Abstract
Objective
This study was designed to evaluate the survival benefit of laparoscopic surgical staging (LSS)-guided tailored radiation therapy (RT) in locally advanced cervical cancer (LACC).
Methods
We retrospectively reviewed 89 LACC patients' medical records who primarily received non-surgical treatment, of which pretreatment LSS was performed in 20 (LSS group) and primary chemoradiation therapy (CCRT) without LSS (CCRT group) was carried out in 69 from January 2000 to January 2006. We analyzed clinical characteristics, pretreatment imaging study results and survival outcomes including disease free survival (DFS) and overall survival (OS) to compare them between the two groups.
Results
There were as many as eight cases (40%) of LSS related complications. The mean time interval between LSS and RT or CCRT was 26.6 days (±18.8 days). Six out of twenty (30%) in LSS group and 10 out of 69 (14.5%) in CCRT group received extended field RT when paraaortic lymph nodes (LNs) were positive based on the pathologic findings after LSS and the results of imaging studies, respectively. Three-year DFS and OS were both better in 33 imaging-negative CCRT group patients than those in 4 imaging-negative/pathology-positive (false negative) patients after LSS (3-year DFS, 50% vs. 87%, p=0.022; 3-year OS, 50% vs. 84%, p=0.033). The 5-year DFS rates were 52% and 55% in LSS group and in CCRT group, respectively (p=0.28). The 5-year OS rates were 68% in LSS group and 62% in CCRT group without significant difference between the two groups (p=0.79).
While early-stage cervical cancer (stage IA-IIA) is treated with surgery followed by adjuvant therapy according to the risk factors after surgery, locally advanced cervical cancer (LACC, stage IIB-IVA) is primarily treated by radiation therapy (RT) or concurrent chemoradiation therapy (CCRT).1 Lymph nodes (LNs) metastasis is an important prognostic factor in not only that it is the most significant prognostic factor in early-stage cervical cancer but also that the field of RT depend on the extent of LN involvement.2
However, the current clinical International Federation of Gynecology and Obstetrics (FIGO) staging system does not consider LN status,3 which results in some discrepancies between FIGO stage and prognosis. In addition, preoperative imaging studies were not known to be satisfactorily sensitive for detecting LN metastasis in cervical cancer.4-7 Choi et al.6 reported that the sensitivity of pretreatment MRI and PET/CT was 30.3% and 57.6%, respectively, compared to pathological diagnosis of LN. These explain the reason why investigators try to ponder the role of laparoscopic surgical staging (LSS) in the treatment of LACC.
As laparoscopic procedures have become more popular, LSS appeared to be an attractive alternative method for gynecologic oncologists to evaluate preoperative LN metastasis, although no conclusive data exist that indicate an advantage to this approach for now.8 They expected that surgical staging could identify metastatic nodes and other intra-abdominal diseases accurately that were not detected by the imaging studies and maximize the effect of RT by modifying radiation field and by resection of bulky metastatic LNs to help them plan the treatment of disease with metastatic nodes.2,9
The purpose of the current study was to report our institution's experience of LSS and evaluate the survival benefit of LSS-guided tailored RT in LACC.
The study population was the patients who pathologically proved cervical cancer and underwent LSS or primary CCRT between January 2000 and January 2006. The inclusion criteria were as follows: Histologically confirmed FIGO stage IB2-IVA cervical cancer patients who were considered inappropriate for radical surgery because of the advanced stage of IIB-IVA or large tumor ≥ 4 cm and patients who consented for this procedure. The exclusion criteria were patients who were considered to have metastatic lesions in distant organs (stage IVB), severe medical problems, poor performance status or old age more than 70 years old. A total of twenty patients underwent LSS during study period, while 69 being treated with primary CCRT. Medical records of all subjects were reviewed for patients' characteristics, surgical results of LSS, accuracy of imaging modality, and clinical parameters of both groups. Statistical analysis was performed using SPSS ver. 11.0 (SPSS Inc., Chicago, IL, USA). The nonparametric, Mann-Whitney test, Kaplan-Meier and log-rank test were used for comparison between the two groups. The Kyungpook National University Hospital (Daegu, Korea) does not require approval from the Institutional Review Board for retrospective chart reviews; hence this analysis was exempted from the approval process.
The primary puncture was made by an 11 mm sharpened triple edge pyramidal trocar after a skin incision along the lower margin of the umbilicus. CO2 gas was used for insufflation of the abdominal cavity. The 2nd puncture was made at the suprapubic area with a 5 mm trocar. The 3rd and 4th punctures were both made in the lower quadrant with 5 mm trocars, and the 5th puncture was made in the left upper quadrant with an 11 mm trocar. The main procedure began after a thorough exploration of the pelvic and abdominal cavities with biopsies of all suspicious lesions.
The lymphadenectomy began from the right paraaortic area by incising the peritoneum covering the upper part of the right iliac artery. The camera showed the aorta and vena cava horizontally with the vena cava on top of the aorta. After identification and lateral pushing of the right ureter, the precaval and paracaval LNs were dissected from the common iliac artery on the right side to the level of the right ovarian vein. For a left side paraaortic lymphadenectomy, after identification of the left ureter and the inferior mesenteric artery, the LN was dissected from the left paraaortic and presacral area and an inframesenteric lymphadenectomy was done. The left paraaortic LN dissection extended up to the interaortocaval region. After completion of the paraaortic lymphadenectomy, the pelvic lymphadenectomy began on the left side by coagulation and cutting of the round ligament. The LN near the external iliac artery was dissected, cautiously preserving the genitofemoral nerve. After identification and traction of the left medial umbilical ligament, the obturator fossa was opened with blunt dissection. The LN along the external iliac vein and obturator nerve was dissected and removed. All LNs dissected were removed through the 11 mm port site enveloped with an endobag for prevention of port site metastasis (Fig. 1).
The patients received one of the 4 combination chemotherapies in both group: weekly cisplatin (40 mg/m2)×6 courses, paclitaxel (175 mg/m2), and cisplatin (75 mg/m2) every 3 weeks×2 courses, weekly cisplatin (50 mg/ m2) and mitomycin (10 mg/m2)×6 courses, cisplatin (75 mg/m2) and mitomycin (10 mg/m2) and Oncovin (1.0 mg/m2) every 3 weeks×2 courses.
The patients received 45 Gy whole pelvic RT at 39.6 Gy in 22 fractions (five fractions per week) and at 5.4 Gy in 3 fractions with middle block, after whole pelvic RT. Brachy-therapy was performed at 30 Gy in 5 fractions (two fractions per week). Paraaortic field was irradiated at 45 Gy in 25 fractions.
Of the 20 patients in LSS group, all of which done without conversion to laparotomy, 16 patients underwent both paraaortic lymphadenectomy and pelvic lymphadenectomy. The rest of four patients underwent either pelvic lymphadenectomy or paraaortic lymphadenectomy, three the former and one the latter.
Patient characteristics were shown in Table 1. Age, histologic type, clinical stage distribution, tumor size and LN positivity in CT or MRI were all similar between the two groups. The time intervals between LSS and definitive treatment ranged from 7 days to 110 days in LSS group with mean 26.6 days. Six out of twenty (30%) in LSS group and 10 out of 69 (14.5%) in CCRT group received extended field RT when paraaortic LNs were positive based on the pathologic findings after LSS and the results of imaging studies, respectively (p=0.01). In LSS group, the patient whose age was older than 70 or WHO/ECOG performance status was 3 or more received RT alone. There was one patient who stopped therapy on her own after only 1 cycle of chemotherapy (Table 1).
Surgical outcomes of LSS are summarized in Table 2. Despite no severe intraoperative complication such as vessel rupture or ureter damage, there were various postoperative complications associated with LSS. Eight patients suffered from several complications and two out of them were grade III surgical complications which needed surgical or radiological interventions.10
The accuracy of an MRI or CT in the assessment of LN involvement is 68.8% for paraaortic LNs and 75.0% for pelvic LNs. Imaging could not detect four cases of metastatic paraaortic LN and two cases of metastatic pelvic LN (Table 3).
During 59 months of mean follow-up period, 10 recurrences (50%) and 7 disease specific deaths occurred in LSS group. Five-year disease-free survival (DFS) rate was 52% and the 5-year overall survival (OS) rate was 68%. In CCRT group, the mean follow-up period was 71 months. Twenty-three patients (50%) recurred and eighteen died of the disease. Five-year DFS rate and 5-year OS rate was 55% and 62%, respectively. There was no difference of DFS and OS between the two groups (p=0.28 and 0.79, respectively) (Fig. 2).
LN metastasis is considered to be the most important prognostic factor.5 The discrepancy between clinical staging and prognosis was largely attributed to the fact that the same therapy was given in LACC regardless of nodal status. A lot of efforts have been made to resolve this problem using imaging modalities. However, the sensitivity of CT and MRI is reported as 34% and 30.3-75%, respectively,4-7 and PET has limitations in detecting metastatic nodes due to lesion sizes and other causes in spite of the reported great clinical usefulness.5,11-13 In this study, PETs were conducted for five patients in LSS group and the sensitivity was the same as that of MRI or CT. Therefore, surgical staging was considered an important pretreatment procedure in LACC, though the effectiveness was controversial in many previous reports (Table 4).
Since the feasibility report of LSS in LACC by Vidaurreta et al.14 in which false negative rate of imaging study was as high as 48.3% (18/38), several studies have shown the same results about the safety and usefulness of surgical staging.15-18 On the other hand, the first randomized study of Lai et al.19 revealed a significant worse DFS rate of surgically staged patients than that of clinically staged patients. After Lai's study, there were a couple of reports which demonstrated survival benefits of LSS through therapeutic effect of removal of metastatic LNs and tailored RT.20-22
Fagotti et al.9 summarized the purpose of pretreatment surgical staging: to identify positive nodes in patients with LACC that is considered clinically negative; to recognize intraperitoneal disease; to tailor the treatment plan based on histological confirmation; to remove macroscopically positive nodes; and to transpose the ovary outside the radiation field. Initial studies were undertaken with the hope of increased survival in patients with advanced cervical cancer. The postulation that a staging operation could increase the survival rate was based on the effect of the tailored extended field RT, appropriate adjuvant treatment modality after histological confirmation of nodal status and debulking of positive LNs.
A previous report described increased survival in patients with macroscopic LN resected.23 Another study also reported increased survival in patients without residual paraaortic LNs.24 Marnitz et al.21 found that the removal of more than five pelvic and/or five positive paraaortic LNs was associated with significant improvement of OS. However, Goff et al.18 mentioned that it would be impossible to verify that surgical excision of grossly involved nodes provides a surgical advantage without sufficient randomized trials.
Debulking effects of the involved LNs also seemed to be reflected in the decrease of squamous cell carcinoma antigen (SCCA) in this study. A significant dropping of SCCA was observed even without any change of the primary cervical tumor mass after LSS in positive LN group (N=13), from 11.0 to 3.4. Meanwhile in the negative LN group, the dropping was relatively small, from 5.3 to 3.7.
Although the significance of the removal of metastatic LNs is controversial, it can allow radiation oncologists to use lower doses in extended field RT. A randomized trial conducted by the Radiation Therapy Oncology Group (RTOG) demonstrated an overall benefit of extended field RT over pelvic RT for patients with cervical cancer, which can be explained by a lower incidence of distant failure in complete responders and a better salvage in the complete responders who later failed locally.25 Extended field RT has been shown to improve local control in stage IIIC endometrial cancer, but radiation-related toxicity occurred more frequently than in other radiation modalities. To minimize the dose to the small bowel and other normal tissue, intensity modulated radiation therapy (IMRT) was recommended.26
DFS and OS were compared between 13 patients with histologically confirmed positive LNs after LSS (true positive) and 36 patients in CCRT group with radiologically suspected metastatic LNs only to show no significant difference (5-year DFS, 44.4% vs. 46.1%, p=0.24; 5-year OS, 58.3% vs. 54.2%, p=0.35). However, three-year DFS and OS were both better in 33 imaging-negative CCRT group patients than those in 4 imaging-negative/pathology-positive (false negative) patients after LSS (3-year DFS, 50% vs. 87%, p=0.022; 3-year OS, 50% vs. 84%, p=0.033). These findings indicate that no additional benefit is given by performing LSS regardless of the pre-LSS imaging study result and there is not that significant therapeutic beneficial effect of metastatic LNs removal. Even though there was no statistical significance, DFS of CCRT group showed a weak trend toward better than that of LSS group (Fig. 2B). It might be resulted from the difference of type of radiations between the two groups and delayed time intervals to treatment mainly due to the morbidity of the LSS procedure (Table 1).
Now gynecologic oncologic group (GOG) is about to start GOG 233 which is as to the utility of preoperative FDG-PET/CT scanning prior to primary CCRT to detect retroperitoneal LN metastasis in advanced gynecologic cancers including LACC. This prospective randomized trial is expected to suggest a good modality that can overcome the low sensitivity/specificity of MRI or CT and improve the survival of LACC.
In conclusion, even though LSS for LACC provided a more accurate LN status, LSS-based RT tailoring did not show survival benefit in LACC. Given the possible bad influence of LSS itself, time delay to definite treatment or its related complications, further studies are required to find new method to improve survival outcomes in LACC.
Figures and Tables
Fig. 1
Paraaortic and pelvic lymphadenectomy (A: left renal vein, B: vena cava, C: aorta, D: inferior mesenteric artery, E: both common iliac artery, F: right ureter).
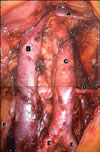
Fig. 2
(A) Five-year disease-free survival rate in laparoscopic surgical staging and primary concurrent cheomoradiation group. (B) Five-year overall survival rate in laparoscopic surgical staging and primary concurrent chemoradiation group.

References
1. Bidus MA, Elkas JC. Berek JS, editor. Cervical and vaginal cancer. Berek & Novak's gynecology. 2007. 14th ed. Philadelphia: Lippincott Williams & Wilkins;1403–1455.
2. Lim MC, Bae J, Park JY, Lim S, Kang S, Seo SS, et al. Experiences of pretreatment laparoscopic surgical staging in patients with locally advanced cervical cancer: results of a prospective study. J Gynecol Oncol. 2008. 19:123–128.
3. Pecorelli S. Revised FIGO staging for carcinoma of the vulva, cervix, and endometrium. Int J Gynaecol Obstet. 2009. 105:103–104.
4. Jones B, Kingston R, Green J, Adams E, Bleasdale C. The effect of treatment duration in the local control of cervix cancer. Radiother Oncol. 1993. 28:179–180.
5. Chung HH, Kang KW, Cho JY, Kim JW, Park NH, Song YS, et al. Role of magnetic resonance imaging and positron emission tomography/computed tomography in preoperative lymph node detection of uterine cervical cancer. Am J Obstet Gynecol. 2010. 203:156.e1. 156.e5.
6. Choi HJ, Roh JW, Seo SS, Lee S, Kim JY, Kim SK, et al. Comparison of the accuracy of magnetic resonance imaging and positron emission tomography/computed tomography in the presurgical detection of lymph node metastases in patients with uterine cervical carcinoma: a prospective study. Cancer. 2006. 106:914–922.
7. Hricak H, Lacey CG, Sandles LG, Chang YC, Winkler ML, Stern JL. Invasive cervical carcinoma: comparison of MR imaging and surgical findings. Radiology. 1988. 166:623–631.
8. Monk BJ, Tewari KS. DiSaia PJ, Creasman WT, editors. Invasive cervical cancer. Clinical gynecologic oncology. 2007. 7th ed. Philadelphia: Mosby;55–124.
9. Fagotti A, Fanfani F, Longo R, Legge F, Mari A, Gagliardi ML, et al. Which role for pre-treatment laparoscopic staging? Gynecol Oncol. 2007. 107:1 Suppl 1. S101–S105.
10. Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004. 240:205–213.
11. Rose PG, Adler LP, Rodriguez M, Faulhaber PF, Abdul-Karim FW, Miraldi F. Positron emission tomography for evaluating para-aortic nodal metastasis in locally advanced cervical cancer before surgical staging: a surgicopathologic study. J Clin Oncol. 1999. 17:41–45.
12. Esthappan J, Mutic S, Malyapa RS, Grigsby PW, Zoberi I, Dehdashti F, et al. Treatment planning guidelines regarding the use of CT/PET-guided IMRT for cervical carcinoma with positive paraaortic lymph nodes. Int J Radiat Oncol Biol Phys. 2004. 58:1289–1297.
13. Havrilesky LJ, Kulasingam SL, Matchar DB, Myers ER. FDG-PET for management of cervical and ovarian cancer. Gynecol Oncol. 2005. 97:183–191.
14. Vidaurreta J, Bermudez A, di Paola G, Sardi J. Laparoscopic staging in locally advanced cervical carcinoma: a new possible philosophy? Gynecol Oncol. 1999. 75:366–371.
15. Sonoda Y, Leblanc E, Querleu D, Castelain B, Papageorgiou TH, Lambaudie E, et al. Prospective evaluation of surgical staging of advanced cervical cancer via a laparoscopic extraperitoneal approach. Gynecol Oncol. 2003. 91:326–331.
16. Vergote I, Amant F, Berteloot P, Van Gramberen M. Laparoscopic lower para-aortic staging lymphadenectomy in stage IB2, II, and III cervical cancer. Int J Gynecol Cancer. 2002. 12:22–26.
17. Hertel H, Kohler C, Elhawary T, Michels W, Possover M, Schneider A. Laparoscopic staging compared with imaging techniques in the staging of advanced cervical cancer. Gynecol Oncol. 2002. 87:46–51.
18. Goff BA, Muntz HG, Paley PJ, Tamimi HK, Koh WJ, Greer BE. Impact of surgical staging in women with locally advanced cervical cancer. Gynecol Oncol. 1999. 74:436–442.
19. Lai CH, Huang KG, Hong JH, Lee CL, Chou HH, Chang TC, et al. Randomized trial of surgical staging (extraperitoneal or laparoscopic) versus clinical staging in locally advanced cervical cancer. Gynecol Oncol. 2003. 89:160–167.
20. Chung HH, Lee S, Sim JS, Kim JY, Seo SS, Park SY, et al. Pretreatment laparoscopic surgical staging in locally advanced cervical cancer: preliminary results in Korea. Gynecol Oncol. 2005. 97:468–475.
21. Marnitz S, Kohler C, Roth C, Fuller J, Hinkelbein W, Schneider A. Is there a benefit of pretreatment laparoscopic transperitoneal surgical staging in patients with advanced cervical cancer? Gynecol Oncol. 2005. 99:536–544.
22. Leblanc E, Narducci F, Frumovitz M, Lesoin A, Castelain B, Baranzelli MC, et al. Therapeutic value of pretherapeutic extraperitoneal laparoscopic staging of locally advanced cervical carcinoma. Gynecol Oncol. 2007. 105:304–311.
23. Cosin JA, Fowler JM, Chen MD, Paley PJ, Carson LF, Twiggs LB. Pretreatment surgical staging of patients with cervical carcinoma: the case for lymph node debulking. Cancer. 1998. 82:2241–2248.
24. Kim PY, Monk BJ, Chabra S, Burger RA, Vasilev SA, Manetta A, et al. Cervical cancer with paraaortic metastases: significance of residual paraaortic disease after surgical staging. Gynecol Oncol. 1998. 69:243–247.
25. Rotman M, Pajak TF, Choi K, Clery M, Marcial V, Grigsby PW, et al. Prophylactic extended-field irradiation of para-aortic lymph nodes in stages IIB and bulky IB and IIA cervical carcinomas. Ten-year treatment results of RTOG 79-20. JAMA. 1995. 274:387–393.
26. Mundt AJ, Murphy KT, Rotmensch J, Waggoner SE, Yamada SD, Connell PP. Surgery and postoperative radiation therapy in FIGO Stage IIIC endometrial carcinoma. Int J Radiat Oncol Biol Phys. 2001. 50:1154–1160.




 PDF
PDF ePub
ePub Citation
Citation Print
Print






 XML Download
XML Download