Introduction
Smell is very important to cats in terms of their communications with each other and they have a larger proportion of their brain devoted to dealing with smells than we do. Odors are detected in the majority of mammals by means of two systems, the main olfactory system and the vomerosensory system (VNS) [1]. The VNS is now widely considered as a secondary olfactory system that mediates a variety of interesting animal behavior, ranging from mating to attack-fighting responses [1 2]. Basically the vomeronasal organ (VNO) pumps liquids and gases in and out of the vomeronasal duct (VND) as has been demonstrated experimentally in hamsters [3]. Some reports could be found on the developing rat [4567], mice [8], rabbit [9], Golden hamster [10], and Syrian hamster [11]. However, a scant attention has been paid to the morphology of the cat VNO which is considered to be of biomedical importance. Cats may harbour many parasites potentially transmissible to humans, which may represent a health risk, especially to children, the elderly and the immunocompromised [12].
The study of Salazar et al. [13] dealt only with the general morphology of the VNO in adult cats; whereas Salazar and Sanchez-Quinteiro [14] reported organization of the VNS of fetal, new born and adult cats. To the best of the author knowledge, no reports are available on the development of the VNO in cat. Therefore, this work was designed to describe the postnatal development of the non-sensory epithelium (NSE) lining the cat VND during the period from birth to the weaning (8 weeks). Using various histological techniques, detailed study of the morphology of the VNO would be of value for elucidating the function of the VNO. Besides, providing sequential description and of a new ultrastructural and histochemical data may be useful for future experimental studies.
Materials and Methods
Fourteen juvenile female cats of four age groups (new born, 2, 4, and 8 weeks) were used. Animal care and use was in accordance with procedures outlined in the National Institutes of Health Guidelines. The experiment was approved by the Institutional Ethics Committee of Assiut University. The cats were anesthetized with ether and their thorax opened to expose the heart, which was used to perfuse the appropriate fixative.
Light microscopy
The cats (two from each age group) were perfused intracardially with 10% formaldehyde solution. After perfusion, the rostral part of the skull containing the intact nasal cavity was removed and immersed into 10% formaldehyde to continue fixation 2 more days. Next, the specimens were decalcified in 13% formic acid for 3–7 days [15]. Then, the decalcified specimens were processed for the preparation of paraffin blocks. Paraffin sections (5–7 µm) were cut in a transverse plane, mounted on glass slides, and every 10th section was stained with hematoxylin and eosin stain. In addition, selected sections were processed for histochemical demonstration of polysaccharides using Alcian blue (AB) at pH 2.5 for acid mucosubstances and periodic acid Schiff (PAS) method for neutral mucosubstances. Orcein stain was used for elastic fibers and Masson's trichrome for collagen fibers. Processing and staining techniques were performed according to Drury and Wallington [15].
Electron microscopy
The cats (two from each age group) were perfused intracardially with 4% glutaraldehyde in cacodylate buffer (pH 7.4). After perfusion, the VNOs in both sides were dissected free. One middle segment (MS) was used for transmission electron microscopy (TEM) and the other used for scanning electron microscopy (SEM).
Transmission electron microscope
The specimens were immersed in glutaraldehyde cacodylate fixative for 24 hours and postfixed in 1% osmium tetroxide in phosphate buffer for 2 hours. Tissues were rinsed in the same buffer, dehydrated with alcohol, cleared with propylene oxide and embedded in Epon 812 (SPI-Pon Araldite Kit Cat. no. 02635-AB., SPI Supplies, West Chester, PA, USA). Semithin sections (0.5–1 µm) were cut and stained with toluidine blue (TB) [16] for examination on a light microscope. Ultrathin sections (500–800 A) were cut from selected areas of the blocks and contrasted with uranyl acetate and lead citrate [17]. These sections were observed with the TEM (Jeol E.M.-100 CX11, Japanese Electron Optic Laboratory, Tokyo, Japan) and photographed at 80 kV.
Scanning electron microscope
After dissection, the lateral side (portion lined by NSE) of the middle segment remaining VNO was obtained by cutting the tissue along the longitudinal axis under a dissecting microscope and the NSE portions were immersed in the fixative for several hours. A graded series of alcohols was used for dehydration, and liquid carbon dioxide was used to dry the specimen. Dried specimens were mounted on aluminum stubs, fixed in place with colloidal silver and sputter coated with gold [18]. A Jeol (J.S.M-5400 LV, Japanese Electron Optic Laboratory) was used to view the specimens. Photographs were taken at 15 kV.
Morphometry and statistical analysis
Using computerized assisted image analysis, the thickness of the NSE in micrometers (µm) was measured by measuring the distance from basement membrane to the free surface using the arbitrary distance method. The measurement was taken viewing each 10th hematoxylin and eosin–stained section using a 10× objective lens (three measurements were taken along the length of the NSE/field and the mean value of NSE thickness was calculated [19]. The morphometric data of each animal group were statistically analyzed. The ANOVA test was employed to compare the studied animal groups: P<0.05 was considered significant.
Results
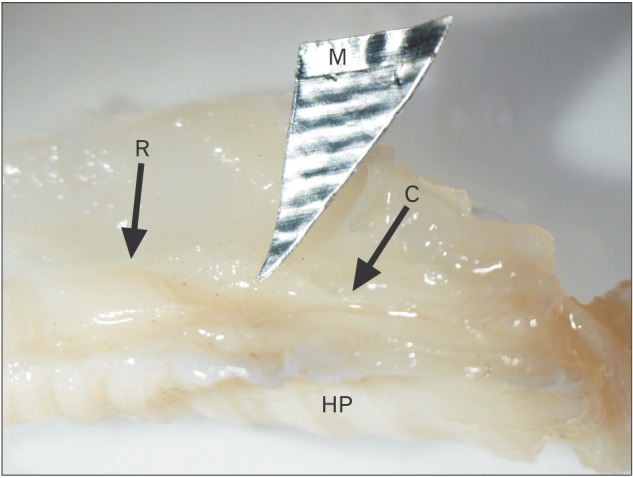
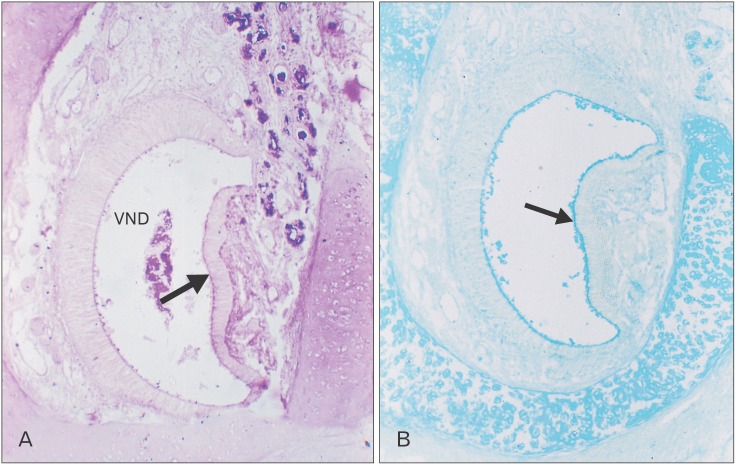
The VNO is a paired structure which appears in new born cat as an enlargement above the hard palate in a direct relationship with the vomer. It consists of three segments; rostral, middle and caudal (Fig. 1). The rostral segment (RS) is narrow and long. It runs posteriorly in a longitudinal direction above and parallel to the palate. The middle segment (MS) is wide and runs poster-ventrally in an oblique direction (Fig. 1). Histologically, the VNO consists of the VND surrounded by soft tissue structures including vomeronasal glands (VNGs), blood vessels, smooth muscles, nerves and connective tissue. The VND consists of three parts: rostral, middle and caudal.
Development of the RS of the VND
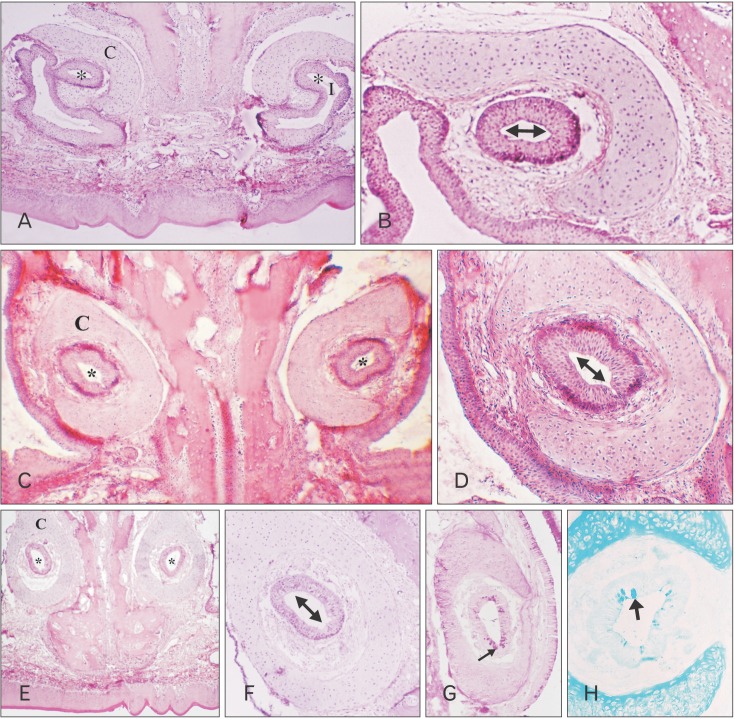
In new born kitten, the RS turns its axis for 90° and changes its lining epithelium as it passes posteriorly to form the MS. The most anterior part of the RS possesses an ovoid transversely directed lumen which opens into the naso-palatine duct (Fig. 2A). It is lined with nonkeratinized stratified squamous epithelium (Fig. 2B). The VND luminal axis change 45° becoming obliquely directed (Fig. 2C), and the lining epithelium becomes ciliated stratified columnar epithelium (Fig. 2D). Then the VND turns for another 45° so that its luminal axis becomes vertically directed (Fig. 2E), and the lining epithelium becomes ciliated pseudo-stratified partially ciliated columnar epithelium with goblet cells (Fig. 2F) which are PAS and AB positive in reaction (Fig. 2G, H).
By 2 weeks, the RS reveals growth of its various parts which are lined by the same types of epithelium as those found at birth with a mild increase in the VNGs present in this area compared with that in the previous age. The most caudal part which is lined by pseudo-stratified columnar ciliated epithelium with goblet cells reveals a mild decrease in PAS and AB reaction compared with that in new born.
By 4 weeks, the RS reveals an increase in thickness of its various parts which are lined by the same types of epithelium as those found in new born cat with an increase in the VNGs present in this area in comparison with the previous age. The most caudal part which is lined by pseudo-stratified columnar ciliated epithelium with goblet cells reveals a mild decrease in PAS and AB reaction compared with that in the previous age.
By 8 weeks, the RS reveals an increase in thickness of its various parts which are lined by the same types of epithelium as those found at birth (Fig. 3A). The VNGs present in this area became numerous (Fig. 3B). The most caudal part which is lined by pseudo-stratified columnar ciliated epithelium with goblet cells reveals a more decrease in PAS and AB reaction in the lining-cells compared to that in the previous age and a mild PAS positive and AB negative reaction in the VNGs (Fig. 3C, D).
Development of the MS-NSE of the VND
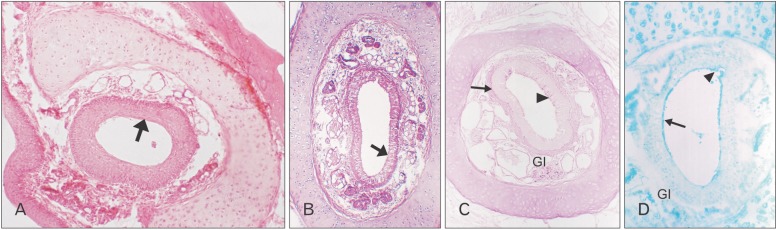
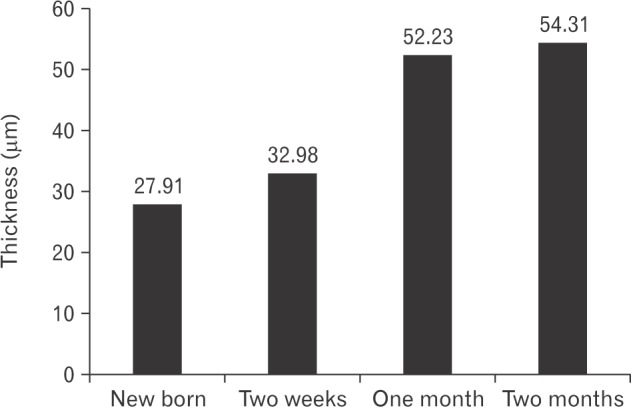
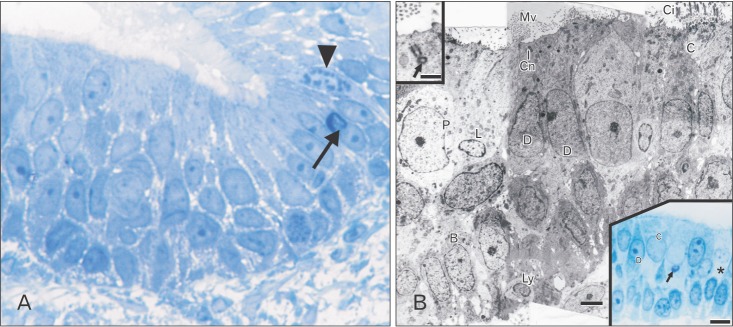
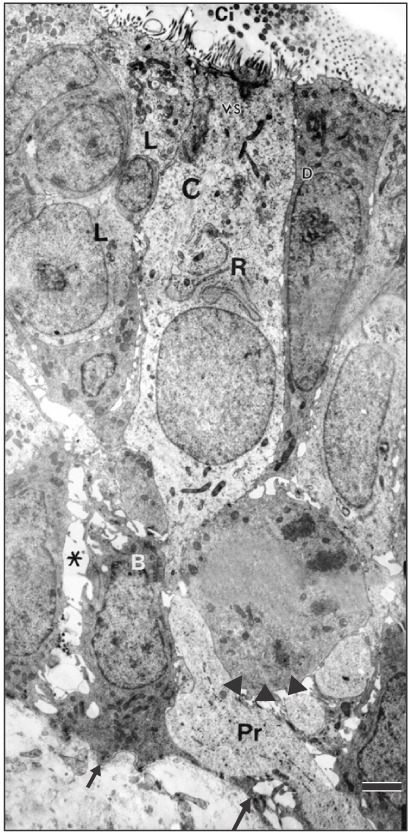
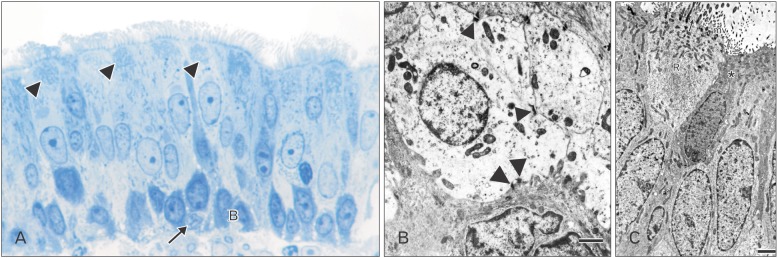
On the day of birth, the cross section of the MS of VND exhibits crescent-shaped, vertically oriented lumen which is maintained throughout its length. It is lined medially with sensory epithelium and laterally with NSE. The NSE thickness measures 27.91±7.18 µm at birth (Table 1, Fig. 4). It consists of pseudostratified partially ciliated columnar epithelium without goblet cells. In semi-thin sections, four types of cells could be distinguished; ciliated columnar, dark, light, and pale nonciliated columnar and basal cells. The ciliated cells are lightly stained and the most common type. The dark nonciliated columnar cells are less common, their cytoplasm may reveal metachromatic areas of variable size. The pale nonciliated columnar cells are the least common type. Both mitotic and apoptotic figures are observed among the lining epithelium (Fig. 5A). Mitotic figures are found both among the apical and basal cells (Fig. 5A). Intra epithelial invading cells as neutrophils and lymphocytes are not uncommon. The basal cell layer forms a continuous raw next to the basement membrane. They are ovoid in shape with their long axis lying perpendicular to the basement membrane (Fig. 5A, B). In ultrastructure, the ciliated columnar cells exhibit surface cilia and microvilli, mild electron-dense cytoplasm containing a few organelles and ovoid euchromatic nuclei containing well-developed nucleoli. The infranuclear cytoplasm forms processes which extend between basal cells and form desmosomal junctions with neighboring columnar and basal cells (Fig. 5B). The dark nonciliated columnar cells exhibit surface microvilli, moderate electron-dense cytoplasm containing mitochondria, rough endoplasmic reticulum (RER), supranuclear moderately developed Golgi bodies, lysosomal dense bodies, and occasional subsurface centrosomes. These cells are connected to other types of cells by junctional complex at the apical part of the lateral plasma membrane which form interdigitating processes. The pale nonciliated columnar cells possess a few surface microvilli and electron-lucent cytoplasm which characteristically contains some clear vesicles, a few free ribosomes, a few mitochondria, Golgi bodies, and euchromatic nuclei containing well developed nucleoli (Fig. 5B). The basal cells possess moderate-electron dense cytoplasm and nuclei and characteristically contain numerous bundles of tonofilaments, free ribosomes and mitochondria. Numerous desmosomal junctions connect them to the neighboring cells, whereas hemidesmosomes link them to the basal lamina (Fig. 5B).
Paraffin section reveals PAS and AB positive reaction of the epithelial surface (Fig. 6A, B).
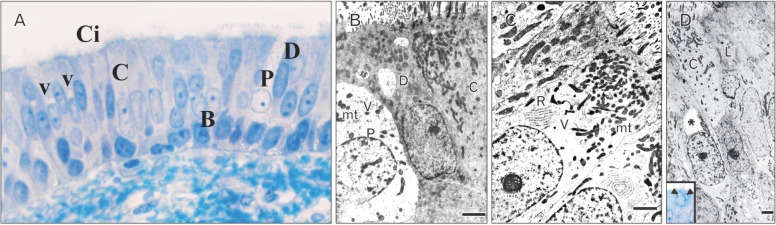
By the age of 2 weeks, the MS NSE of the VND reveals a marked increase in its thickness (32.98±8.47 µm). The ciliated cells reveal a mild increase in cytoplasmic organelles. The dark and light nonciliated cells exhibit an increase in supranuclear cytoplasm (Fig. 7).
By 4 weeks, the MS NSE of the VND exhibits an increase in thickness (52.23±18.68 µm). The lining cells reveal supranuclear cytoplasmic vacuoles. The ciliated columnar cells reveal subsurface darkened granular cytoplasm. Some cells contain metachromatic-stained areas in the cytoplasm (Fig. 8A). In ultrastructure, the ciliated cells reveals a marked increase in mitochondria and RER in the supranuclear areas of the cytoplasm which may reveal rounded vacuoles (Fig. 8B) and/or irregular shaped area containing electron lucent matrix (Fig. 8C). The dark and light nonciliated columnar cells contains fewer supranuclear mitochondria compared with those in ciliated cells (Fig. 8B, C). The pale nonciliated cells reveal a mild increase in mitochondria and small clear vesicles which are present around the nucleus (Fig. 8B). The basal cells reveal a mild increase in cell organelles particularly the short segments of RER. The desmosomal junctions which connect the basal cells to the basal processes of the columnar cells are frequently detected with reduction in the intercellular spaces. Invading inflammatory cells mainly lymphocytes and neutrophils are infrequently detected in the basal area (Fig. 8A).
By 8 weeks, the MS NSE of the VND reveals a mild, statistically insignificant, increase in thickness (54.31±18.68 µm); however, the lining cells exhibit prominent increase in the surface processes and the amount of cytoplasm relative to the nucleus and the subsurface granulated areas. The ciliated cells characteristically reveal prominently stained ovoid area in the supranuclear cytoplasm (Fig. 9A). Some cells reveal markedly darkened nucleus and cytoplasm. Ultrastructure of ciliated cells reveal supranuclear areas formed of free and attached RER and subsurface accumulation of mitochondria (Fig. 9B). The pale nonciliated cells reveal a mild increase in cytoplasmic organelles and numerous desmosomal junctions attach them to the adjacent columnar cells (Fig. 9C).
Topography of the NSE of the MS
At birth, the ciliated cells extend an unevenly distributed long processes which are of two forms: thick ciliary and thin microvillar processes whereas the nonciliated cells extend short processes whereas the nonciliated cells extend only thin microvillar processes. By the age of 2 weeks postnatal, the topography exhibits a mild increase in height of the cells processes (Fig. 10B), whereas at 4 weeks, there is a marked increase in height (Fig. 10C), which continue to the age of 8 weeks. The short processes show variation in density from an area to another (Fig. 10D).
Discussion
The rotation of the VND, which we have detected in the cat, occurs in its RS for 90° and results in a transverse orientation of its lumen, there the VND opens into the nasopalatine duct. Salazer et al. [13 14] in their studies did not point out to the rotation of the RS of the VND. We found that this form of rotation is related only to the orientation of the duct lumen which changes from being vertical caudally, into transverse rostrally. The nasopalatine duct which is a route for stimulus access, connects the VNO into the oral and nasal cavities. The rotation in cat differs from that reported in the rat in its location, as it occurs in the posterior part of the MS. In rat, the duct rotation results in a change in the orientation of the epithelium lining of the duct so that the NSE changes from the lateral position into the dorsal position [7]. Although both the rat and the cat have patent nasopalatine duct during early fetal life [20 21], rotation in the VND RS is not recorded in rat.
Salazar et al. [13] reported presence of only two types of epithelia. However, we reported here the presence of a partially ciliated stratified columnar epithelium among the lining of the VND as a transition between the stratified squamous epithelium rostrally and the pseudostratified epithelium caudally. In accordance with our findings, Steinberg [22] reported that the characteristics of the cat epithelium vary depending on position.
NSE lining the MS of the VND contains no goblet cells, whereas the posterior part of the RS contains goblet cells which produce neutral and acid mucopolysaccharides. Their presence could be a compensation for the paucity of VNGs in this area.
Besides, the frequently detected cytoplasmic supranuclear vacuoles which appear in TEM as nonmembrane bound vacuoles and metachromatic when stained with TB suggest the presence of some secretory activity of the columnar cells.
The pale nonciliated columnar cells are negative in reaction for CD96 though they coincide morphologically with those reported in the developing rabbit NSE [9]. This could be attributed to the paucity of invading inflammatory cells compared with those reported in rabbit. Pale cells similar in structure to those found in the cat have been reported in bandicoot olfactory epithelium [23]. These cells have been suggested to be concerned with ion exchange [23].
The results regarding the SEM correlate well with both the light microscope and TEM. The topographic long processes which are unevenly distributed along the surface of the VND correlate with the ultrastructural ciliary and microvillar processes extending from the ciliated cells. Their irregular presence does not substantiate the function of spreading out of mucous and/or mucous removal from the lumen of the VNO as suggested by Salazar et al. [13]. Adams and Wiekamp [24] suggested that cilia may serve a purpose in mixing the fluid content of the vomeronasal lumen. These ciliated cells characteristically possess apical vesicular smooth endoplasmic reticulum (SER) at birth and their gradual increase as development progressed coincides with our work in rabbit [9] where a possibility of an immune function is suggested. The paucity of invading intraepithelial neutrophils, which are suggested to function in immune surveillance lend support to the suggestion of the immune function. In addition, the ciliated cells reveal apical cytoplasmic granulations and mitochondrial aggregation. In contrary, the other types of nonciliated columnar cells always exhibit evenly distributed mitochondria. Several functions including detoxification have been linked to abundance of SER in other cellular systems [25]. Besides, Ferrari et al. [26] hypothesized that the great development of SER in the apical location of the supporting cells of armadillo olfactory epithelium is related to nasal xenobiotic metabolism. The topographic short processes correlate to the ultrastuctural microvillar processes which extend from the nonciliated columnar cells (dark, light, or pale). The stage during which the great topographic development occurs in the height of cellular processes of the VND (between 2- and 4-week postnatal) correlates well with the morphometric increase in epithelial thickness and the ultrastructural increase in cytoplasmic organelles. The topographic continuous increase in the height of these cells processes till the age of 2 months correlates with the ultrastructural maturational changes of the cytoplasmic cell organelles including the well-developed Golgi bodies, several stacks of RER and numerous mitochondria. To close up, the cat VND possesses several characteristics including presence of three types of epithelium lining the RS and the NSE of the MS characteristically has numerous ciliated cells and a few invading cells.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download