초록
Background
Xpert Flu (Cepheid, USA) allows for fully automated real-time RT-PCR using a single-use dis-posable cartridge. The aim of this study was to evaluate Xpert Flu for the detection of influenza A virus and subtype A/H1N1/2009 pandemic virus.
Methods
We conducted a prospective comparison study for Xpert Flu with the RealTime ready Influenza A/H1N1 Detection Set (Roche Diagnostics, Germany). Analytical specificities of the assays were determined by testing commonly encountered respiratory viral pathogens, including parainfluenza virus type 1/2/3, rhinovirus A, rhinovirus B, metapneumovirus, adeno-virus, and coronavirus. The analytical sensitivities and workflow of both methods were also assessed.
Results
A total of 102 consecutive clinical specimens were tested by both methods. Total agreement between the two methods was estimated to be 99.0% (101/102): 11 A/H1N1/2009 and 3 seasonal influenza A by the RealTime ready Influenza A/H1N1 Detection Set; 10 and 3 by Xpert Flu. No cross-reactivity was observed between influenza A/H1N1/2009 and other respiratory viral pathogens in either method. The lim-its of detection of the RealTime ready Influenza A/ H1N1 Detection Set and Xpert Flu were 500 TCID50/ mL and 20 TCID50/mL, respectively. Xpert Flu requi-red 85 minutes (10 minutes of hands-on time) for processing, while RealTime ready Influenza A/ H1N1 Detection Set took 128 minutes (30 minutes of hands- on time).
Conclusion
The results of Xpert Flu were compara-ble to those of the RealTime ready Influenza A/H1N1 Detection Set. It is of note that the fully automated and closed system of Xpert Flu could be advanta-geous for reducing hands-on time and for preventing cross-contamination during the testing process.
REFERENCES
1.Thompson WW., Shay DK., Weintraub E., Brammer L., Cox N., Anderson LJ, et al. Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA. 2003. 289:179–86.

2.Muscatello DJ., Newall AT., Dwyer DE., Macintyre CR. Mortality attributable to seasonal and pandemic influenza, Australia, 2003 to 2009, using a novel time series smoothing approach. PLoS One. 2013. 8:e64734.

3.Yi K., Kang JO., Oh JW., Ham SY., Choi TY. Trends of viral respiratory pathogens detected in pediatric patients, 1996 through 2001. Korean J Clin Microbiol. 2002. 5:77–83.
4.WHO on behalf of the Special Programme for Research and Training in Tropical Diseases. 2010. Use of Influenza Rapid Diagnostic Tests.http://whqlibdoc.who.int/publications/2010/9789241599283_eng.pdf/. [Online] (last visited on 6 August, 2013).
5.Treanor JJ. Influenza Virus, Including Avian Influenza and Swine Influenza. Mandell GL, Bennett JE, Dolin R, editors. Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases. 7th ed. New York: Churchill Livingstone Inc;2010. p. 2265–88.
6.Kwon A., Kim JS., Kim HS., Song W., Park JY., Cho HC, et al. Comparison of rapid antigen test and real-time reverse transcriptase PCR for diagnosing novel swine influenza A (H1N1). Korean J Clin Microbiol. 2010. 13:109–13.

7.Popowitch EB., Rogers E., Miller MB. Retrospective and prospective verification of the Cepheid Xpert influenza virus assay. J Clin Microbiol. 2011. 49:3368–9.

8.Salez N., Ninove L., Thirion L., Gazin C., Zandotti C., de Lamballerie X, et al. Evaluation of the Xpert Flu test and comparison with in-house real-time RT-PCR assays for detection of influenza virus from 2008 to 2011 in Marseille, France. Clin Microbiol Infect. 2012. 18:E81–3.

9.Li M., Brenwald N., Bonigal S., Chana K., Osman H., Oppenheim B. Rapid diagnosis of influenza: an evaluation of two commercially available RT-PCR assays. J Infect. 2012. 65:60–3.

Fig. 1.
Workflow analysis of the RealTime ready Influenza A/H1N1 Detection Set and Xpert Flu systems. A, Transfer nasopharyngeal samples into the cartridges; B, RNA extraction and real time RT-PCR in automated Xpert Flu system; C, results analysis; D, sample preparation for RNA extraction; E, RNA extraction using MagNa Pure LC Total Nucleic Acid Isolation Kit; F, Preparation for real time RT-PCR; G, real time RT-PCR using RealTime ready Influenza A/H1N1 Detection Set, H. results analysis.
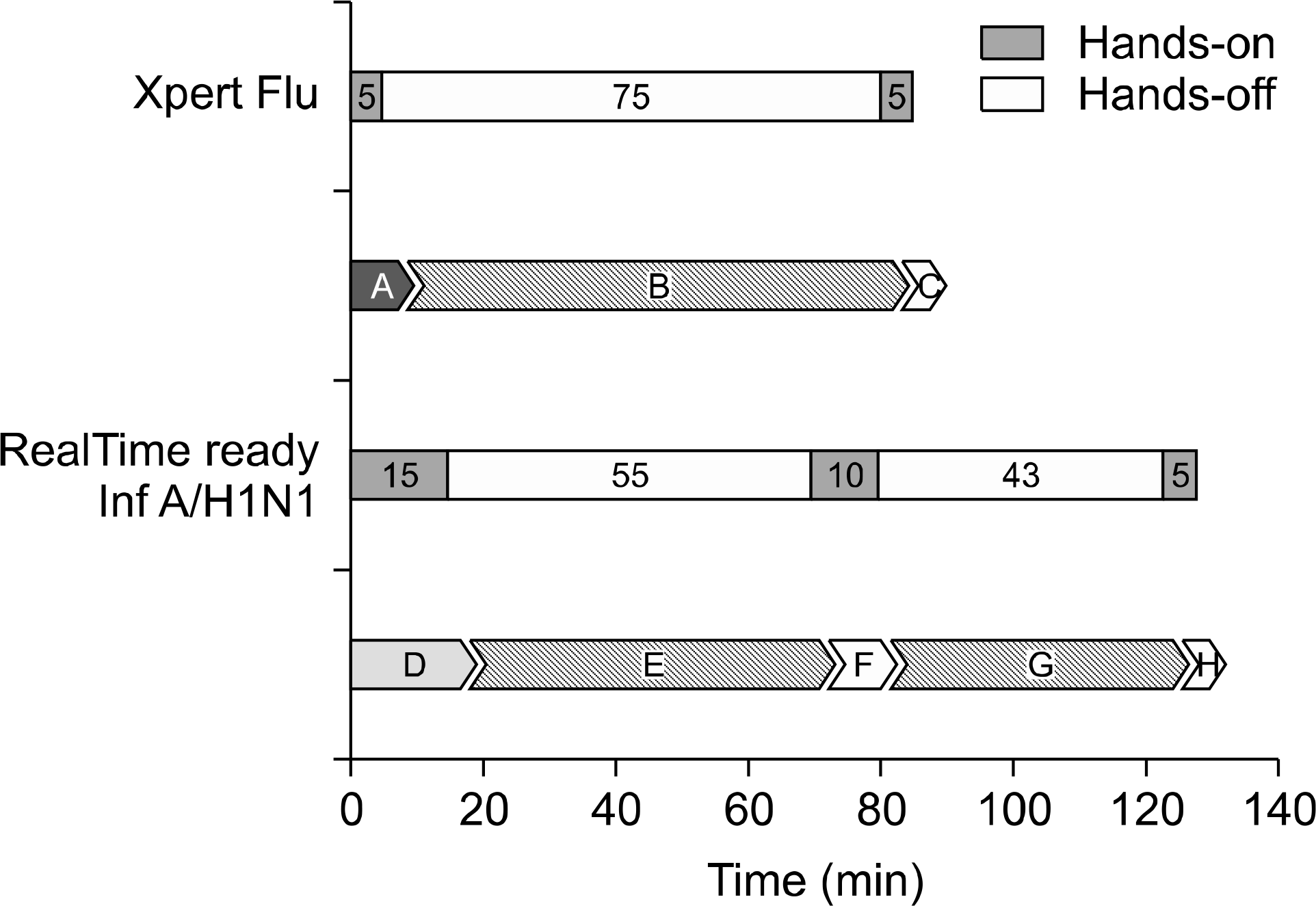
Table 1.
Xpert Flu (Cepheid, Sunnyvale, CA, USA) performance compared to RealTime ready Influenza A/H1N1 Detection Set (Roche Diagnostics, Mannheim, Germany)
| Xpert Flu | RealTime ready Influenza A/H1N1 Detection Set | |||
|---|---|---|---|---|
| Influenza A | A/H1N1/2009 | Negative | Total | |
| Influenza A | 3 | 0 | 0 | 3 |
| A/H1N1/2009 | 0 | 10 | 0 | 10 |
| Negative | 0 | 1 | 88 | 89 |
| Total | 3 | 11 | 88 | 102 |
Table 2.
Analytical sensitivity of RealTime ready Influenza A/H1N1 Detection Set and Xpert Flu
| Conc. (TCID50∗/mL | RealTime ready Influenza A/H1N1 Detection Set (Ct† value for M2 and N1) | Xpert Flu (Ct value for influenza A/H1N1) | ||||
|---|---|---|---|---|---|---|
| )1 st | 2 nd | 3 rd | 1 st | 2 nd | 3 rd | |
| 500 | Positive (34.1, 32.0) | Positive (34.0, 32.3) | Positive (35.7, 35.1) | Not tested | Not tested | Not tested |
| 250 | Positive (36.1, 34.7) | Positive (35.2, 35.0) | Positive (35.7, 34.4) | Not tested | Not tested | Not tested |
| 200 | Positive (37.0, 35.33) | Positive (37.50, 35.61) | Positive (36.97, 34.95) | Not tested | Not tested | Not tested |
| 150 | Negative (-, 36.77) | Positive (34.01, 36.08) | Negative (-, 36.52) | Not tested | Not tested | Not tested |
| 100 | Negative | Negative | Negative | Positive (32.8, 32.8) | Positive (32.4, 32.4) | Positive (33.1, .33.5) |
| 75 | Not tested | Not tested | Not tested | Positive (35.1, 34.4) | Positive (33.5, 33.5) | Positive (33.4, 34.4) |
| 50 | Not tested | Not tested | Not tested | Positive (39.3, 34.7) | Positive (35.4, 35.5) | Positive (35.7, 35.2) |
| 25 | Not tested | Not tested | Not tested | Positive (35.7, 35.6) | Negative (-, 39.9) | Positive (34.5, 35.6) |
| 10 | Negative | Negative | Negative | Positive (35.7, 35.6) | Negative (-, 37.9) | Negative (-, 37.0) |
| 5 | Not tested | Not tested | Not tested | Negative (-, 35.5) | Negative | Negative |
| 1 | Negative | Negative | Negative | Negative | Negative | Negative |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download