Abstract
Objectives
Several researchers have shown that three dimensional (3D) distribution analysis of prostate cancer is helpful when initiating needle biopsy procedures. Knowledge regarding the distribution of prostate cancer could enhance understanding of the pathophysiology involved and improve detection of these malignancies. We propose utilizing digital processing techniques to analyze prostate cancer distribution in a 3D setting.
Methods
Pre-made radical prostatectomy sample slices were digitized with a resolution of 76 dpi. Slices of each sample were aligned and registered by deformation algorithm and interpolated for analysis of relative distribution statistics. We analyzed 80 samples saved in electronic medical record and compared the detection rate of preoperative needle biopsies and radical prostatectomies using our 3D analysis technique.
Results
The statistical 3D distribution of prostate cancer was evaluated using a 36-sector process. Results were represented in the following two ways: distribution of a single patient, and statistical distribution of prostate cancers of multiple patients. The overall concordance rate was 62.7% between the two methods; therefore a technique is needed which can raise this percentage.
Conclusions
We suggest using the normalization method to develop a software tool which permits reconstruction of the 3D distribution of prostate cancer from 2D legacy images and reduces the loss of image quality as well. This application will facilitate detection of prostate cancer by aiding in the determination of the most effective clinical position via partial sampling with decreased patient inconvenience.
As increase of old age population, prostate cancer is more important cancer increasingly in Korea. According to report of Korea central cancer registry, prostate cancer has increased up to 270% between 1999-2008 [1]. It shows a highest increasing rate than other cancers.
The sextant biopsy technique has been used widely with success in diagnosing prostate cancer. However concern has arisen that the original sextant method may not include an adequate sampling of the prostate. Complete sampling of the entire prostate which has increased prostate specific antigen (PSA) level most accurately provides prognostic information. However, it demands time, cost, and burden of patients. Therefore, most pathologists adopt various partial sampling methods [2].
A partial sampling method has different probability depends on sites of biopsy. There are some partial sampling methods for improve detecting prostate cancer. Zeng et al. [3] predicted number of biopsies and clinical positions for accurate diagnosis through modeling three dimensional (3D) prostate and simulation. Egevad et al. [4] developed for three dimensional modeling of prostate cancer and transrectal biopsy. It validated 10 standardized virtual biopsies, 24% would have remained undetected with sextant biopsies.
As result of these studies shows the distribution of prostate cancer would be very useful for increasing probability of diagnosis prostate cancer [3-6]. These studies not only present general biopsy protocols but also develop three dimensional tools for showing cancer distribution of prostates using various and useful computer algorithms.
In this study, we present a tool for distribution of prostate cancer in Korean from 2D legacy images, this would be helpful to detect prostate cancer accurately. As technical point of view, Kang et al. [7] does not defined with different size of prostate, we enhanced that reconstructing the 3D distribution of prostate cancer using normalization method. Therefore we presented distribution of prostate cancers regardless of their size.
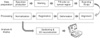
Figure 1 shows the concept in order for this experiment. Each radical prostatectomy specimen was measured, weighed, and fixed in 10% buffered neutral formalin. All prostates were processed using the complete sampling method. In brief, after application of four different color inks on the surface of the right, left, anterior, and posterior prostate. The basal portion of the prostate was shaved using a transverse cut. The apical portion was amputated 5 mm from the apical margin in all cases, thus generating a cone-shaped specimen which was sliced serially at 3-5 mm intervals perpendicular to the prostatic urethra. To generate the tumor map, the slices were arranged orderly and their outline, direction, prostatic urethra, any suspicious area of tumor, and cassette number were then recorded on an overlain overhead projector film. Each slice was embedded in one cassette or divided into 2-4 cassettes, according to its size. Both shaved basal and amputated apical prostates were submitted separately. We scanned each slice as 76 dpi resolution and it used as input data for analysis software of cancer distribution. The scanning resolution is determined by the redrawing size of the specimen on A4 paper and the appropriate resolution of 640 pixels (horizontal) on computer processing.
It is difficult to know the standard distribution because of discordances in size and shape between samples. We should comply shape and size of samples to analysis the distribution of prostate cancers in objectively and quantitatively. We call this procedure 'Registration.' This procedure is composed of 'Alignment,' 'Deformation,' and 'Normalization.'
It is difficult to align slices when they were stacked for reconstructing the 3D distribution without standard position. One of solution is marking out two or more point through the needle. We marked two points on the sliced prostate sample with needle. These two marks make reference points to align sample itself. But on older samples does not have these markers. In this case we apply next deformation procedure.
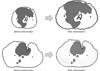
In case of multiple analysis module requires consistent distribution even if prostate size are different. However, there is no reference in early studies [7-12] thus we defined our own normalization method for prostate cancer distribution of group patients. So we need some standardization procedure on sample slices. We call this procedure a 'Deformation.' First of all, we made a standard template of a prostate slice. We got an image of prostate slice from textbook and post-process it. And then we smooth the edge of sample. We adapt the polar coordinate. Then the pixel movement of sample in outer direction is,
Figure 2 shows the standard template and the deformation procedure. Where PθL denotes the pixel position of the smoothed sample outline in the direction of θ and length L. Fθ denotes the difference between sample outline and template outline in the direction of θ. And Pθi denotes the inner pixel position of sample in the direction of θ and length i. First, the sample edge is smoothed and then deformed to template outline.
After this pixel movement procedure we restore the rectangular coordinate. And then apply tri-linear interpolation algorithm to fill holes between sparsely relocated pixels.
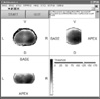
When we make a digital sample from legacy images, there was 4 mm space between slices. This is a main reason of reducing quality of images. In this study, we suggested the normalization method which is a digital sampling method such as Figure 3. For example, if number of slices is decreased than average number of slice, we make increased number of pixels such as (a) in Figure 3. On the other hand, number of slices is increased, decreased number of pixels like (c) in Figure 3. After normalization method, we carry out trilinear interpolation [13] for improve to image quality. Figure 7 shows the results which are normalized distribution of 148 patient's specimens.
A number of needle biopsies are changeable at the partial sampling. Generally, 10-12 preoperative needle biopsies are preferable, more than 20 times as the case may be [14]. The prostate was divided into 36 parts by dividing the prostate into anterior (A) and posterior (P) potions and into lateral (L), middle (M), central (C) potions of the right and left lobes horizontally and into upper (U), middle (M), lower (L) levels vertically (Figure 4). The tumor size and volume were measured by the computer program according to the tumor positive area among the 36 parts of the prostate.
In this study, LabVIEW 8.2 (National Instruments, Austin, TX, USA) was used a programming language and hardware environment consisted of Pentium D CPU 3.0 GHz PC using Windows XP as the operating system.
Figure 5 illustrates the deformation results. Sample slices are deformed to template shape.
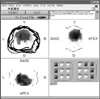
Figure 6 illustrates distribution of single sample module. Each image consists of slices with 4 mm interval, and they are piled up one on another. All images represent X-Y, Y-Z, X-Z plane and the direction indicate with single characters such as ventral (V), dorsal (D), left (L), right (R). The digital image of each case showed boundaries of the prostate and tumor. The color of intensity graph means accumulated density of prostate cancer probability.
In the multiple analysis module processes the normalization method and interpolation to the group have different size of prostate. Figure 7 shows 148 samples that are merged into one average size of prostate volume. In case of multiple analysis module has a threshold valued boundaries of prostate and tumor. Because if does not the threshold value, all images would be filled with boundaries. The threshold value is also variable so we could modulate the thickness of boundaries.
This study initially included 190 consecutive prostate cancer patients who received radical prostatectomy at Asan Medical Center from January 1 to December 31, 2006. Among these patients, 42 were excluded; 28 patients were excluded because of no glass slides or tumor maps and 14 patients undergone preoperative hormonal therapy. A total 148 patients were therefore included in this study. Clinical information and the results of digital rectal examination and radiological studies were obtained from the patients' electronic medical records. Table 1 shows comparison between preoperative needle biopsy and radical prostatectomy specimens. On the preoperative needle biopsies, the right and left lobes showed tumor positivity in 57 cases (71.3%) and 50 cases (62.5%), respectively. On the resected prostates, the right and left lobes showed tumor positivity in 73 cases (91.3%) and 65 cases (81.3%), respectively. The mean overall concordance rate between preoperative needle biopsy and radical prostatectomy specimens, was 62.7% (Table 2). As the tumor was more frequent in the right side than in the left side of both needle biopsy and prostatectomy specimens, the concordance rate was slightly higher for the right side (67.1%) than for the left side (58.3%). Needle biopsy revealed high specificity (82.4%) but relatively low sensitivity (50.7%) for the regional detection of prostate cancer, with 82.5% and 50.5% positive and negative predictive values, respectively. These results show that it needed improved protocol for cancer detection rate by biopsy.
Table 2 presents 36 section distribution rates in this study which has 80 samples. We expect this result would be helpful to build a new method for improvement of preoperative needle biopsy afterward.
The knowledge of distribution of prostate cancer could be helpful for planning preoperative biopsy and well treatment to the patients. The importance of this issue emphasized in several researches. In 1988, McNeal et al. [15] suggested zonal distribution of prostatic adenocarcinoma. In 2000, Chen et al. [16], developed computer based algorithm for distribution of prostate cancer about 180 patients who received radical prostatectomy. But in that study, they just standardize the width (x) and height (y) at a given length (z). They insert additional virtual slices for a total 19 slices. In the contrary, we adopt deformation and normalization algorithms.
In case of compare with westerner, Korean has less prevalence, volume, and also characteristics different depends on races. Consequently, in 2006 Kang et al. [7], they made a Korean patients oriented prostate tumor map through 186 patients. However, they only suggested distribution of prostate cancer about similar size. In this study, we defined the normalization method which applicable every size of prostates into distribution analysis objectively. Especially, this method is useful for converting legacy 2D images to 3D views, as well as analysis of approximate prostate volume.
In this study, we considered this application will not only help to find effective clinical position by partial sampling that detecting prostate cancers but also release burden of patients.
Figures and Tables
Figure 3
Normalization of samples which have different specimen sizes. Sample (A) has less slices for normal. So the thickness is increased to 21 pixels for each slice. (C) Sample has more slices for normal. So the thickness is reduced to 7 pixels for each slice.

References
1. Korea Central Cancer Registry. Annual report of cancer statistics in Korean in 2008. 2011. 1st ed. Goyang: Korea Central Cancer Registry;24.
2. True LD. Surgical pathology examination of the prostate gland. Practice survey by American society of clinical pathologists. Am J Clin Pathol. 1994. 102:572–579.

3. Zeng J, Bauer JJ, Mun SK. Modeling and mapping of prostate cancer. Comput Graph. 2000. 24:683–694.

4. Egevad L, Frimmel H, Norberg M, Mattson S, Carlbom I, Bengtsson E, Busch C. Three-dimensional computer reconstruction of prostate cancer from radical prostatectomy specimens: evaluation of the model by core biopsy simulation. Urology. 1999. 53:192–198.

6. Mazzucchelli R, Santinelli A, Lopez-Beltran A, Scarpelli M, Montironi R. Evaluation of prognostic factors in radical prostatectomy specimens with cancer. Urol Int. 2002. 68:209–215.

7. Kang T, Song C, Song GH, Shin GH, Shin DI, Kim CS, Ahn H. The anatomic distribution and pathological characteristics of prostate cancer: a mapping analysis. Korean J Urol. 2006. 47:578–585.

8. Frimmel H, Egevad L, Bengtsson E, Busch C. Modeling prostate cancer distributions. Urology. 1999. 54:1028–1034.

9. Song C, Kim J, Chung H, Kim CS, Ro JY, Ahn HJ. Nomograms for the prediction of the pathological stage of the clinically localized prostate cancer in Korean men. Korean J Urol. 2003. 44:753–758.
10. Lee SE, Kim DY, Kwak C. Interrelationship among age, prostate specific antigen and prostate volume in Korean men living at the metropolitan area. Korean J Urol. 1999. 40:1311–1317.
11. Hodge KK, McNeal JE, Terris MK, Stamey TA. Random systematic versus directed ultrasound guided transrectal core biopsies of the prostate. J Urol. 1989. 142:71–74.

12. Vachani C. Understanding your pathology report: prostate cancer. cited at 2010 Dec 22. Available from: http://www.oncolink.org/types/article.cfm?c=16&s=57&ss=452&id=9597.
13. Goshtasby A, Turner DA, Ackerman LV. Matching of tomographic slices for interpolation. IEEE Trans Med Imaging. 1992. 11:507–516.

14. Mazzucchelli R, Santinelli A, Lopez-Beltran A, Scarpelli M, Montironi R. Evaluation of prognostic factors in radical prostatectomy specimens with cancer. Urol Int. 2002. 68:209–215.

15. McNeal JE, Redwine EA, Freiha FS, Stamey TA. Zonal distribution of prostatic adenocarcinoma. Correlation with histologic pattern and direction of spread. Am J Surg Pathol. 1988. 12:897–906.
16. Chen ME, Johnston DA, Tang K, Babaian RJ, Troncoso P. Detailed mapping of prostate carcinoma foci: biopsy strategy implications. Cancer. 2000. 89:1800–1809.




 PDF
PDF ePub
ePub Citation
Citation Print
Print






 XML Download
XML Download