Abstract
Objective
Although nerve-sparing radical surgery (NSRS) is an emerging technique for reducing surgery-related dysfunctions, its efficacy is controversial in patients with cervical cancer. Thus, we performed a meta-analysis to compare clinical outcomes, and urinary, anorectal, and sexual dysfunctions between conventional radical surgery (CRS) and NSRS.
Methods
After searching PubMed, Embase, and the Cochrane Library, two randomized controlled trials, seven prospective and eleven retrospective cohort studies were included with 2,253 patients from January 2000 to February 2014. We performed crude analyses and then conducted subgroup analyses according to study design, quality of study, surgical approach, radicality, and adjustment for potential confounding factors.
Results
Crude analyses showed decreases in blood loss, hospital stay, frequency of intraoperative complications, length of the resected vagina, duration of postoperative catheterization (DPC), urinary frequency, and abnormal sensation in NSRS, whereas there were no significant differences in other clinical parameters and dysfunctions between CRS and NSRS. In subgroup analyses, operative time was longer (standardized difference in means, 0.948; 95% confidence interval [CI], 0.642 to 1.253), while intraoperative complications were less common (odds ratio, 0.147; 95% CI, 0.035 to 0.621) in NSRS. Furthermore, subgroup analyses showed that DPC was shorter, urinary incontinence or frequency, and constipation were less frequent in NSRS without adverse effects on survival and sexual functions.
Various types of conventional radical surgery (CRS), such as radical hysterectomy, radical trachelectomy, and radical parametrectomy, have shown 5-year survival rates of more than 90%, and remain the standard treatment for patients with early-stage cervical cancer [1,2]. However, CRS is known to cause urinary dysfunctions, such as bladder hypotonia, urinary incontinence, and abnormal sensation, in 12% to 85% of patients [3,4,5]. Furthermore, anorectal dysfunctions, including constipation, have been reported in 5% to 10% of patients after CRS [6,7]. Considerable sexual dysfunctions, including decrease in sexual interest and orgasm, and vaginal dryness, are also noticed after CRS, which compromise sexual activity and result in substantial distress [8].
Urinary, anorectal, and sexual dysfunctions are known to be caused by injury to of the pelvic autonomic nerves during CRS. These nerves play a major role for the neurogenic control of urinary and anorectal functions. Moreover, they supply blood vessels of the female genital tract and thereby affect sexual activity by neurogenically controlling its lubrication or swelling response [9]. Thus, nerve-sparing radical surgery (NSRS) has emerged in the last 30 years for reducing surgery-related dysfunctions without compromising oncologic outcomes [10]. However, the efficacy and safety of NSRS are still controversial in comparison with CRS despite a growing number of studies addressing the issue of NSRS. In particular, major limitations are no consensus on which part of the uterine-supporting ligaments the nerve-sparing technique should be directed to, an unresolved concern about whether NSRS may interfere with radicality necessary for treating cervical cancer, and a debate on the discrepancy in prognosis between CRS and NSRS.
Although three prospective randomized controlled trials (RCTs) and one systematic review with a meta-analysis have been published up to now [11,12,13,14], they are not enough to clarify the efficacy and safety of NSRS in cervical cancer due to small numbers of enrolled patients and non-English literature that hinder extraction of relevant data. Thus, this meta-analysis was performed to compare clinical outcomes, and urinary, anorectal, and sexual dysfunctions between CRS and NSRS in patients with early-stage cervical cancer.
This meta-analysis was conducted in line with the recommendations from the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [15]. For this meta-analysis, we searched PubMed, Embase, and the Cochrane Central Register of Controlled Trials (CENTRAL) in the Cochrane Library for relevant studies published from January 2000 to February 2014 inclusive using the following terms: "cervical neoplasm(s)" or "cervical cancer" or "cervical carcinoma," and "nerve sparing."
We included relevant studies that met the following criteria: cervical cancer; comparison of clinical outcomes between CRS and NSRS; and comparison of urinary, anorectal, or sexual dysfunctions between CRS and NSRS. However, review articles, case reports, editorials or letters to the editor, and non-English studies that did not meet the selection criteria were excluded from this meta-analysis.
Two of the authors (HSK and KK) independently evaluated the potential eligibility of all studies retrieved from the database based on the predetermined selection and exclusion criteria, and the third author (CWJ) resolved disagreement between the two authors through consensus conference. A total of 166 studies were identified, and we excluded 27 duplicates and 51 studies, including reviews (n=23), non-English literature (n=13), editorials or letters to the editor (n=9), and case reports (n=6). In addition, we excluded 54 studies due to non-comparative studies (n=41) and non-cervical cancer (n=13). Thirteen studies were also excluded due to data on surgical technique or anatomy only (n=8), and lack of data for comparison (n=5). Finally, two RCTs [12,13], seven prospective cohort [7,9,16,17,18,19,20], and 11 retrospective cohort studies [10,21,22,23,24,25,26,27,28,29,30] were included in this meta-analysis (Fig. 1).
Data extraction was performed by two authors (HSK and SBR), and any discrepancies were addressed by a joint reevaluation of the article with the third author (KSH). The following data were independently extracted from each study for this meta-analysis: the first author; period of enrollment; study design; the International Federation of Gynecology and Obstetrics (FIGO) stage; surgical approach; surgical types such as radical hysterectomy, radical trachelectomy, or radical parametrectomy; radicality of surgery; number of patients with cervical cancer who underwent CRS or NSRS; neoadjuvant chemotherapy (NAC); adjustment for potential confounding factors; clinical outcomes, including operative time (minute), blood loss (mL), hospital stay (day), intraoperative or postoperative complications, length of the resected vagina or parametrium (mm), disease-free survival (DFS), and overall survival (OS); urinary dysfunctions, including duration of postoperative catheterization (DPC, day), urinary incontinence and frequency, urinary retention and urgency and dysuria; anorectal dysfunctions including constipation, diarrhea and fecal incontinence; and sexual dysfunctions, including a decrease in sexual interest, dyspareunia, decreases in orgasm and sexual satisfaction, and vaginal dryness.
Since the classification of radical surgeries for cervical cancer has been changed from the Piver-Rutledge system to the Querleu-Morrow system since 2008 [31,32], we considered that types II and III were similar to types B and C, respectively. In particular, NSRS was evaluated by full review of some studies where the Piver-Rutledge system was used due to no subtype for defining NSRS [7,9,10,12,13,16,17,18,20,21,22,27,28,29,30], whereas type C1 was considered to be NSRS in other studies based on the Querleu-Morrow system [19,23,24,25,26].
Bladder injury, bowel perforation, vessel injury, and hemorrhage with estimated blood loss >1,000 mL were considered to be intraoperative complications [25,27,28]. Postoperative complications included acute renal failure, bleeding from surgical sites, dysesthesia, febrile morbidity, ileus, infection on surgical sites, lymphocele, metabolic complications, pyelonephritis, reoperation, thromboembolism, and ureteral fistula or stenosis [10,12,18,21,24, 27, 28, 30]. DPC was defined as the time to achieve postvoid residual urine of ≤50 [13,16,18,19,23,25] or ≤100 mL [12,21,24,28,30]. Urinary, anorectal, and sexual dysfunctions were evaluated through interviews or self-reports 6 [19,30] or 12 months after surgery [9,12,20,22,28,29].
In CRS sacrificing the pelvic autonomic nerves-the hypogastric nerve containing sympathetic nerves, the pelvic splanchnic nerve containing parasympathetic nerves, and the vesical branch of pelvic plexus containing both sympathetic and parasympathetic nerves-are known to be easily injured during dissection of the uterosacral ligament, the parametrium, and the posterior part of the vesicouterine ligament, respectively [9]. Thus, we focused on whether each of the three nerves was preserved by NSRS to assess the quality of individual studies because they used different nerve-sparing techniques. As a result, it was found that the three nerves were preserved in all except one study [16], and only five studies confirmed the success rate of the nerve-sparing technique on at least one side (Supplementary Table 1) [17,18,19,22,29].
Furthermore, we assessed the quality of individual studies using the Newcastle-Ottawa Scale (NOS) for 18 enrolled cohort studies [33]. The NOS consists of the following three parameters of quality: selection, comparability, and outcome. It assigns a maximum of four points for selection, two points for comparability, and three points for outcome. In this meta-analysis, we considered a study with an NOS score of ≥8 to be a high-quality study because the mean NOS score was 7.6. As a result, 10 studies (55.6%) showed high quality (Supplementary Table 2).
Continuous variables were shown as standard difference in means (SDMs) with 95% confidence intervals (CIs), which were calculated from mean, SD or p-value, and sample size in each study. Dichotomous data eligible in each study were demonstrated as an odds ratio (OR) with 95% CIs. Furthermore, we conducted survival analysis using a statistical method describer by Tierney et al. [34]. Heterogeneity was assessed using Higgins I2, evaluating the percentage of total variation across studies which was due to heterogeneity rather than chance [35]. Thus, an I2 of >50% was considered to represent substantial heterogeneity, and thereby we used the random effects model using the DerSimonian and Laird method. On the other hand, the fixed effect model using the Mantel-Haenszel method was employed when I2 was ≤50% because it meant no heterogeneity.
Funnel plots were represented to identify publication bias, which were scattered plots of SMDs, ORs, or hazard ratios of individual studies on the x axis against the standard error on the y axis. As a result, all funnel plots resembled symmetric inverter funnels, suggesting no publication bias in this meta-analysis. Moreover, we performed Egger's test if at least three studies were included for each outcome and thereby found no publication bias (p>0.05) (Supplementary Fig. 1). For this meta-analysis, we used SPSS ver. 19.0 (SPSS Inc., Chicago, IL, USA) and Comprehensive Meta-Analysis ver. 2.0 (Biostat Inc., Englewood, NJ, USA). A p-value of <0.05 was considered to be statistically significant.
Supplementary Table 3 shows the general characteristics of 20 comparative studies that included 2,253 patients with cervical cancer. Among them, 1,130 (50.2%) underwent CRS, while 1,145 (49.8%) received NSRS, and there was no significant difference in the frequency of NAC between CRS and NSRS in all except two studies [27,30]. Furthermore, potential confounding factors, including age, adjuvant treatment, body mass index, FIGO stage, depth of stromal invasion, extent of lymphadenectomy, grade, histology, lymph node metastasis, number of resected lymph nodes, parametrial invasion, positive resection margin, and tumor size were adjusted in most of the studies.
As a result, crude analyses showed that blood loss (SDM, -0.251; 95% CI, -0.391 to -0.110) and hospital stay (SDM, -0.224; 95% CI, -0.400 to -0.047) were less, and intraoperative complications (OR, 0.273; 95% CI, 0.105 to 0.715) were less common in patients treated with NSRS. Moreover, the length of the resected vagina was significantly shorter in NSRS than in CRS (SDM, -0.498; 95% CI, -0.795 to -0.201) (Fig. 2). However, there were no significant differences in operative time, postoperative complications, the length of the resected parametrium, DFS, and OS between the two groups (Supplementary Fig. 2).
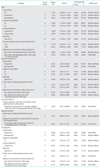
When we performed subgroup analyses for at least three studies to evaluate each outcome according to study design, quality of study, surgical approach, radicality, and adjustment for potential confounding factors, operative time (SDM, 0.948; 95% CI, 0.642 to 1.253) was longer, and intraoperative complications were less common (OR, 0.147; 95% CI, 0.035 to 0.621) in NSRS (Table 1), whereas there were no differences in blood loss, hospital stay, and postoperative complications between NSRS and CRS after adjustment for age, body mass index, extent of lymphadenectomy, FIGO stage, and number of resected lymph nodes (Supplementary Table 4). In terms of survival, DFS and OS were not different between the two treatments (Table 2).
In regard to urinary dysfunctions, crude analyses demonstrated that DPC was shorter (SDM, -1.369; 95% CI, -1.865 to -0.873), and urinary frequency and abnormal sensation were less common in NSRS (OR, 0.347 and 0.067; 95% CI, 0.183 to 0.658 and 0.013 to 0.340, respectively) (Fig. 3). However, there were no significant differences in urinary incontinence, urinary retention, dysuria, and urinary urgency between CRS and NSRS (Supplementary Fig. 3). In terms of anorectal dysfunctions, there were no significant differences in constipation, diarrhea, and fecal incontinence between the two treatments. Furthermore, a decrease in sexual interest, dyspareunia, a decrease in orgasm, or sexual satisfaction, and vaginal dryness were not significantly different between CRS and NSRS (Supplementary Fig. 4).
In subgroup analyses based on study design, quality of study, surgical approach, radicality, postvoid residual urine volume not requiring DPC, follow-up for evaluating dysfunctions, and adjustment for potential confounding factors, DPC was still shorter, and urinary incontinence and urinary frequency were less common in NSRS. Furthermore, constipation was less frequent in NSRS after adjustment for age, adjuvant treatment, extent of lymphadenectomy, and FIGO stage (OR, 0.177; 95% CI, 0.078 to 0.401) (Table 3). However, there were no significant differences in sexual dysfunctions between CRS and NSRS (Supplementary Table 5).
Recent RCTs and one systematic review with a meta-analysis have reported the advantages of NSRS [11,14]. However, they have some limitations as follows: small numbers of studies with low quality of RCTs: seven studies (41.2%) published in the Chinese literature in the systematic review with a metaanalysis, which can act as a bias to interpret meta-analytic results because of difficulty in accessing full papers and the disadvantage that most of the relevant studies have been performed in the limited area [11,36,37,38,39,40,41]; a lack of relevant studies comparing prognosis, anorectal or sexual dysfunctions between the two treatments. Although this meta-analysis could not also overcome these limitations completely, it has major advantages as follows: (1) inclusion of the largest number of relevant English literature which enabled us to compare most of the surgery-related issues between CRS and NSRS; (2) definition of the pelvic autonomic nerves which should be spared in NSRS in spite of different techniques; (3) comparison of urinary, anorectal, and sexual dysfunctions between the two treatments in terms of long-term outcomes (6 or 12 months after surgery); (4) subgroup analyses based on study design, quality of study, surgical approach, radicality, and adjustment for potential confounding factors in order to minimize bias.
As a result, we found that operative time was longer, and intraoperative complications were less common in NSRS despite no significant differences in blood loss, hospital stay, and postoperative complications. Longer operative time and less frequent intraoperative complications may result from more care taken to avoid damaging the pelvic nerves during NSRS. Thus, the surgical field can be dissected even wider than CRS, and meticulous and precise dissection can contribute to decreases in blood loss and injury to adjacent organs [12,42].
Secondly, the length of the resected vagina was shorter in NSRS, while the length of the resected parametrium, DFS, and OS were not significantly different between CRS and NSRS. These finding can be supported by some studies suggesting that the level of colpectomy should be restricted to 2 cm in order to preserve the most distal portion of the vesical branch of the pelvic plexus [16,24,43]. On the other hand, the safety of NSRS still remains controversial because of the concerns of less radicality of NSRS [44,45]. Although this meta-analysis was performed under the conditions that radicality could affect prognosis (up to 90% of patients with a large tumor of >4 cm, and less than 50% of them who received adjuvant radiotherapy), it demonstrated that NSRS may not reduce the radicality affecting prognosis.
Thirdly, DPC was shorter, and urinary incontinence and frequency were less common in NSRS. Sympathetic nerves in the hypogastric nerve and the vesical branch of the pelvic plexus stimulate the urethral sphincter and inhibit the detrusor muscle of the bladder, whereas parasympathetic nerves in the pelvic splanchnic nerve and the vesical branch of the pelvic plexus relax the urethral sphincter and stimulate the detrusor muscle of the bladder [10,46]. Thus, CRS may increase DPC, and urinary incontinence and frequency can be expected by autonomic dysregulation after surgical disruption [21,22,47,48]. Thus, these meta-analyses is meaningful in supporting the efficacy of autonomic nerve preservation by NSRS on urinary functions.
Fourthly, constipation was less common in NSRS. Sympathetic nerves inhibit the expulsion of feces and stimulate the internal sphincter of the anus, whereas parasympathetic nerves show opposite effects [10,46]. In particular, a previous study has suggested the hypothesis that injury to the pelvic autonomic nerves by CRS disrupts the spinal reflex, which causes internal sphincter dysregulation and decreased rectal sensation [6]. Thus, this meta-analysis supports the hypothesis and suggests that NSRS may reduce the incidence of functional defecation disorders, such as constipation.
Fifthly, there were no significant differences in sexual dysfunctions between CRS and NSRS. Theoretically, autonomic nerve fibers in the vascular smooth muscle cells of the vagina innervate reproductive organs and are responsive to circulating steroids [49]. Thus, damage to autonomic nerves caused by CRS may change the neurogenic control of the blood vessels of the vagina wall and thereby disturbs vaginal blood flow during sexual arousal and lubrication-swelling response [9,49,50,51]. However, this meta-analysis failed to show a decrease in sexual dysfunctions by NSRS in comparison with CRS, which means that autonomic nerve preservation may not be associated with the improvement in sexual functions and that multiple factors, including vaginal shortness, tissue fibrosis, radiotherapy, a decrease in ovarian function, and psychological factors, may be more important to improve sexual functions [30].
When we consider that the nerve-sparing technique is not currently uniform, and thereby a large-scale RCT is not easy to perform, this meta-analysis is important because it showed the possibility that NSRS can give better quality of life by preserving urinary and anorectal functions without adverse effects on clinical outcomes and sexual functions in patients with early-stage cervical cancer. Furthermore, it is helpful in planning large-scale prospective randomized trials for valuable epidemiologic evidence.
Figures and Tables
 | Fig. 1PRISMA diagram. The search strategy and number of studies identified for inclusion in this meta-analysis. |
 | Fig. 2Forest plots for standard differences (Std diffs) in means or odds ratios with 95% confidence intervals (CIs) to compare (A) blood loss, (B) hospital stay, (C) intraoperative complications, and (D) the length of the resected vagina between conventional radical surgery (CRS) and nerve-sparing radical surgery (NSRS) for cervical cancer. |
 | Fig. 3Forest plots for standard differences (Std diffs) in means or odds ratios with 95% confidence intervals (CIs) to compare (A) the duration of postoperative catheterization, (B) urinary frequency between conventional radical surgery (CRS) and nerve-sparing radical surgery (NSRS) for cervical cancer. |
Table 1
Subgroup analyses for comparing clinical outcomes between conventional and nerve sparing radical surgery for cervical cancer

Table 2
Subgroup analyses for comparing survival between conventional and nerve sparing radical surgery for cervical cancer

Table 3
Subgroup analyses for comparing postoperative urinary and anorectal functions between conventional and nerve sparing radical surgery for cervical cancer
ACKNOWLEDGMENTS
We appreciate the Medical Research Collaborating Center (MRCC) of Seoul National University Hospital for statistical analysis. This research was supported by grants (No. 04-2012-0890; 03-2012-0170) from the Seoul National University Hospital research fund and the Priority Research Centers Program (No. 2009-0093820), Basic Science Research Program (No. 2011-0025394), and BK21 plus program (No. 5256-20140100) through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology. Moreover, it was also supported by a grant of the Korean Health Technology R&D Project, Ministry of Health of Welfare (HI14C2404).
References
1. Ditto A, Martinelli F, Borreani C, Kusamura S, Hanozet F, Brunelli C, et al. Quality of life and sexual, bladder, and intestinal dysfunctions after class III nerve-sparing and class II radical hysterectomies: a questionnaire-based study. Int J Gynecol Cancer. 2009; 19:953–957.
2. Kim HS, Choi CH, Lim MC, Chang SJ, Kim YB, Kim MA, et al. Safe criteria for less radical trachelectomy in patients with early-stage cervical cancer: a multicenter clinicopathologic study. Ann Surg Oncol. 2012; 19:1973–1979.
3. Suh DH, Kim JW, Kang S, Kim HJ, Lee KH. Major clinical research advances in gynecologic cancer in 2013. J Gynecol Oncol. 2014; 25:236–248.
4. Choi SY, Lee KH, Suk HJ, Chae HD, Kang BM, Kim CH. Successful pregnancy by direct intraperitoneal insemination in an infertile patient with failure of recanalization of isthmic stenosis after laparoscopic radical trachelectomy. Obstet Gynecol Sci. 2014; 57:82–85.
5. Zullo MA, Manci N, Angioli R, Muzii L, Panici PB. Vesical dysfunctions after radical hysterectomy for cervical cancer: a critical review. Crit Rev Oncol Hematol. 2003; 48:287–293.
6. Barnes W, Waggoner S, Delgado G, Maher K, Potkul R, Barter J, et al. Manometric characterization of rectal dysfunction following radical hysterectomy. Gynecol Oncol. 1991; 42:116–119.
7. Trimbos JB, Maas CP, Deruiter MC, Peters AA, Kenter GG. A nervesparing radical hysterectomy: guidelines and feasibility in Western patients. Int J Gynecol Cancer. 2001; 11:180–186.
8. Bergmark K, Avall-Lundqvist E, Dickman PW, Henningsohn L, Steineck G. Vaginal changes and sexuality in women with a history of cervical cancer. N Engl J Med. 1999; 340:1383–1389.
9. Pieterse QD, Ter Kuile MM, Deruiter MC, Trimbos JB, Kenter GG, Maas CP. Vaginal blood flow after radical hysterectomy with and without nerve sparing. A preliminary report. Int J Gynecol Cancer. 2008; 18:576–583.
10. Raspagliesi F, Ditto A, Fontanelli R, Zanaboni F, Solima E, Spatti G, et al. Type II versus type III nerve-sparing radical hysterectomy: comparison of lower urinary tract dysfunctions. Gynecol Oncol. 2006; 102:256–262.
11. Li B, Zhang R, Wu LY, Zhang GY, Li X, Yu GZ. A prospective study on nerve-sparing radical hysterectomy in patients with cervical cancer. Zhonghua Fu Chan Ke Za Zhi. 2008; 43:606–610.
12. Wu J, Liu X, Hua K, Hu C, Chen X, Lu X. Effect of nerve-sparing radical hysterectomy on bladder function recovery and quality of life in patients with cervical carcinoma. Int J Gynecol Cancer. 2010; 20:905–909.
13. Chen C, Li W, Li F, Liu P, Zhou J, Lu L, et al. Classical and nerve-sparing radical hysterectomy: an evaluation of the nerve trauma in cardinal ligament. Gynecol Oncol. 2012; 125:245–251.
14. Long Y, Yao DS, Pan XW, Ou TY. Clinical efficacy and safety of nerve-sparing radical hysterectomy for cervical cancer: a systematic review and meta-analysis. PLoS One. 2014; 9:e94116.
15. Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009; 62:e1–e34.
16. Possover M, Stober S, Plaul K, Schneider A. Identification and preservation of the motoric innervation of the bladder in radical hysterectomy type III. Gynecol Oncol. 2000; 79:154–157.
17. van den Tillaart SA, Kenter GG, Peters AA, Dekker FW, Gaarenstroom KN, Fleuren GJ, et al. Nerve-sparing radical hysterectomy: local recurrence rate, feasibility, and safety in cervical cancer patients stage IA to IIA. Int J Gynecol Cancer. 2009; 19:39–45.
18. Skret-Magierlo J, Narog M, Kruczek A, Kluza R, Kluz T, Magon T, et al. Radical hysterectomy during the transition period from traditional to nerve-sparing technique. Gynecol Oncol. 2010; 116:502–505.
19. Tseng CJ, Shen HP, Lin YH, Lee CY, Wei-Cheng Chiu W. A prospective study of nerve-sparing radical hysterectomy for uterine cervical carcinoma in Taiwan. Taiwan J Obstet Gynecol. 2012; 51:55–59.
20. Bogani G, Cromi A, Uccella S, Serati M, Casarin J, Pinelli C, et al. Nervesparing versus conventional laparoscopic radical hysterectomy: a minimum 12 months\' follow-up study. Int J Gynecol Cancer. 2014; 24:787–793.
21. Querleu D, Narducci F, Poulard V, Lacaze S, Occelli B, Leblanc E, et al. Modified radical vaginal hysterectomy with or without laparoscopic nerve-sparing dissection: a comparative study. Gynecol Oncol. 2002; 85:154–158.
22. Todo Y, Kuwabara M, Watari H, Ebina Y, Takeda M, Kudo M, et al. Urodynamic study on postsurgical bladder function in cervical cancer treated with systematic nerve-sparing radical hysterectomy. Int J Gynecol Cancer. 2006; 16:369–375.
23. Cibula D, Slama J, Velechovska P, Fischerova D, Zikan M, Pinkavova I, et al. Factors affecting spontaneous voiding recovery after radical hysterectomy. Int J Gynecol Cancer. 2010; 20:685–690.
24. Espino-Strebel EE, Luna JT, Domingo EJ. A comparison of the feasibility and safety of nerve-sparing radical hysterectomy with the conventional radical hysterectomy. Int J Gynecol Cancer. 2010; 20:1274–1283.
25. Liang Z, Chen Y, Xu H, Li Y, Wang D. Laparoscopic nerve-sparing radical hysterectomy with fascia space dissection technique for cervical cancer: description of technique and outcomes. Gynecol Oncol. 2010; 119:202–207.
26. Cibula D, Pinkavova I, Dusek L, Slama J, Zikan M, Fischerova D, et al. Local control after tailored surgical treatment of early cervical cancer. Int J Gynecol Cancer. 2011; 21:690–698.
27. Ditto A, Martinelli F, Mattana F, Reato C, Solima E, Carcangiu M, et al. Class III nerve-sparing radical hysterectomy versus standard class III radical hysterectomy: an observational study. Ann Surg Oncol. 2011; 18:3469–3478.
28. Ceccaroni M, Roviglione G, Spagnolo E, Casadio P, Clarizia R, Peiretti M, et al. Pelvic dysfunctions and quality of life after nerve-sparing radical hysterectomy: a multicenter comparative study. Anticancer Res. 2012; 32:581–588.
29. Pieterse QD, Kenter GG, Maas CP, de Kroon CD, Creutzberg CL, Trimbos JB, et al. Self-reported sexual, bowel and bladder function in cervical cancer patients following different treatment modalities: longitudinal prospective cohort study. Int J Gynecol Cancer. 2013; 23:1717–1725.
30. Wang W, Li B, Zuo J, Zhang G, Yang Y, Zeng H, et al. Evaluation of pelvic visceral functions after modified nerve-sparing radical hysterectomy. Chin Med J (Engl). 2014; 127:696–701.
31. Piver MS, Rutledge F, Smith JP. Five classes of extended hysterectomy for women with cervical cancer. Obstet Gynecol. 1974; 44:265–272.
32. Querleu D, Morrow CP. Classification of radical hysterectomy. Lancet Oncol. 2008; 9:297–303.
33. Wells GA, Shea B, Oconnell D, Peterson J, Welch V, Losos M, et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses [Internet]. Ottawa: Ottawa Hospital Research Institute;c2014. cited 2015 Mar 17. Available from: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp.
34. Tierney JF, Stewart LA, Ghersi D, Burdett S, Sydes MR. Practical methods for incorporating summary time-to-event data into metaanalysis. Trials. 2007; 8:16.
35. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003; 327:557–560.
36. Sun L, Wu LY, Zhang WH, Li XG, Song Y, Zhang X. Preliminary study of nerve sparing radical hysterectomy in patients with cervical cancer. Zhonghua Zhong Liu Za Zhi. 2009; 31:607–611.
37. Long Y, Yao DS, Gao K, Xie XL. Preliminary study on clinical effect of nerve sparing radical hysterectomy for cervical cancer. Chin Clin Oncol. 2010; 15:1083–1090.
38. Zhu T, Yu AJ, Shou HF, Chen X, Zhu JQ, Yang ZY, et al. Feasibility of unilateral or bilateral nerve-sparing radical hysterectomy in patients with cervical cancer and evaluation of the post-surgery recovery of the bladder and rectal function. Zhonghua Zhong Liu Za Zhi. 2011; 33:53–57.
39. Chen Y, Li Y, Xu HC, Li JN, Li YY, Liang ZQ. Laparoscopic anatomical nerve sparing radical hysterectomy for cervical cancer: a clinical analysis of 37 cases. Zhonghua Fu Chan Ke Za Zhi. 2009; 44:359–363.
40. Zhang Y, Wang D, Ma Y, Pan CQ. A clinical study of surgical treatment of the cervical cancer with laparoscopic nerve sparing radical hysterectomy. Prog Obstet Gynecol. 2010; 19:31–32.
41. Lu Y, Yao DS, Mo LZ, Li F, Pan ZM. Preliminary clinical study of laparoscopic pelvic autonomic nerve-plane sparing radical hystrectomy. Chin Clin Oncol. 2012; 17:347–350.
42. Liang JT, Chien CT, Chang KJ, Hsu HC, Chu SH, Lai MK, et al. Neurophysiological basis of sympathetic nerve-preserving surgery for lower rectal cancer: a canine model. Hepatogastroenterology. 1998; 45:2206–2214.
43. Raspagliesi F, Ditto A, Fontanelli R, Solima E, Hanozet F, Zanaboni F, et al. Nerve-sparing radical hysterectomy: a surgical technique for preserving the autonomic hypogastric nerve. Gynecol Oncol. 2004; 93:307–314.
44. Hockel M. Do we need a new classification for radical hysterectomy? Insights in surgical anatomy and local tumor spread from human embryology. Gynecol Oncol. 2007; 107:1 Suppl 1. S106–S112.
45. Dursun P, LeBlanc E, Nogueira MC. Radical vaginal trachelectomy (Dargent\'s operation): a critical review of the literature. Eur J Surg Oncol. 2007; 33:933–941.
46. Rob L, Halaska M, Robova H. Nerve-sparing and individually tailored surgery for cervical cancer. Lancet Oncol. 2010; 11:292–301.
47. Brown JS, Sawaya G, Thom DH, Grady D. Hysterectomy and urinary incontinence: a systematic review. Lancet. 2000; 356:535–539.
48. Raspagliesi F, Ditto A, Hanozet F, Martinelli F, Solima E, Zanaboni F, et al. Nerve-sparing radical hysterectomy in cervical cancer: evolution of concepts. Gynecol Oncol. 2007; 107:1 Suppl 1. S119–S121.
49. Papka RE, Mowa CN. Estrogen receptors in the spinal cord, sensory ganglia, and pelvic autonomic ganglia. Int Rev Cytol. 2003; 231:91–127.
50. Maas CP, ter Kuile MM, Laan E, Tuijnman CC, Weijenborg PT, Trimbos JB, et al. Objective assessment of sexual arousal in women with a history of hysterectomy. BJOG. 2004; 111:456–462.
51. Ercoli A, Delmas V, Gadonneix P, Fanfani F, Villet R, Paparella P, et al. Classical and nerve-sparing radical hysterectomy: an evaluation of the risk of injury to the autonomous pelvic nerves. Surg Radiol Anat. 2003; 25:200–206.
SUPPLEMENTARY MATERIALS
Supplementary Fig. 1
Funnel plots with Egger's test representing no publication bias in this meta-analysis for comparing clinical outcomes: (A) operative time, (B) blood loss, (C) hospital stay, (D) intraoperative complications, (E) postoperative complications, (F) disease-free survival, (G) overall survival, (H) duration of postoperative catheterization, (I) urinary incontinence, (J) urinary retention, (K) urinary frequency, (L) constipation, (M) decrease in sexual interest, and (N) dyspareunia between conventional and nerve-sparing radical surgery for cervical cancer.
Supplementary Fig. 2
Forest plots for standard differences (Std diffs) in means or odds ratios with 95% confidence intervals (CIs) to compare (A) operative time, (B) postoperative complications, (C) the length of the resected parametrium, (D) disease-free survival, and (E) overall survival between conventional radical surgery (CRS) and nerve-sparing radical surgery (NSRS) for cervical cancer.
Supplementary Fig. 3
Forest plots for standard differences (Std diffs) in means or odds ratios with 95% confidence intervals (CIs) to compare (A) urinary incontinence, (B) urinary retention, (C) dysuria, and (D) urinary urgency between conventional radical surgery (CRS) and nerve-sparing radical surgery (NSRS) for cervical cancer.
Supplementary Fig. 4
Forest plots for odds ratios with 95% confidence intervals (CIs) to compare (A) constipation, (B) diarrhea, (C) fecal incontinence, (D) a decrease in sexual interest, (E) dyspareunia, (F) decrease in orgasm, (G) decrease in sexual satisfaction, and (H) vaginal dryness between conventional radical surgery (CRS) and nerve-sparing radical surgery (NSRS) for cervical cancer.
Supplementary Table 2
The Newcastle-Ottawa Scale for assessing qualities of 19 included cohort studies




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download