Abstract
A case of a female patient with local and systemic complications of metallosis, following catastrophic wear of a revised hip arthroplasty, is presented. The patient had a history of a fractured ceramic-on-ceramic implant, exchanged with a metal-on-polyethylene prosthesis. Systemic complications included sarcoidosis-like reactions, presenting as granulomatous lung disease, along with chorioretinitis, erythema nodosum, and cardiomyopathy. High local and circulating cobalt and chromium levels established the diagnosis. The patient underwent extensive debridement and implant revision. One year postoperatively, she had no respiratory symptoms or functional impairment. Local and systemic complications of metallosis after hip arthroplasty should be promptly recognized and treated operatively.
One of the catastrophic complications of total hip arthroplasty (THA) is wear of articulating surfaces, leading to local metallosis. Metallosis has been reported with some designs of metal-on-metal articulations or after the exchange of fractured ceramic bearings with a metal head and a polyethylene acetabular liner1). Metal ions and nanoparticles can disseminate through blood and lymphatic vessels and cause systemic complications12). We present a case of pulmonary granulomatous disease secondary to revision of a fractured ceramic THA head.
In 2005, an otherwise healthy 55 year-old female patient, with a primary diagnosis of hip osteoarthritis and otherwise unremarkable history, underwent bilateral staged THA at another institution. A cementless system with a ceramic-on-ceramic articulation was used on both occasions (R3 acetabulum, Synergy femoral stem; Smith & Nephew, Memphis, TN, USA). Commencing early postoperatively, the patient had recurrent dislocation of her right arthroplasty (Fig. 1A) as often as 3-4 times a year, which she managed to reduce on her own. Six years postoperatively, the patient sustained a fracture of the right ceramic head (Fig. 1B). The acetabular prosthesis was exchanged with a tantalum shell and an ultra-high molecular weight polyethylene liner. The existing stem was well fixed and therefore was left in situ. The fractured ceramic head was exchanged with one made of cobalt-chromium alloy (Continuum acetabulum, Versys head; Zimmer, Warsaw, IN, USA) (Fig. 1C).
In the following months, the patient gradually developed severe activity related right hip pain. Six months post-revision, the patient developed erythema nodosum of the left leg and bilateral chorioretinitis and papilledema. A chest computed tomography (CT) revealed diffuse radiopaque nodules in both lungs and enlargement of the right hilum (Fig. 2). Biopsy of the right lung showed fibrosis and extensive inflammation with multiple non-necrotizing granulomata, consisting of histiocytes, multinucleated giant cells, and lymphocytes (Fig. 3). Special stains for mycobacteria and fungi were negative and no atypical cells were found. A diagnosis of sarcoidosis was made. The patient received corticosteroids and azathioprine for six months, which led to clinical and radiological improvement. Between 2013 and 2015, echocardiograms and chest magnetic resonance imaging showed severe cardiomyopathy with the ejection fraction decreasing from 70% to 35% and evidence of non-ischemic type fibrosis of the lower and posterior ventricular wall, as well as moderate dilation of the left ventricle.
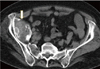
The patient sought advice from our team in February 2015. Physical examination revealed a 76 kg, 159 cm tall woman, with a severely antalgic gait and painful, decreased range of motion of the right hip. Radiographic imaging showed severe osteolysis of the proximal femur (Fig. 1D). A CT of pelvis revealed an iliac pseudotumor (Fig. 4). The presence of a pseudotumor, extensive osteolysis, and the history of a fractured ceramic implant exchanged with a metal alloy head and a polyethylene liner, aroused suspicion for metallosis. A hip aspiration produced black fluid with high concentrations of chromium and cobalt, 25,400 µg/L and 26,500 µg/L respectively (Fig. 5). Whole blood chromium and cobalt concentrations were also significantly elevated, 31.8 µg/L and 22.2 µg/L respectively.
It was therefore decided to proceed with a second revision operation. Intraoperatively, gross local metallosis was found and large volumes of black fluid poured out from a trochanteric pseudotumor. There was extensive osteolysis of the proximal femur but the stem remained fixed (Fig. 6A). Multiple ceramic fragments were still present in the periprosthetic tissues. Ceramic debris was also embedded in the articular surface of the polyethylene liner (Fig. 6B). The metallic head was distorted due to massive wear (Fig. 6C). The Morse taper of the stem was also damaged, allowing free rotation of the prosthetic head (Fig. 6D).
Meticulous soft tissue debridement was carried out. The acetabular shell, which remained well fixed, was left in place but the worn polyethylene liner was removed. The femoral stem was exchanged with a long revision implant (Wagner SL revision uncemented stem; Zimmer GmbH, Winterthur, Switzerland). To prevent recurrence of the problem, a ceramic-on-ceramic articulation was inserted (Biolox Delta; Zimmer GmbH) (Fig. 1E).
Recovery was uneventful. At three months, blood chromium and cobalt levels had returned to normal, 1.17 µg/L and 0.19 µg/L respectively. One year later, the patient was able to mobilize painlessly, using one crutch. She had no respiratory symptoms and her pulmonary function tests were normal. Her heart function had stabilized to an ejection fraction of 50%.
To our knowledge, this is the first report, associating granulomatous lung disease with wear of metal implants, following the revision of a failed ceramic-onceramic THA. Granulomatous lung disease has been attributed to exposure to a variety of environmental and occupational agents, including mycobacteria, fungi, pollen, building materials and metallic dust and fumes34). Sarcoidosis is a diagnosis of exclusion, suggesting an unrecognized causal factor, causing non-necrotic, non-infectious granulomas that are clinically and histologically indistinguishable from secondary granulomas3). Since initially no causative agent had been identified, the overall presentation of the patient had been characterized as sarcoidosis. The identification of the hip arthroplasty as a source of heavy metal ions and microparticles offered a reasonable specific etiology.
Inhalation of metallic fumes and dust has been associated with pulmonary fibrosis and giant cell infiltration of pulmonary interstitial tissue4). Metal nanoparticles are internalized by distant tissue cells, inducing chromosomal damage and oxidative stress2). Macrophage and monocyte activation, following exposure to metal nanoparticles and ions, has been proven in vitro5). This event could be the first step in a cascade leading to granuloma formation6). It can be expected that similar tissue reactions occur with hematogenous spread of heavy metal particles. Péoc’h et al.7) have reported granulomatous lesions in the spleen and liver after a metal-on-polythylene THA, with histological evidence of titanium and polyethylene infiltration of macrophages.
In young patients, ceramic implants are considered more appropriate than the usual, metal-on-polyethylene bearings, because they produce less wear debris, allowing for increased longevity of the reconstruction. However, ceramic materials are associated with an up to 1.5% risk of fracture8). Revision of a fractured ceramic implant, using a metal head and a polyethylene acetabular liner, may result in catastrophic wear of the new head1). Since complete removal of the ceramic debris from the periprosthetic tissues is impossible, embedding of remaining debris in the polyethylene liner acts as sandpaper, damaging the relatively softer new metallic head and producing metal nanoparticles and ions, causing local metallosis and systemic toxicity.
Gessner et al.9) identified twenty five cases of systemic toxicity following metallosis from chrome/cobalt hip arthroplasty, with thyroid dysfunction, psychosocial manifestations, sensory and motor neuropathy, visual and auditory disturbances, or cardiomyopathy. However, since five of these cases have been identified by the same team in Alaska only, it is probable that systemic complications following metallosis from hip replacement are underreported9). It is therefore important, for both physicians and orthopedic surgeons, to suspect metallosis in patients who present with systemic disorders and have a history of a fractured ceramic bearing exchanged with a metal-on-polyethylene combination. Diagnostic difficulties arise with elderly patients, who, due to a lower activity level, have only mild symptoms from a potentially malfunctioning prosthetic joint, and whose medical problems can be easily attributed to comorbidities or medications. Guidelines, such as those of the UK Medicines and Health-related Products Regulatory Agency10), imposing frequent monitoring of blood ion levels and repeated imaging in arthroplasties with high risk for metal release, can lead to earlier diagnosis. With prompt synovectomy, lavage and revision to a ceramic-on-ceramic bearing, systemic complications of metallosis can be avoided.
Figures and Tables
Fig. 1
(A) Posterior dislocation of the original arthroplasty. (B) Multiple episodes of dislocation ultimately led to a fracture of the ceramic head. (C) At the first revision, a metal head and a new metal shell with a polyethylene liner were inserted. (D) Absorption of the greater trochanter occurred within three years. (E) A second revision with a long stem and ceramic bearings was undertaken (radiograph at 12 months postoperatively).
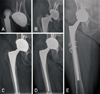
Fig. 3
Resection biopsy from the upper right lung revealed extensive, non-necrotizing inflammation, with multiple granulomata (white arrows), consisting of histiocytes, lymphocytes and multinucleated giant cells (black arrows), arranged around vessels and bronchial walls (H&E stain, ×100).
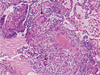
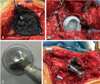
Fig. 6
(A) At second revision, metallosis in the periprosthetic region was evident, necessitating generous debridement. (B) Ceramic debris was found embedded in the polyethylene liner, causing wear of the metallic head. (C) Excessive wear resulted in loss of the spherical shape of the prosthetic femoral head. (D) The Morse taper of the femoral prosthesis appeared scratched and distorted.
References
1. Oldenburg M, Wegner R, Baur X. Severe cobalt intoxication due to prosthesis wear in repeated total hip arthroplasty. J Arthroplasty. 2009; 24:825.e15–825.e20.

2. Keegan GM, Learmonth ID, Case CP. Orthopaedic metals and their potential toxicity in the arthroplasty patient: A review of current knowledge and future strategies. J Bone Joint Surg Br. 2007; 89:567–573.
5. Newman KL, Newman LS. Occupational causes of sarcoidosis. Curr Opin Allergy Clin Immunol. 2012; 12:145–150.

6. Caicedo MS, Desai R, McAllister K, Reddy A, Jacobs JJ, Hallab NJ. Soluble and particulate Co-Cr-Mo alloy implant metals activate the inflammasome danger signaling pathway in human macrophages: a novel mechanism for implant debris reactivity. J Orthop Res. 2009; 27:847–854.

7. Péoc’h M, Moulin C, Pasquier B. Systemic granulomatous reaction to a foreign body after hip replacement. N Engl J Med. 1996; 335:133–134.

8. Koo KH, Ha YC, Jung WH, Kim SR, Yoo JJ, Kim HJ. Isolated fracture of the ceramic head after third-generation alumina-on-alumina total hip arthroplasty. J Bone Joint Surg Am. 2008; 90:329–336.

9. Gessner BD, Steck T, Woelber E, Tower SS. A systematic review of systemic cobaltism after wear or corrosion of chrome-cobalt hip implants. J Patient Saf. 2015; 06. 12. Published online June 12, 2015. DOI: 10.2106/JBJS.F.01489.

10. Medicines and Healthcare Products Regulatory Agency. Appendix. Management recommendations for patients with metal-on-metal hip replacement implants [Internet]. Medicines and Healthcare Products Regulatory Agency;2012 Jun 25. Available from: https://assets.digital.cabinet-office.gov.uk/media/5485abf640f0b6024400027d/con155766.pdf.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download