INTRODUCTION
Primary cardiac lymphoma (PCL) is a very rare neoplasm originating in the heart. Traditionally, PCL is defined as an extranodal lymphoma limited to the heart or pericardium. However, some previous reports demonstrated PCL cases with extracardiac involvement.1,2,3 PCL occupies 1.3% of primary cardiac tumors and 0.5% of extranodal lymphomas.4 Patients with PCL generally complain of nonspecific symptoms such as dyspnea, edema, and arrhythmia.5,6 Treatment of PCL is variable because the optimal treatment strategy has not yet been established. However, previous studies have shown that chemotherapy is the most effective therapy.5,7
In this report, we delineate a case of diffuse infiltrative type of PCL with spread to the mediastinum and cervical lymph nodes.
CASE REPORT
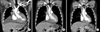
A 48-year-old man visiting the Korea University Ansan Hospital presented with the symptoms of exertional dyspnea and cough. He had no specific past history or family history. Physical examination showed no lymph node enlargement. Chest radiography showed pleural effusion, pulmonary congestion, and cardiomegaly. In a transthoracic two-dimensional echocardiography, a large amount of pericardial effusion and diffuse cardiac wall thickening were observed (Fig. 1A). Cytologic analysis of the pleural fluid showed scattered lymphocytes only. We performed cardiac magnetic resonance imaging (MRI) for differential diagnosis of myocardial infiltrative diseases such as cardiac amyloidosis. MRI showed diffuse cardiac wall thickening and delayed enhancement in all chambers (Fig. 1B). However, there was no lymphadenopathy or a mass lesion in the MRI images (Fig. 1C). The patient underwent a myocardial and pericardial biopsy. However, the myocardial biopsy showed myocyte hypertrophy, and the pericardial biopsy showed chronic inflammation with fibrosis and the Congo red staining was negative. We could not identify a definite cause of the pericardial effusion; hence, the patient underwent conservative treatment and received outpatient follow-up.
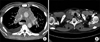
After 10 months, he was readmitted for recurrent massive pericardial effusion. Chest computed tomography (CT) revealed an infiltrative soft tissue mass in the anterior mediastinum and diffuse thickening of all ventricular and atrial walls (Fig. 2A). We suspected the mediastinal mass to be a lymphoma and performed a biopsy. The biopsy results of the mediastinal mass showed chronic inflammation with fibrosis. Immunohistochemical staining was positive for CD45, but owing to the lack of specimen tissue, we could not perform more tests. We considered repeating the biopsy by performing a thoracostomy. However, the patient refused to undergo further evaluation and aggressive management.
Clinically, we suspected a diagnosis of fibrosing mediastinitis, and glucocorticoids were used empirically. We prescribed prednisolone 60 mg daily for 1 month, which was tapered to 15 mg per day within 5 months. After tapering of prednisolone, we maintained prednisolone 15 mg daily.
Ten months later, the patient complained of a nontender mass on the right side of the neck (Fig. 2B). We performed an ultrasound-guided neck mass biopsy. Microscopic examination revealed a diffuse proliferation of large atypical lymphoid cells. Immunohistochemistry demonstrated a large lymphoid neoplasm, which was positive for CD45, CD79a, and CD20. Finally, the patient was diagnosed with primary cardiac diffuse large B cell lymphoma (DLBCL). He commenced chemotherapy with rituximab and CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone). After the second cycle of chemotherapy, we performed chest CT. In the chest CT, the infiltrative lesion, involving the myocardium and multiple lymph nodes, was decreased (Fig. 3B) compared with the chest CT performed before the R-CHOP therapy (Fig. 3B). On the basis of this result, we are confident that the DLBCL originated in the heart and the final diagnosis was PCL.
After 6 cycles of chemotherapy, chest CT and positron emission tomography scans were performed to assess the response to treatment. Imaging studies showed an improvement in the lesions, including the cardiac manifestations. However, the cardiophrenic angle lymph nodes were persistent (Fig. 3C). He commenced radiation therapy (total 40 Gy) for the cardiophrenic angle lymph nodes. After radiation therapy, the lymph node size was slightly decreased.
DISCUSSION
PCL is very rare and makes up only 0.5% of extranodal lymphomas. It is defined as an extranodal lymphoma limited to the heart or pericardium.4,5 Histologically, diffuse large B cell type of non-Hodgkin's lymphoma is the most common of the PCLs, and its incidence ranges from 60% to 81%.6,8,9 In previous reports, most of the cases showed a mass-like lesion.6 The right atrium is the most common site of invasion, followed by the right ventricle, left ventricle, and left atrium.7 Patients commonly present with nonspecific, different symptoms; and clinical findings such as dyspnea, arrhythmia, pericardial effusion, and congestive heart failure are observed.5,6 Because of its nonspecific clinical manifestation, it is difficult to diagnose PCL. Imaging studies such as echocardiography, CT, and MRI are helpful for the diagnosis of PCL. However, pathologic examination is the only way to obtain a definite diagnosis.10 Chemotherapy is effective for treatment of any subtype of primary cardiac lymphoma. A previous report demonstrated that rituximab in combination with the CHOP regimen has a good outcome in the DLBCL type of PCL.1,9,10 Petric et al.5 stated that the median overall survival after diagnosis was 12 months. Among the patients, the chemotherapy group had a longer overall survival up to 30 months.5 However, the optimal treatment strategy has not yet been established.
There are some interesting points about this case. First, the PCL was observed as thickened myocardium without an intracardiac mass. To our knowledge, this is the second case report of a diffuse infiltrative PCL without an intracardiac mass.11 Second, in this case, the DLBCL originated in the heart and delayed extracardiac involvement was noted. Three PCL cases with extracardiac involvement have previously been reported. Two cases presented with an extracardiac metastasis at the first diagnosis.1,2 One case had a PCL involving only the heart at the primary diagnosis, but microscopic involvement of the lungs, peritoneal, and retroperitoneal adipose tissue was detected on autopsy.3 However, these cases did not clearly show delayed metastasis of the PCL to an extracardiac site. Our case clearly showed that the PCL had no extracardiac involvement initially but afterwards showed extracardiac involvement. Mediastinal lymph node biopsy was performed at the second admission, and lymphoma was suspected. However, because of insufficient specimens and refusal of the patient to undergo further testing, we could not confirm the diagnosis of lymphoma, and we administered steroids for a possible diagnosis of fibrosing mediastinitis. We think that the use of steroids led to an immunocompromised state, and this state may have contributed to the delayed extracardiac involvement. It is well known that steroids have a lympholytic effect, and this effect probably caused a disturbance of the detection time from the symptom onset to the administration of proper treatment. Although this is only a hypothesis, clinicians should be aware of the possibility of delayed diagnosis of a PCL owing to the use of steroids.
Despite these interesting points, this report had one large limitation. Because we failed to confirm the diagnosis of PCL from previous cardiac and mediastinal biopsies, the tissues from the enlarged lymph node in the right neck were used to confirm the diagnosis of PCL. Although the biopsy was not performed at the primary site of lymphoma, given that the initial cardiac MRI demonstrated diffuse cardiac infiltration without lymph node enlargements, it appears proper to diagnose PCL.
In summary, we have reported a case of an infiltrative type of PCL without an intracardiac mass and with delayed extracardiac involvement.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download