.55%. 1×105 periodontal ligament fibroblasts were applied to each scaffold. The cells were seeded into scaffolds by static, agitating and ABBS seeding method.
Results
The number of periodontal ligament fibroblasts attached was greater for ABBS seeding method than for static or agitating method (P<0.05). No meaningful difference has been observed among seeding methods with scanning electron microscopy images. However, increased strength of cell attachment of ABBS could be deduced from the high affinity between avidin and biotin (Kd=10-15 M).
Once tissue destruction has occurred, one of the major goals of periodontal treatment is to regenerate the affected tissue, reproducing the original architecture and function. However, although many techniques have been advocated for achieving true regeneration, clinical results are often unpredictable, and functional restoration of periodontal tissues remains a challenge. Recent progress in the understanding of the molecular processes associated with tissue repair and regeneration has led to the development of periodontal tissue engineering as an improved method for achieving periodontal regeneration. Periodontal tissue engineering involves the application of the principles of molecular and cell biology, developmental biology, and tissue engineering [1].
Tissue engineering is understanding the principles of tissue growth, and applying this to produce functional replacement tissue for clinical use which requires external regenerative resources that include cells, scaffolds, and growth factors, either in combination or singly; these are known as the triad of tissue engineering [2]. Selection of the optimum types of cell, the scaffold properties, and the growth factor combination are fundamental requisite to reconstruct lost tissue close to native one.
The periodontal ligament is composed of a complex vascular and highly cellular connective tissue [3]. Previous studies have demonstrated the importance of the periodontal ligament in periodontal regeneration [4,5]. They suggest that a new attachment can be achieved using cells originating from the periodontal ligament. More recently, some studies have examined the use of periodontal ligament cells in the field of periodontal regeneration. Seo et al. [6] concluded that human periodontal ligament stem cells can be induced to form mineralized deposits in vitro, and form organized, functional tissues following implantation in vivo.
In the body, nearly all tissue cells reside in an extracellular matrix (ECM) comprising a complex three-dimensional network. However, we commonly rely on two-dimensional cell-culture systems that do not accurately replicate the structure, function, or physiology of living tissues. In the field of tissue engineering, three-dimensionally reconstructed scaffolds play a role similar to that of the ECM in natural tissues, supporting cell attachment, proliferation, differentiation, and migration [7].
It has been established that scaffolds require the following properties [2]: 1) a three-dimensional environment similar to the in vivo condition, 2) an ability to activate cell function, 3) biodegradability, and 4) sufficient mechanical strength for a clinical application. The ECMs of hard tissue are composed of organic and inorganic phases, with the inorganic phase consisting mainly of nanohydroxyapatite (n-HA) crystals, and the organic phase consisting primarily of type I collagen and a small amount of ground substance [8]. Thus, n-HA scaffolds have been developed for use in bone-tissue engineering [9].
The majority of cell-culture studies have been performed on two-dimensional surfaces such as microwell plates and petri dishes because of their convenience and the established high cell viability of two-dimensional cultures. Although these conventional cell-culture systems have notably improved the understanding of basic cell biology, in the field of tissue engineering, there are fundamental disadvantages associated with using a two-dimensional substrate.
Several methods of seeding cells into three-dimensional scaffolds have been reported, such as dynamic perfusion seeding [10], centrifugal seeding [11], agitation seeding [12], and rotating seeding [13]. These authors have asserted the usefulness and effectiveness of each of these methods. Furthermore, several strategies using chemical bonding strength have been applied to improve the affinity between cells and the scaffold surfaces, the most favored being the conjugation of ECM adhesion proteins to synthetic surfaces [14]. Alternatively, precoating the scaffold surface with antibodies against cell membrane antigens can improve cell adhesion to biomaterials [15].
Another approach involves the application of a pair of molecules that bind to each other [16], wherein one molecule is bound to the cell membrane, and the other to the scaffold surface. A good example of a binding molecule pair is avidin and biotin. Avidin is a glycoprotein found in egg white that can strongly bind biotin, which is known as a small-molecule vitamin. The avidin-biotin binding system (ABBS) is distinguishable by the extraordinarily high affinity of avidin for biotin, and it has been applied extensively in the field of biotechnology [16].
In the present study we evaluated the ability of periodontal ligament fibroblasts to attach to three-dimensionally constructed n-HA scaffolds using various cell-seeding methods, thus enabling exploration of the cell attachments associated with the ABBS.
Human periodontal ligament fibroblasts were isolated from healthy teeth extracted for orthodontic reasons. After washing with α-minimum essential medium (α-MEM, Gibco, Grand Island, NY, USA) containing 100 U/mL penicillin and 100 µg/mL streptomycin (Gibco), periodontal ligament tissue was collected by scraping the root surface from the middle one-third to the apex. The collected tissues were attached to the bottom of T75 cell culture flasks with α-MEM containing 15% fetal calf serum (Gibco), 100 mol/L ascorbic acid 2-phosphate (Gibco), 2 mmol/L L-glutamine (Gibco), 100 U/mL penicillin, and 100 µg/mL streptomycin. The flask was initially kept upright and incubated at 37℃ in 5% CO2 for 4 hours, and then laid on its side after confirming the rigid attachment of tissue onto the floor of the flask. The culture medium was exchanged every 3 to 4 days, and when the dishes became subconfluent, the periodontal ligament fibroblasts were detached by applying 0.25% trypsin-ethylenediaminetetraacetic acid (EDTA) solution for 3 minutes at 37℃. Third-passage periodontal ligament fibroblasts were used in this experiment.
This study was approved by the Ethics Committee of Clinical Experiments, Yonsei University Dental Hospital (IRB number 2-2008-0014).
A simple porous scaffold was produced according to published methods [17]. Hydroxyapatite nanoparticles were prepared using the sol-gel process. Ca(NO3)2-4H2O (99%; Sigma-Aldrich Co., St. Louis, MO, USA) and (OC2H5)3P (97%; Sigma-Aldrich Co.) were selected as precursors of the HA sol. The Ca precursor was dissolved in methyl alcohol at a stoichiometric Ca:P ratio of 1.67. The solution was then dehydrated at 180℃ after solvent evaporation and was refluxed in methyl alcohol in an atmosphere of Ar. The P precursor was prehydrolyzed for 10 hours; HCl and H2O were added as catalysts before the reaction with the Ca precursor. The hydroxyapatite sol was prepared by the reaction of the two precursors, and then dried at 950℃. The finished scaffold was an 8-mm-diameter and 3-mm-high disk with 81.55% porosity, 31.25 mm2/mm3 of specific surface, 140.14 mm of strut thickness, and 523.12 mm of strut spacing. Scanning electron microscopy (SEM) (S-3000N, Hitachi, Ibaraki, Japan) was used to examine the microstructure of the n-HA scaffolds.
Cells were biotinylated with a commercially available reagent, EZ-Link Sulfo-NHS-LC-Biotin (Pierce Chemical Co., Rockford, IL, USA) according to the manufacturer's instructions. After detachment from the culture dishes with 0.25% trypsin-EDTA, the suspension of periodontal ligament fibroblasts was collected in a 50-mL tube by centrifugation for 5 minutes at 1,500 rpm (260 g). A biotinylation reagent was applied to the cell suspension for 30 minutes at room temperature. The cells were then washed with 100 mM glycine in phosphate-buffered saline (PBS) and used for subsequent assays.
Eight n-HA scaffolds were prepared for each of the seeding procedures. The n-HA scaffolds were prepared as follows for the static and agitating cell-seeding procedures. Each scaffold was placed into a cryotube. Prior to cell seeding, the scaffolds were pre-wet with culture medium for 30 minutes, and then the cells were loaded onto them. The procedure was the same for cells seeded using the ABBS, except that prior to their use, the n-HA scaffolds were preabsorbed with avidin (Sigma-Aldrich Co.) for 2 hours, and then the modified surface was observed using SEM. In the case of the static and ABBS cell-seeding procedures, cells were incubated with their respective n-HA scaffolds (i.e., unmodified and avidin-modified) for 6 hours at room temperature. For the agitating cell-seeding procedure, the cells were incubated on an orbital shaker at room temperature for 6 hours, and for rotating cell seeding, the culture plate was set down onto an orbital shaker and agitated at 100 rpm for 6 hours. Regardless of the seeding method, after incubation of the scaffolds with the cells, any unattached cells were removed by flushing with PBS. The degree of cell attachment was determined by WST-1 assay. The periodontal ligament fibroblasts attached to the disks were observed using SEM.
The cell proliferation reagent WST-1 was used to assess the degree of attachment of live cells on the n-HA scaffolds. For evaluation of viable cells, the cell-seeded n-HA scaffolds were placed into 48-well microtiter plates, each well containing 1,000 µL of culture medium and 100 µL of WST-1. The reactants were then treated with the test articles at 37℃ in a humidified 5% CO2/95% air incubator for 4 hours. The absorbance of the samples relative to a background control (medium alone) as a blank was measured at 480 nm using a microtiter plate (ELISA) reader.
Statistical assessment of significant variations was performed using SPSS software (SPSS Inc., Chicago, IL, USA). Mean and standard deviation (SD) values were calculated for each of the three groups. The significance of any differences was determined using the Kruskal-Wallis test, and the level of statistical significance was set at P<0.05.
Fig. 1 shows the WST-1 results. The WST-1 value, which is a ratio of attached living cells, did not differ significantly between the agitating cell-seeding method (2.24±0.16, mean±SD) and the static cell-seeding method (2.05±0.11, mean±SD), but it did differ significantly between the ABBS (3.16±0.19, mean±SD) and both the static and agitating cell-seeding methods (P<0.05).
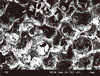
Typical examples of SEM images are shown in Figs. 2-4. Smooth surfaces with some elevations and depressions were observed on the scaffolds. There were no significant differences between the appearances of the n-HA and the avidin-modified n-HA scaffold (Figs. 2 and 3). They both appear to be macroporous and have a high degree of interconnectivity. Furthermore, the carbon-bead-derived micropores are visible.
Fig. 4 shows the SEM morphology of periodontal ligament fibroblasts adhered to scaffolds after static cell seeding (Fig. 4A), agitating cell seeding (Fig. 4B), and ABBS cell seeding (Fig. 4C and D). In the statically seeded group, it appears as if previously attached cells have become detached from the scaffold. No meaningful difference was observed between the morphology of these cells after agitating and ABBS seeding.
Efficient cell attachment is an important first step for tissue reconstruction using appropriated organ-derived cells and three-dimensionally constructed biodegradable scaffolds. This study has demonstrated the enhanced attachment of periodontal ligament-derived fibroblasts to highly porous three-dimensional n-HA surfaces using an ABBS.
Several scaffold parameters play a significant role in tissue-engineering applications. We designed a porous n-HA scaffold with a porosity value of 81.55%. Compared to the background controls, WST-1 results confirm the presence of cells on all n-HA scaffolds, suggesting successful loading of the cells and indicating that the n-HA has good cytocompatibility with periodontal ligament fibroblasts.
While the initial loading was almost equal for the three test groups, the rate of cell attachment varied among the tests. The ABBS demonstrated WST-1 values superior to the other two methods, implying a more effective cell attachment with that method. The effectiveness of ABBS for cell attachment has been demonstrated previously [16]. We also found that conjugation of the biotin molecule to periodontal ligament fibroblast cell membranes enhanced the cell attachment to the avidin-modified n-HA scaffold.
Various seeding methods have been tested to investigate cell attachment to various scaffolds; we used the agitating technique as a control [12]. Takahashi and Tabata [12] argued that the agitating cell-seeding method was an improvement over the traditional cell-seeding protocol for attaching cells to scaffolds. However, although the results in the present study showed that the agitating method did induce cell attachment, the differences between the traditional method and agitating method were not statistically significant. The difference between the findings of our study and that of Takahashi and Tabata may be attributable to the cells and scaffolds used and differences in seeding conditions, such as the volume of cell suspension used, the rotating speed, and the culture time.
The dissociation constant for the ABBS (Kd=10-15 M) [18] is several times smaller than the typical dissociation constant of other biospecific cell-attachment reactions, such as between integrin and fibronectin (Kd=10-6 M) [19] and between integrin and laminin (Kd=10-9 M) [20]. In spite of a lack of a meaningful distinction among the SEM images, a higher strength of cell attachment associated with the ABBS could be deduced from the high affinity between avidin and biotin. Furthermore, this high affinity would increase the initial cell adhesion and strength of the cell attachment in conditions under which the cell-to-surface contact times are much shorter than when using static conditions or when a limited number of cells is prepared.
In particular, it is likely that biotinylation can denature cell-surface receptor molecules for hormones or cytokines. However, Kojima et al. [21] found that biotinylation of cells did not severely inhibit cell proliferation and functions or signal transfer via transmembrane receptors. Therefore, we can assume that the application of periodontal ligament fibroblasts on n-HA scaffolds via the ABBS potentially induced the formation of cementum or periodontal ligament-like structure. However, further studies are required to evaluate this assumption.
Previous studies have shown that periodontal ligament cells can be transplanted into periodontal defects without any adverse immunologic or inflammatory consequences [22]. In general, for proper periodontal regeneration, implanted cells should be positioned correctly via chemotactic events. The high affinity of avidin for biotin may inhibit this chemotactic event. This limitation should be borne in mind when carrying out the research required to further develop this method for in vivo applications.
In conclusion, a high-affinity ABBS enhances the ability of periodontal ligament fibroblasts to attach to three-dimensionally constructed n-HA scaffolds. We have demonstrated herein the applicability of the ABBS to periodontal tissue engineering.
Figures and Tables
ACKNOWLEDGEMENTS
This study was supported by a grant of the Korea Healthcare technology R&D Project, Ministry of Health & Welfare, Republic of Korea. (A101578).
References
1. Bartold PM, McCulloch CA, Narayanan AS, Pitaru S. Tissue engineering: a new paradigm for periodontal regeneration based on molecular and cell biology. Periodontol 2000. 2000. 24:253–269.

2. Lynch SE, Marx RE, Nevins M, Wisner-Lynch LA. Tissue engineering: applications in maxillofacial surgery and periodontics. 2008. 2nd ed. Chicago: Quintessence.
3. McCulloch CA, Lekic P, McKee MD. Role of physical forces in regulating the form and function of the periodontal ligament. Periodontol 2000. 2000. 24:56–72.

4. Nyman S, Gottlow J, Karring T, Lindhe J. The regenerative potential of the periodontal ligament. An experimental study in the monkey. J Clin Periodontol. 1982. 9:257–265.

5. Nyman S, Lindhe J, Karring T, Rylander H. New attachment following surgical treatment of human periodontal disease. J Clin Periodontol. 1982. 9:290–296.

6. Seo BM, Miura M, Gronthos S, Bartold PM, Batouli S, Brahim J, et al. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet. 2004. 364:149–155.

7. Lee J, Cuddihy MJ, Kotov NA. Three-dimensional cell culture matrices: state of the art. Tissue Eng Part B Rev. 2008. 14:61–86.

8. Zhao F, Yin Y, Lu WW, Leong JC, Zhang W, Zhang J, et al. Preparation and histological evaluation of biomimetic three-dimensional hydroxyapatite/chitosan-gelatin network composite scaffolds. Biomaterials. 2002. 23:3227–3234.

9. Wang H, Li Y, Zuo Y, Li J, Ma S, Cheng L. Biocompatibility and osteogenesis of biomimetic nano-hydroxyapatite/polyamide composite scaffolds for bone tissue engineering. Biomaterials. 2007. 28:3338–3348.

10. Kim SS, Sundback CA, Kaihara S, Benvenuto MS, Kim BS, Mooney DJ, et al. Dynamic seeding and in vitro culture of hepatocytes in a flow perfusion system. Tissue Eng. 2000. 6:39–44.

11. Yang TH, Miyoshi H, Ohshima N. Novel cell immobilization method utilizing centrifugal force to achieve high-density hepatocyte culture in porous scaffold. J Biomed Mater Res. 2001. 55:379–386.

12. Takahashi Y, Tabata Y. Homogeneous seeding of mesenchymal stem cells into nonwoven fabric for tissue engineering. Tissue Eng. 2003. 9:931–938.

13. Lueders C, Sodian R, Shakibaei M, Hetzer R. Short-term culture of human neonatal myofibroblasts seeded using a novel three-dimensional rotary seeding device. ASAIO J. 2006. 52:310–314.

14. Balcells M, Edelman ER. Effect of pre-adsorbed proteins on attachment, proliferation, and function of endothelial cells. J Cell Physiol. 2002. 191:155–161.

15. Dekker A, Poot AA, van Mourik JA, Workel MP, Beugeling T, Bantjes A, et al. Improved adhesion and proliferation of human endothelial cells on polyethylene precoated with monoclonal antibodies directed against cell membrane antigens and extracellular matrix proteins. Thromb Haemost. 1991. 66:715–724.

16. Tsai WB, Wang MC. Effects of an avidin-biotin binding system on chondrocyte adhesion and growth on biodegradable polymers. Macromol Biosci. 2005. 5:214–221.

17. Saggiowoyansky J, Scott CE, Minnear WP. Processing of porous ceramics. Am ceram soc bull. 1992. 71:1674–1682.
19. Akiyama SK, Yamada KM. The interaction of plasma fibronectin with fibroblastic cells in suspension. J Biol Chem. 1985. 260:4492–4500.

20. Terranova VP, Rao CN, Kalebic T, Margulies IM, Liotta LA. Laminin receptor on human breast carcinoma cells. Proc Natl Acad Sci U S A. 1983. 80:444–448.





 PDF
PDF ePub
ePub Citation
Citation Print
Print






 XML Download
XML Download