Abstract
An anomalous aortic origin of a coronary artery is rare and surgical intervention is recommended when the patient is symptomatic. We performed coronary artery bypass graft surgery in a 21-year-old male patient with a right coronary artery anomalously originating from the left coronary sinus. The artery was significantly stenosed by external compression between the aorta and the pulmonary artery. However, the graft became occluded 1 year after the operation. In such cases, the dynamic nature of the stenosis can cause relatively intact antegrade competitive flow from the native coronary artery and lead to an occlusion of the grafted artery. Methods for evaluating flow rates or intraluminal pressures of native arteries could be helpful in decision-making in similar cases.
An anomalous aortic origin of a coronary artery is a rare congenital anomaly with an incidence of about 0.17%.1 Such anomalies are reported as an important cause of sudden death in specific populations such as athletes or military recruits.2,3 Generally, surgical interventions like coronary artery bypass grafting (CABG), reimplantation, or unroofing are recommended when the patient has symptoms of ischemia or arrhythmia.4 Here we report a case of failed CABG of an anomalously originating right coronary artery (RCA) in a young and symptomatic patient.
A 21-year-old male soldier visited the division of cardiology of our medical center with the complaint of effort angina. He had no history of smoking and no family history of heart disease. His vital signs were stable, his electrocardiogram showed a normal sinus rhythm, and the results of baseline laboratory findings were nonspecific. However, owing to the typical nature of his chest pain, which suggested an ischemic origin, we decided to perform cardiac computed tomography angiography (CTA) to rule out coronary artery diseases. The cardiac CTA images showed normal left coronary arteries but an RCA anomalously originating from the left sinus of Valsalva. Also, the proximal RCA had an interarterial course, which meant that the RCA was trapped between the aorta and the pulmonary artery (Fig. 1).
We performed coronary angiography (CAG) to verify the anomaly. CAG from the left sinus of Valsalva showed all 3 coronary arteries, suggesting the anomalously originating RCA (Fig. 2). CAG from the right coronary sinus showed no significant stenosis in the left anterior oblique view. In the right anterior oblique view, however, significant stenosis was noted in the proximal RCA as the result of external compression (Fig. 3). Also during CAG, an intravascular ultrasonography (IVUS) study of the RCA showed that the minimum lumen area in the proximal RCA was 3.8 mm2, which suggested significant stenosis. After CAG, we could not proceed to additional diagnostic tests to verify the evidence of ischemia in the RCA territory, such as myocardial single photon emission computed tomography (M-SPECT) or fractional flow reserve (FFR), because the patient was on a mandatory military service and did not have enough time for many diagnostic tests. However, given the findings from the CAG and IVUS, we regarded the chest pain to be originating from ischemia of the RCA territory. We therefore decided to perform surgical intervention. Owing to a slit-like stenosis of the RCA ostium, we could not choose reimplantation of the RCA ostium, and surgical unroofing was not favored because of a concern for persistent myocardial ischemia in such cases.5 Finally, CABG [right internal mammary artery (RIMA) to RCA] was selected as the surgical intervention. After the CABG, the patient's symptom was relieved, which suggested that the effort angina was caused by myocardial ischemia. A follow-up CAG done 1 week after the operation showed a patent RIMA to RCA graft (Fig. 4). The patient was discharged from our hospital with medications including aspirin.
The patient had no chest pain for more than 10 months after the operation. However, nearly 1 year after the operation, he visited our center and reported suffering from chest pain for about 2 weeks. We recommended that he undergo CAG again. The follow-up CAG revealed no interval changes of the 3 native coronary arteries, but the RIMA to RCA graft was totally occluded (Fig. 5). The right subclavian artery was intact and the patient had no other risk factors for atherosclerosis. Thus, the total occlusion seemed to have developed as the result of complete inhibition by good antegrade flow of the RCA. At this point, we decided to control the patient's symptom with medical treatment, and we recommended that he undergo reimplantation of the right coronary ostium if his symptom persisted after optimal medical treatment.
Graft patency of the internal mammary artery is generally excellent, with a 5-year patency rate of more than 90% and a 10-year patency rate of about 80-95%.6 It is well known that the degree of stenosis in the native vessel is a major predictor of internal mammary artery bypass graft patency.7 In the present case, however, the patient's RCA was significantly stenosed, but the graft became occluded. In fact, the degree of stenosis in the anomalously originating RCA may have changed, because the diameter of the 2 major vessels may vary according to sympathetic and parasympathetic tone. Thus, the degree of antegrade flow through the native RCA, and, consequently, the degree of competitive flow, can also vary. This suggests that we should be particularly careful when choosing a surgical intervention for an anomalously originating RCA with an interarterial course. If we could measure flow rates or intraluminal pressures of native arteries and estimate the intensity of possible competitive antegrade flow through the native coronary arteries, we could avoid CABG surgeries in patients who have higher risks of graft occlusion by competitive flows. The use of methods such as FFR for such purposes should be investigated.
One question arises from this case. Did this patient really have to undergo a surgical intervention? Despite the lack of other evidence of myocardial ischemia, such as results from a treadmill test, M-SPECT, or FFR, the patient's chest pain was present with a significantly stenosed RCA, which was relieved by CABG and unfortunately redeveloped with occlusion of the graft. This suggests that the symptom was caused by myocardial ischemia. However, in one report, no significant difference in 10-year survival was seen in patients with an anomalously originating coronary artery from the opposite sinus of Valsalva, which had an interarterial course, after a surgical intervention.8 Of course, this does not mean that a surgical intervention is useless for such cases. The efficacy of surgical intervention versus medical treatment for reducing symptoms is not yet validated in such cases, and this remains a question that should be investigated.
In summary, physicians should think twice before performing a surgical intervention in patients who have an anomalously originating coronary artery with an interarterial course, and methods to evaluate flow rates or the intraluminal pressures of native arteries could be helpful in making a decision. It should be investigated whether such methods would really be helpful, and the treatment efficacy of surgical intervention should also be evaluated.
Figures and Tables
FIG. 1
Cardiac Computed Tomography Angiography. The right coronary artery is originated from the left coronary sinus (blue long arrow) and was trapped between the aorta (red short arrow) and the pulmonary artery (yellow arrowhead).

FIG. 2
Coronary angiography showed that both the right coronary artery (blue long arrow) and the left coronary arteries (red short arrows) originated from the left coronary sinus simultaneously, suggesting an anomalously originating RCA (left image). The left coronary arteries are normal (right image).
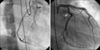
FIG. 3
The right coronary artery seems to be normal in the left anterior oblique view (left image), but in the right anterior oblique view, significant stenosis in the proximal RCA due to external compression is shown (right image, blue arrow).

ACKNOWLEDGEMENTS
This study was supported by a grant of the National Research Foundation of Korea Grant funded by the Korean Government (2011-0008875), and a grant of the Korean Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (HI13C0163), and the Bio & Medical Technology Development Program of the National Research Foundation (NRF) funded by the Korean government (MEST) (2012M3A9C6049744), and the Korea Healthcare technology R&D Project, Ministry for Health, Welfare and Family Affairs (HI12C0275), and a grant of the Korean Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (HI13C1527), and Chonnam National University Hospital Research Institute of Clinical Medicine (CRI 11080-21), Republic of Korea.
References
1. Davis JA, Cecchin F, Jones TK, Portman MA. Major coronary artery anomalies in a pediatric population: incidence and clinical importance. J Am Coll Cardiol. 2001; 37:593–597.

2. Maron BJ, Carney KP, Lever HM, Lewis JF, Barac I, Casey SA, et al. Relationship of race to sudden cardiac death in competitive athletes with hypertrophic cardiomyopathy. J Am Coll Cardiol. 2003; 41:974–980.

3. Eckart RE, Scoville SL, Campbell CL, Shry EA, Stajduhar KC, Potter RN, et al. Sudden death in young adults: a 25-year review of autopsies in military recruits. Ann Intern Med. 2004; 141:829–834.

4. Gersony WM. Management of anomalous coronary artery from the contralateral coronary sinus. J Am Coll Cardiol. 2007; 50:2083–2084.

5. Brothers JA, McBride MG, Seliem MA, Marino BS, Tomlinson RS, Pampaloni MH, et al. Evaluation of myocardial ischemia after surgical repair of anomalous aortic origin of a coronary artery in a series of pediatric patients. J Am Coll Cardiol. 2007; 50:2078–2082.

6. Tatoulis J, Buxton BF, Fuller JA. Patencies of 2127 arterial to coronary conduits over 15 years. Ann Thorac Surg. 2004; 77:93–101.

7. Berger A, MacCarthy PA, Siebert U, Carlier S, Wijns W, Heyndrickx G, et al. Long-term patency of internal mammary artery bypass grafts: relationship with preoperative severity of the native coronary artery stenosis. Circulation. 2004; 110:11 Suppl 1. II36–II40.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download