Abstract
High intensity focused ultrasound (HIFU) is a technique that was first introduced in the 1940s as a potential method of destroying selective regions within the brain to aid neurobehavioral studies. A beam of ultrasound can be delivered to a targeted focus at a distance from its source, and if a sufficient amount of energy is concentrated in the focus, the cells lying within this focal volume are selectively killed. This is, therefore, a non-invasive method of producing selective and "trackless" tissue destruction in deep-seated targets in the body without damage to the overlying tissues. Although it had not been in clinical use for a long time, HIFU is now widely used as a non-invasive treatment method for malignant tumors of the liver, kidney, breast, bone, uterus and pancreas, as well as for the relief of chronic pain of malignant origin. Further improvement of technology and imaging of HIFU in the near future will make it one of the most important tools in the treatment of solid tumors, further expanding its clinical applications.
Figures and Tables
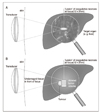 | Figure 1Schematic showing the principle of high-intensity focused ultrasound
A) An extracorporeal source generates an ultrasound beam, which forms a cigar-shaped focus deep within the target tissue(liver). The volume of ablation('lesion') following a single high-intensity focused ultrasound exposure is small and will vary according to transducer characteristics, but is typically in the order of 1~3mm wide by 8~15mm in length along the beam axis.
B) Schematic illustrating application of sequential "single lesions" to achieve tumour volume ablation. The lesions must be placed side by side systematically to "paint out" the target tumour and some of the surrounding normal tissue margin.
|
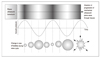 | Figure 2The principle of inertial cavitation
A mechanical ultrasound wave progresses through tissues(top), causing alternating cycles of increased and reduced pressure (compression and rarefaction respectively-middle). Gas is drawn out of solution during rarefaction, creating bubbles. These can oscillate in size in a stable fashion with the chaging tissue pressure, but ultimately might collapse, causing local energy release and temperature rises at the microscopic level (bottom).
|
 | Figure 3A case of high-intensity focused ultrasound for hepatocelluar carcinoma
A) Before HIFU: Abdominal CT shows a large, 9cm-diameter, hepatocelluar carcinoma(arrows) at right hepatic lobe in 67-year-old man.
B) After HIFU: MR obtained 2 months later shows the lesion(arrows) has markedly decreased without contrast enhancement.
|
References
1. Vogl TJ, Straub R, Eichler K, Soller O, Mack MG. Colorectal carcinoma metastases in liver: laser-induced interstitial thermotherapy : local tumor control rate and survival data. Radiology. 2003. 230:450–458.

2. Nagaoka Y, Nakayama R, Iwata M. Cutaneous seeding following percutaneous ethanol injection therapy for hepatocellular carcinoma. Intern Med. 2004. 43:268–269.

3. Liu C, Frilling A, Dereskewitz C, Broelsch CE. Tumor seeding after fine needle aspiration biopsy and percutaneous radiofrequency thermal ablation of hepatocellular carcinoma. Dig Surg. 2003. 20:460–463.

4. Thuroff S, Chaussy C, Vallancien G, Wieland W, Kiel HJ, Gelet A, et al. High-intensity focused ultrasound and localized prostate cancer : efficacy results from the European multicentric study. J Endourol. 2003. 17:673–677.
5. Kennedy JE, Wu F, ter Haar GR, Gleeson FV, Phillips RR, Middleton MR, et al. High-intensity focused ultrasound for the treatment of liver tumours. Ultrasonics. 2004. 42:931–935.

6. Wu F, Wang ZB, Chen WZ, Zou JZ, Bai J, Gao GW, et al. Extracorporeal focused ultrasound surgery for treatment of human solid carcinomas: early Chinese clinical experience. Ultrasound Med Biol. 2004. 30:245–250.

7. Gianfelice D, Kheat A, Boulanger Y, Amara M, Belblidia A. Feasibility of magnetic resonance imaging-guided focused ultrasound surgery as an adjunct to tamoxifen therapy in high-risk surgical patients with breast carcinoma. J Vasc Interv Radiol. 2003. 14:1275–1282.

8. Wu F, Wang ZB, Chen WZ, Bai J, Zhu H, Qiao TY. Preliminary experience using high intensity focused ultrasound for the treatment of patients with advanced stage renal malignancy. J Urol. 2003. 170:2237–2240.

9. Chen L, Rivens L, ter Haar G, Riddler S, Hill CR, Bensted JP. Histological changes in rat liver tumours treated with high-intensity focused ultrasound. Ultrasound Med Biol. 1993. 19:67–74.

10. ter Haar G, Clarke RL, Vaughan MG, Hill CR. Trackless surgery using focused ultrasound: technique and case report. Minimally invasive Therapy. 1991. 1:13–19.

11. Hill CR, ter Haar GR. High intensity focused ultrasound: potential for cancer treatment. Br J Radiol. 1995. 68:1296–1303.
12. Wu F, Chen WZ, Bai j, Zou JZ, Wang ZL, Wang ZB, et al. Pathological changes in human malignant carcinoma treated with high-intensity focused ultrasound. Ultrasound Med Biol. 2001. 27:1099–1106.

13. Wu F, Wang ZB, Jin CB, Zhang JP, Chen WZ, Zou JZ, et al. Circulating tumor cells in patients with solid malignancy treated by high-intensity focused ultrasound. Ultrasound Med Biol. 2004. 30:511–517.

14. Vallejo R, Hord ED, Barna SA, Santiago-Palma J, Ahmed S. Perioperative immunosuppression in cancer patients. J Environ Pathol Toxicol Oncol. 2003. 22:139–146.

15. Mafune K, Tanaka Y. Influence of multimodality therapy on the cellular immunity of patients with esophageal cancer. Ann Surg Oncol. 2000. 7:609–616.

16. Kramer G, Steiner GE, Grobl M, Hrachowitz K, Reithmayr F, Marberger M, et al. Response to sublethal heat treatment of prostatic tumor cells and of prostatic tumor infiltrating T-cells. Prostate. 2004. 58:109–120.

17. Visioli AG, Rivens IH, ter Haar GR, Horwich A, Huddart RA, Glees J, et al. Preliminary results of a phase I dose escalation clinical trial using focused ultrasound in the treatment of localized tumours. Eur J Ultrasound. 1999. 9:11–18.

18. Vallancien G, Harouni M, Guillonneau B, Veillon B, Bougaran J. Ablation of superficial bladder tumors with focused extracorporeal pyrotherapy. Urology. 1996. 47:204–207.

19. Kennedy JE. High-intensity focused ultrasound in the treatment of solid tumors. Cancer. 2005. 5:321–327.
20. Fry WJ, Mosberg WH, Bamard JW, Fry FJ. Production of focal destructive lesions in the central nervous system with ultrasound. J Neurosurg. 1954. 11:471–478.

21. Blana A, Walter B, Rogenhofer S, Wieland WF. High-intensity focused ultrasound for the treatment of localized prostate cancer: 5-year experience. Urology. 2004. 63:297–300.

22. Gelet A, Chapelon JY, Poissonnier L, Bouvier R, Rouviere O, Vallancien G, et al. Local recurrence of prostate cancer after external beam radiotherapy: early experience of salvage therapy using high-intensity focused ultrasonography. Urology. 2004. 63:625–629.

23. Stewart EA, Gedroyc WM, Tempany CM, Quade BJ, Inbar Y, Rabinovici J, et al. Focused ultrasound treatment of uterine fibroid tumors: safety and feasibility of a noninvasive thermoablative technique. Am J Obstet Gynecol. 2003. 189:48–54.

24. McDannold N, Moss M, Killiany R, Rosene DL, King RL, Hynynen K, et al. MRI-guided focused ultrasound surgery in the brain: tests in a primate model. Magn Reson Med. 2003. 49:1188–1191.

25. Wu F, Wang ZB, Cao YD, Chen WZ, Bai J, Zhu H, et al. A randomised clinical trial of high-intensity focused ultrasound ablation for the treatment of patients with localised breast cancer. Br J Cancer. 2003. 89:2227–2233.

26. Marbeger M, Schatzl G, Cranston D, Kennedy JE. Extracorporeal ablation of renal tumors with high intensity focused ultrasound. Br J Urol. 2005. 95:Suppl 2. 52–55.
27. Wu F, Wang JB, Chen WZ, Zou JZ, Bai J, Su HB, et al. High intensity focused ultrasound ablation combined with transcatheter arterial embolisation in the treatment of advanced hepatocellular carcinoma. Radiology. 2005. 235:659–667.

28. Bohris C, Jenne JW, Rastert R, Simiantonakis I, Brix G, Debus J, et al. MR monitoring of focused ultrasound surgery in a breast tissue model in vivo. Magn Reson Imaging. 2001. 19:167–175.

29. Sedelaar JP, Aarnink RG, van Leenders GJ, Beerlage HP, Debruyne FM, de La Rosette JJ, et al. The application of three-dimensional contrast-enhanced ultrasound to measure volume of affected tissue after HIFU treatment for localized prostate cancer. Eur Urol. 2000. 37:559–568.

30. Anderson GS, Brinkmann F, Soulen MC, Alavi A, Zhuang H. FDG positron emission tomography in the surveillance of hepatic tumors treated with radiofrequency ablation. Clin Nucl Med. 2003. 28:192–197.

31. Cannon JW, Stroll JA, Salgo IS, Knowles HB, Howe RD, del Nido PJ, et al. Real-time three-dimensional ultrasound for guiding surgical tasks. Comput Aided Surg. 2003. 8:82–90.





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download