1. Stone JH, Zen Y, Deshpande V. IgG4-related disease. N Engl J Med. 2012; 366:539–551. PMID:
22316447.

2. Kamisawa T, Funata N, Hayashi Y, Eishi Y, Koike M, Tsuruta K, et al. A new clinicopathological entity of IgG4-related autoimmune disease. J Gastroenterol. 2003; 38:982–984. PMID:
14614606.

3. Inoue D, Zen Y, Abo H, Gabata T, Demachi H, Kobayashi T, et al. Immunoglobulin G4-related lung disease: CT findings with pathologic correlations. Radiology. 2009; 251:260–270. PMID:
19221056.

4. Hamano H, Kawa S, Horiuchi A, Unno H, Furuya N, Akamatsu T, et al. High serum IgG4 concentrations in patients with sclerosing pancreatitis. N Engl J Med. 2001; 344:732–738. PMID:
11236777.

5. Zen Y. The pathology of IgG4-related disease in the bile duct and pancreas. Semin Liver Dis. 2016; 36:242–256. PMID:
27466794.

6. Kawano M, Yamada K. IgG4-related kidney disease and IgG4-related retroperitoneal fibrosis. Semin Liver Dis. 2016; 36:283–290. PMID:
27466797.

7. Perugino CA, Wallace ZS, Meyersohn N, Oliveira G, Stone JR, Stone JH. Large vessel involvement by IgG4-related disease. Medicine (Baltimore). 2016; 95:e3344. PMID:
27428181.

8. Williams MM, Mashaly H, Puduvalli VK, Jin M, Mendel E. Immunoglobulin G4-related disease mimicking an epidural spinal cord tumor: case report. J Neurosurg Spine. 2017; 26:76–80. PMID:
27517527.

9. Kim JH, Byun JH, Lee SS, Kim HJ, Lee MG. Atypical manifestations of IgG4-related sclerosing disease in the abdomen: imaging findings and pathologic correlations. AJR Am J Roentgenol. 2013; 200:102–112. PMID:
23255748.

10. Kamisawa T, Zen Y, Pillai S, Stone JH. IgG4-related disease. Lancet. 2015; 385:1460–1471. PMID:
25481618.

11. Aalberse RC, Stapel SO, Schuurman J, Rispens T. Immunoglobulin G4: an odd antibody. Clin Exp Allergy. 2009; 39:469–477. PMID:
19222496.

12. Khosroshahi A, Wallace ZS, Crowe JL, Akamizu T, Azumi A, Carruthers MN, et al. International consensus guidance statement on the management and treatment of IgG4-related disease. Arthritis Rheumatol. 2015; 67:1688–1699. PMID:
25809420.

13. Deshpande V, Zen Y, Chan JK, Yi EE, Sato Y, Yoshino T, et al. Consensus statement on the pathology of IgG4-related disease. Mod Pathol. 2012; 25:1181–1192. PMID:
22596100.

14. Inoue D, Yoshida K, Yoneda N, Ozaki K, Matsubara T, Nagai K, et al. IgG4-related disease: dataset of 235 consecutive patients. Medicine (Baltimore). 2015; 94:e680. PMID:
25881845.
15. Vlachou PA, Khalili K, Jang HJ, Fischer S, Hirschfield GM, Kim TK. IgG4-related sclerosing disease: autoimmune pancreatitis and extrapancreatic manifestations. Radiographics. 2011; 31:1379–1402. PMID:
21918050.

16. Takahashi N, Kawashima A, Fletcher JG, Chari ST. Renal involvement in patients with autoimmune pancreatitis: CT and MR imaging findings. Radiology. 2007; 242:791–801. PMID:
17229877.

17. Inoue D, Zen Y, Abo H, Gabata T, Demachi H, Yoshikawa J, et al. Immunoglobulin G4-related periaortitis and periarteritis: CT findings in 17 patients. Radiology. 2011; 261:625–633. PMID:
21803920.

18. Inoue D, Yoneda N, Yoshida K, Nuka H, Kinoshita J, Fushida S, et al. Imaging and pathological features of gastric lesion of immunoglobulin G4-related disease: a case report and review of the recent literature. Mod Rheumatol. 2016; 1–5.

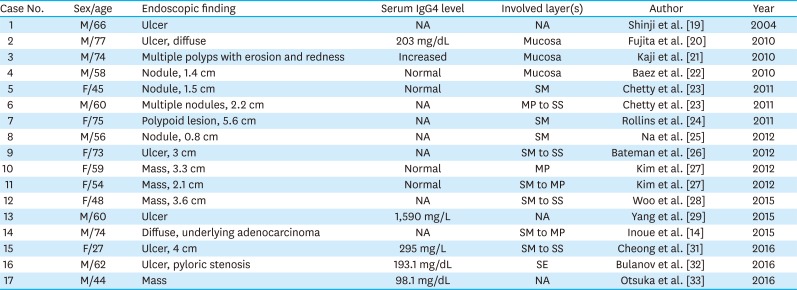
19. Shinji A, Sano K, Hamano H, Unno H, Fukushima M, Nakamura N, et al. Autoimmune pancreatitis is closely associated with gastric ulcer presenting with abundant IgG4-bearing plasma cell infiltration. Gastrointest Endosc. 2004; 59:506–511. PMID:
15044886.

20. Fujita T, Ando T, Sakakibara M, Hosoda W, Goto H. Refractory gastric ulcer with abundant IgG4-positive plasma cell infiltration: a case report. World J Gastroenterol. 2010; 16:2183–2186. PMID:
20440861.

21. Kaji R, Okabe Y, Ishida Y, Takedatsu H, Kawahara A, Aino H, et al. Autoimmune pancreatitis presenting with IgG4-positive multiple gastric polyps. Gastrointest Endosc. 2010; 71:420–422. PMID:
19846081.

22. Baez JC, Hamilton MJ, Bellizzi A, Mortele KJ. Gastric involvement in autoimmune pancreatitis: MDCT and histopathologic features. JOP. 2010; 11:610–613. PMID:
21068496.
23. Chetty R, Serra S, Gauchotte G, Markl B, Agaimy A. Sclerosing nodular lesions of the gastrointestinal tract containing large numbers of IgG4 plasma cells. Pathology. 2011; 43:31–35. PMID:
21240062.

24. Rollins KE, Mehta SP, O'Donovan M, Safranek PM. Gastric IgG4-related autoimmune fibrosclerosing pseudotumour: a novel location. ISRN Gastroenterol. 2011; 2011:873087. PMID:
21991533.

25. Na KY, Sung JY, Jang JY, Lim SJ, Kim GY, Kim YW, et al. Gastric nodular lesion caused by IgG4-related disease. Pathol Int. 2012; 62:716–718. PMID:
23005600.

26. Bateman AC, Sommerlad M, Underwood TJ. Chronic gastric ulceration: a novel manifestation of IgG4-related disease? J Clin Pathol. 2012; 65:569–570. PMID:
22259178.

27. Kim DH, Kim J, Park DH, Lee JH, Choi KD, Lee GH, et al. Immunoglobulin G4-related inflammatory pseudotumor of the stomach. Gastrointest Endosc. 2012; 76:451–452. PMID:
21981816.

28. Woo CG, Yook JH, Kim AY, Kim J. IgG4-related disease presented as a mural mass in the stomach. J Pathol Transl Med. 2016; 50:67–70. PMID:
26420251.

29. Yang L, Jin P, Sheng JQ. Immunoglobulin G4-related disease (IgG4-RD) affecting the esophagus, stomach, and liver. Endoscopy. 2015; 47(Suppl 1 UCTN):E96–E97. PMID:
25734809.

30. Inoue K, Okubo T, Kato T, Shimamura K, Sugita T, Kubota M, et al. IgG4-related stomach muscle lesion with a renal pseudotumor and multiple renal rim-like lesions: a rare manifestation of IgG4-related disease. Mod Rheumatol. 2018; 28:188–192. PMID:
26381653.

31. Cheong HR, Lee BE, Song GA, Kim GH, An SG, Lim W. Immunoglobulin G4-related inflammatory pseudotumor presenting as a solitary mass in the stomach. Clin Endosc. 2016; 49:197–201. PMID:
26867551.

32. Bulanov D, Arabadzhieva E, Bonev S, Yonkov A, Kyoseva D, Dikov T, et al. A rare case of IgG4-related disease: a gastric mass, associated with regional lymphadenopathy. BMC Surg. 2016; 16:37. PMID:
27255154.

33. Otsuka R, Kano M, Hayashi H, Hanari N, Gunji H, Hayano K, et al. Probable IgG4-related sclerosing disease presenting as a gastric submucosal tumor with an intense tracer uptake on PET/CT: a case report. Surg Case Rep. 2016; 2:33. PMID:
27059471.

34. Søreide K, Sandvik OM, Søreide JA, Giljaca V, Jureckova A, Bulusu VR. Global epidemiology of gastrointestinal stromal tumours (GIST): a systematic review of population-based cohort studies. Cancer Epidemiol. 2016; 40:39–46. PMID:
26618334.

35. Ko SY, Lee JS, Kim JJ, Park SM. Higher incidence of gastroesophageal reflux disease after gastric wedge resections of gastric submucosal tumors located close to the gastroesophageal junction. Ann Surg Treat Res. 2014; 86:289–294. PMID:
24949319.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download