Abstract
The toxic effects of morphine sulfate in the adult cerebral cortex and one-day neonatal cerebellum have been studied. This study was carried out to evaluate the effect of maternal morphine exposure during gestational and lactation period on the Purkinje cells and cerebellar cortical layer in 18- and 32-day-old mice offspring. Thirty female mice were randomly allocated into cases and controls. In cases, animals received morphine sulfate (10 mg/kg/body weight intraperitoneally) during the 7 days before mating, gestational day (GD 0-21) 18 or 32. The controls received an equivalent volume of saline. The cerebellum of six infants for each group was removed and each was stained with cresyl violet. Quantitative computer-assisted morphometric study was done on cerebellar cortex. The linear Purkinje cell density in both experimental groups (postnatal day [P]18, 23.40±0.5; P32, 23.45±1.4) were significantly reduced in comparison with the control groups (P18, 28.70±0.9; P32, 28.95±0.4) (P<0.05). Purkinje cell area, perimeter and diameter at apex and depth of simple lobules in the experimental groups were significantly reduced compared to the controls (P<0.05). The thickness of the Purkinje layer of the cerebellar cortex was significantly reduced in morphine treated groups (P<0.05). This study reveals that morphine administration before pregnancy, during pregnancy and during the lactation period causes Purkinje cells loss and Purkinje cell size reduction in 18- and 32-day-old infant mice.
Morphine (C17H19O3N) is one of the strongest known anal gesic compounds [1]. The prevalence of opioid abuse is increasing globally, particularly in women of child-bearing age. Opioid administration during pregnancy can delay embryonic development, induce preterm labor, cause fetus death, produce chromosomal anomalies and neural tube defects, and result in low birth weight [2-7]. Infants of opioid-dependent mothers can display several behavioral abnormalities including hyperactivity, lower mental acuity, and motor development index [2, 3, 5].
An animal model has been used to document the neurotoxicity of high dosages of morphine [8]. Chronic administration of morphine can induce neuronal apoptosis in the central nervous system [9]. Morphine neurotoxicity was also shown in a human neuron cell culture model [10]. Morphine sulphate reduces the number of neurons and the thickness of cortex of cerebrum in mice neonates [11]. Morphine decreases the number and diameter of Purkinje cells, and the thickness of both the molecular and granular cerebellum layers of adult rats [12]. We previously reported that maternal morphine exposure during gestation reduces Purkinje cells in the one-day-old mice off spring [13].
These results highlight the concern regarding the high prevalence of opioid abuse in the world, particularly in women of child-bearing age, in light of the continued cerebellum development in the postnatal period. Very little is known of the effect of morphine sulphate on cerebellar development during infancy and after weaning. The present study was conducted to clarify the Purkinje cell and cortical layer alterations in the cerebellum of mice pups born to morphine-exposed mothers.
This experimental study was performed at the Gorgan Faculty of Medicine, Golestan University of Medical Sciences, Gorgan, Iran. Guidelines on the care and use of laboratory animals and approval of the ethic committee of Golestan University of Medical Sciences were observed.
Balb/c mice, weighing 28-30 g, and 8-9-weeks-of-age were maintained in a climate-controlled room under a 12-hour alternating light/dark cycle at a controlled temperature of 20-22℃. Dry food pellets and water were provided ad libitum.
Each vial contained l ml of morphine sulphate (Darou Pakhsh, Tehran, Iran) dissolved in 3.3 ml of sterile 0.9% saline. The morphine sulphate was intraperitoneally (i.p.) injected into each mouse at a concentration of 10 mg/kg body weight.
After 2 weeks of acclimation to the diet and the environment, females were randomly divided into control and treated groups. Twelve female mice in the treated groups received morphine sulphate as detailed above during 7 days before mating and during the gestational period (GD 0-21), at 18 days after delivery in experimental group I and 32 days after delivery in experimental group II. Twelve female mice in the control groups received an equivalent volume of normal saline i.p. during the 7 days before mating, and at GD 18 (group III) or 32 (group IV). After parturition, on postnatal day 18 (P18) and 32 (P32), six mice pups of each control and experimental group were randomly selected and scarified quickly with chloroform anesthesia. The brain was exposed and fixed by immersion into 10% neutral-buffered formalin. After processing, 6 µm-thick sections were acquired using a model HM 325 microtome (Microm, Walldorf, Germany). Serial coronal sections were selected according to anatomical landmarks corresponding to bregma (-6.255 mm to -6.955 mm) of the cerebellar simple lobule. The sections were stained with cresyl violet for morphometric examination.
In each sample, six similar sections of simple lobule of cerebellum were selected and images of six separate fields in the apex and depth position were captured using a model BX 51 microscope and DP12 digital camera, with associated OLYSIA autobioreport software (Olympus Optical, Tokyo, Japan). Morphometric analyses of cerebellar cortex including thickness of the molecular layer (ML), Purkinje cell layer (PCL), and granular cell layer (GCL) were measured by low magnification (40× objective) (Figs. 1-3). Morphometric analyses of the Purkinje cells, which included the linear density of Purkinje cells, soma area, perimeter and diameter of Purkinje cells in the apex and depth of folium, were measured at high magnification (100× objective) (Figs. 1-3). Purkinje cells that exhibited an appropriate grid was applied on the image and the linear density of the cells was determined from the number of cells per 1,000 µm length of Purkinje cell line. Also the relative area, perimeter and diameter of the cell body was calculated by the software only for those Purkinje cells with a fixed geometric point of nucleus and with prominent nucleolus (Figs. 2, 3). The results are expressed as the mean of thickness of layers, diameter (µm), soma area (µm2), and linear densities of the Purkinje cells in each group.
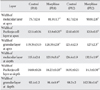
No significant increase in average body weight, brain weight, and brain-to-body weight ratio were found between the morphine sulphate and control groups at P18 and P32 (Table 1).
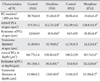
There was significant increase in the width of ML and granular layer at apex and depth of the simple lobule in the treated mice in comparison with controls at P18 and P32. The thickness of the Purkinje layer of cerebellar cortex was significantly reduced at P18 and P32 in the morphine-exposed mice in comparison with the controls (P<0.05) (Table 2, Fig. 2).
Concerning linear Purkinje cells density, the number of Purkinje cells per 1,000 µm length of Purkinje cell line of the experimental groups at P18 (23.40±0.5) and P32 (23.45±1.4) was reduced in comparison with control groups (28.70±0.9 at P18, 28.95±0.4 at P32) (both P<0.001) (Fig. 3). Concerning the area, perimeter, and diameter of the Purkinje cells, the cell area and perimeter in apex and depth of simple lobule in P18 and P32 cerebellar mice in the treated groups were significantly reduced in comparison with mice in the control groups (Fig. 3). The transverse diameter of Purkinje cells in the experimental groups was reduced in comparison with the control groups at P18 and P32 (P<0.001) (Table 3).
According to our findings, administration of morphine sulfate before and during pregnancy, and during lactation, causes Purkinje cell loss and reduces cell size in 18- and 32-day-old infant mice. The toxic effect of morphine continues on the Purkinje cells of cerebellum even after the lactation period.
We previously reported that the maternal morphine exposure during gestation reduces Purkinje cells in one-day-old mice offspring [13]. Furthermore, two studies reported that morphine sulfate administration can cause neurotoxic effects on the cerebellum and cerebral cortex [11, 12]. One of these studies showed that oral morphine exposure during the GD reduces both cortical thickness and the number of neurons in the developing fetal frontal cerebral cortex [11]. The second study reported that the long-term administration of morphine sulphate in adult rats significantly reduced the Purkinje cells [12]. Other substances, such as alcohol and nicotine, can cause cerebellar Purkinje cell loss [14, 15].
Regarding the effect of morphine on the central nervous system, several possible mechanisms can be considered. Loss of the Purkinje cells in morphine treated animals can be due to apoptosis and or necrosis [16]. On the other hand, morphological alterations of astrocytes due to morphine can increase both Ca2+ and production of carbonyl oxidation, which subsequently promote apoptosis and or necrosis in neurons [17]. Furthermore, morphine can block the proliferation of neuroblasts in ML by preventing of DNA synthesis [18]. Several studies have shown that acute opioids exposure blocks the proliferation, differentiation, and survival of neuroblasts and astroglia of the cerebellum [19-23]. Reduction of the Purkinje cells in morphine chronic users has been reported [24]. The involvement of opioids in cerebellar growth regulation has been revealed by experimentally perturbing the endogenous opioid system. Endogenous opioid peptides and receptors are widely expressed by developing cerebellar cells [25-29]. Although heroin and morphine preferentially activate µ opioid receptors, at high concentrations they can activate δ and κ receptors [30]. Continuous opioid receptor blockage accelerates cerebellar growth in postnatal rats, while over-stimulating opioid receptors, as occurs with opiate drugs, retards cerebellar growth [19, 23, 31]. Also, several studies have reported that morphine as heroin can reduce Purkinje cell proliferation [16, 32], cell differentiation, and increase Purkinje cells death in the cerebellum [21, 28]. Neurotoxic effects of opioids can be inducing by the N-methyl-D-aspartate receptor-caspase pathway [8]. Indeed, cell death can be due to mitochondrial damage [33-35].
Another possible mechanism for the alteration of the Purkinje cells may be the significant decrease of calbindin protein as a neuroprotective agent in neurons [36, 37]. Furthermore, opioids may block neuronal activity, causing the neurons to receive internal signals to commit suicide (apoptosis) [38]. Other possible mechanisms for Purkinje cells loss may be due to derangement of the Purkinje cells in the rat cerebellum following prenatal exposure to morphine. Reelin has been shown to play an important role in the migration of neural cells, prompting the suggestion that the decrease in Reelin by X-ray irradiation is an important factor for the derangement of Purkinje cells [39].
Also, morphine presently induced a significant reduction of the thickness of the Purkinje layer. Our finding is similar to other studies [12, 40]. The cause of the reduction of the thickness of Purkinje layer may reflect decreased Purkinje cell size. Morphine presently induced a significant increase in the thickness of the molecular and granular layers in all treated mice. Our finding contrasts with other studies [12, 41]. Bekheet et al. [12] reported that morphine significantly decreased the thickness of molecular and granular layers in all treated animals. Also, another study reported that morphine induced a significant decrease in the molecular and granular layers thickness of the rat cerebellum [41]. The differences between the present and previopus findings regarding the thickness of molecular and granular layers of the cerebellum may be due to the timing of the studies. Our study was done in infancy but the other studies were done on adult animals. According to Simat et al. [42], proliferation and migration of basket and stellate cells in the ML is initiated after 7 postnatal days. Further studies are required to survey the type of cells and layers of cerebellum in different phase of postnatal periods.
In conclusion, this study reveals that morphine administration before and during gestational and lactation period causes Purkinje cell loss and reduction of the size and thickness of PCL of the cerebellar cortex in 18- and 32-day-old infant mice. Comparing the present findings with our previous study, we can conclude that the neurotoxic effect of morphine will be continued, even with discontinuation of exposure.
Figures and Tables
Fig. 1
A representative coronal section stained with cresyl violet of cerebellum at control Balb/c mice (P18) group. The left picture shows a simple lobule of cerebellum in 40× magnification. A1, apex of lobule in 100× magnification (grid: 200 µm×200 µm); B1, depth of lobule in 100× magnification (grid: 200 µm×200 µm). P, postnatal day. Scale bar=200 µm.
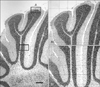
Fig. 2
Coronal section stained with cresyl violet of cerebellum cortex Balb/c mice (P18) in the control group. (A) A2, apex of lobule in 200× magnification (grid, 100 µm×100 µm). (B) A3, apex of lobule in 1,000× magnification (grid, 20 µm×20 µm). ML, molecular layer; PC, Purkinje cell; PCL, Purkinje cell layer (that Purkinje cells are located in one line); GL, granule cell layer; WM, white matter.

Fig. 3
Coronal section stained with cresyl violet of cerebellum cortex Balb/c mice (P18) in control group. (A) B2, depth of lobule in 200× magnification (grid, 100 µm×100 µm). (B) B3, depth of lobule in 1,000× magnification (grid, 20 µm×20 µm). ML, molecular layer; PC, Purkinje cell; PCL, Purkinje cell layer (that Purkinje cells are located in one line); GL, granule cell layer; WM, white matter. Arrows showing the dendritic branches of PCs, white arrow line showing the width of PCL and white line is showing the diameter of PCs.

Table 1
Body weight, brain weight, brain to body weight ratio in morphine treated and control groups of postnatal Balb/c mice (P18, P32)

Acknowledgements
We thank the Deputy Research of Golestan University of Medical Sciences for financial support (Grant number: 7236) and Dr. Abbas Ali Keshtkar for analysis of Data.
References
1. Zhang Y, Chen Q, Yu LC. Morphine: a protective or destructive role in neurons? Neuroscientist. 2008. 14:561–570.
2. National Institute on Drug Abuse. National pregnancy and health survey: drug use among women delivering livebirths, 1992 (NIH Publication 96-3819). 1996. Rockville, MD: U. S. Department of Health and Human Services, National Institutes of Health, National Institute of Drug Abuse, Division of Epidemiology and Prevention Research;1–157.
3. Ornoy A, Michailevskaya V, Lukashov I, Bar-Hamburger R, Harel S. The developmental outcome of children born to heroindependent mothers, raised at home or adopted. Child Abuse Negl. 1996. 20:385–396.
4. Raye JR, Dubin JW, Blechner JN. Fetal growth retardation following maternal morphine administration: nutritional or drug effect? Biol Neonate. 1977. 32:222–228.
5. Wilson GS, McCreary R, Kean J, Baxter JC. The development of preschool children of heroin-addicted mothers: a controlled study. Pediatrics. 1979. 63:135–141.
6. Fazel A, Jalali M. Experimentally-induced exencephaly and spina bifida in mice. Arch Iran Med. 2002. 5:179–183.
7. Nasiraei-Moghadam S, Sahraei H, Bahadoran H, Sadooghi M, Salimi SH, Kaka GR, Imani H, Mahdavi-Nasab H, Dashtnavard H. Effects of maternal oral morphine consumption on neural tube development in Wistar rats. Brain Res Dev Brain Res. 2005. 159:12–17.
8. Mao J, Sung B, Ji RR, Lim G. Neuronal apoptosis associated with morphine tolerance: evidence for an opioid-induced neurotoxic mechanism. J Neurosci. 2002. 22:7650–7661.
9. Atici S, Cinel L, Cinel I, Doruk N, Aktekin M, Akca A, Camdeviren H, Oral U. Opioid neurotoxicity: comparison of morphine and tramadol in an experimental rat model. Int J Neurosci. 2004. 114:1001–1011.
10. Turchan-Cholewo J, Liu Y, Gartner S, Reid R, Jie C, Peng X, Chen KC, Chauhan A, Haughey N, Cutler R, Mattson MP, Pardo C, Conant K, Sacktor N, McArthur JC, Hauser KF, Gairola C, Nath A. Increased vulnerability of ApoE4 neurons to HIV proteins and opiates: protection by diosgenin and L-deprenyl. Neurobiol Dis. 2006. 23:109–119.
11. Sadraie SH, Kaka GR, Sahraei H, Dashtnavard H, Bahadoran H, Mofid M, Nasab HM, Jafari F. Effects of maternal oral administration of morphine sulfate on developing rat fetal cerebrum: a morphometrical evaluation. Brain Res. 2008. 1245:36–40.
12. Bekheet SH, Saker SA, Abdel-Kader AM, Younis AE. Histopathological and biochemical changes of morphine sulphate administration on the cerebellum of albino rats. Tissue Cell. 2010. 42:165–175.
13. Ghafari S, Roshandel D, Golalipour MJ. Effect of intrauterine morphine sulfate exposure on cerebellar histomorphological changes in neonatal mice. Folia Neuropathol. 2011. 49:328–334.
14. Lee Y, Rowe J, Eskue K, West JR, Maier SE. Alcohol exposure on postnatal day 5 induces Purkinje cell loss and evidence of Purkinje cell degradation in lobule I of rat cerebellum. Alcohol. 2008. 42:295–302.
15. Abou-Donia MB, Khan WA, Dechkovskaia AM, Goldstein LB, Bullman SL, Abdel-Rahman A. In utero exposure to nicotine and chlorpyrifos alone, and in combination produces persistent sensorimotor deficits and Purkinje neuron loss in the cerebellum of adult off spring rats. Arch Toxicol. 2006. 80:620–631.
16. Hauser KF, Gurwell JA, Turbek CS. Morphine inhibits Purkinje cell survival and dendritic differentiation in organotypic cultures of the mouse cerebellum. Exp Neurol. 1994. 130:95–105.
17. Hauser KF, Harris-White ME, Jackson JA, Opanashuk LA, Carney JM. Opioids disrupt Ca2+ homeostasis and induce carbonyl oxyradical production in mouse astrocytes in vitro: transient increases and adaptation to sustained exposure. Exp Neurol. 1998. 151:70–76.
18. Hauser KF, Houdi AA, Turbek CS, Elde RP, Maxson W 3rd. Opioids intrinsically inhibit the genesis of mouse cerebellar granule neuron precursors in vitro: differential impact of mu and delta receptor activation on proliferation and neurite elongation. Eur J Neurosci. 2000. 12:1281–1293.
19. Hauser KF, McLaughlin PJ, Zagon IS. Endogenous opioids regulate dendritic growth and spine formation in developing rat brain. Brain Res. 1987. 416:157–161.
20. Lorber BA, Freitag SK, Bartolome JV. Effects of beta-endorphin on DNA synthesis in brain regions of preweanling rats. Brain Res. 1990. 531:329–332.
21. Schmahl W, Funk R, Miaskowski U, Plendl J. Long-lasting effects of naltrexone, an opioid receptor antagonist, on cell proliferation in developing rat forebrain. Brain Res. 1989. 486:297–300.
22. Hammer RP Jr, Ricalde AA, Seatriz JV. Effects of opiates on brain development. Neurotoxicology. 1989. 10:475–483.
23. Zagon IS, McLaughlin PJ. Endogenous opioid systems regulate cell proliferation in the developing rat brain. Brain Res. 1987. 412:68–72.
24. Oehmichen M, Meissner C, Reiter A, Birkholz M. Neuropathology in non-human immunodeficiency virus-infected drug addicts: hypoxic brain damage after chronic intravenous drug abuse. Acta Neuropathol. 1996. 91:642–646.
25. Zagon IS, McLaughlin PJ. Identification of opioid peptides regulating proliferation of neurons and glia in the developing nervous system. Brain Res. 1991. 542:318–323.
26. Hauser KF, Osborne JG, Stiene-Martin A, Melner MH. Cellular localization of proenkephalin mRNA and enkephalin peptide products in cultured astrocytes. Brain Res. 1990. 522:347–353.
27. Spruce BA, Curtis R, Wilkin GP, Glover DM. A neuropeptide precursor in cerebellum: proenkephalin exists in subpopulations of both neurons and astrocytes. EMBO J. 1990. 9:1787–1795.
28. Shinoda H, Marini AM, Cosi C, Schwartz JP. Brain region and gene specificity of neuropeptide gene expression in cultured astrocytes. Science. 1989. 245:415–417.
29. Zhu Y, Hsu MS, Pintar JE. Developmental expression of the mu, kappa, and delta opioid receptor mRNAs in mouse. J Neurosci. 1998. 18:2538–2549.
30. Goldstein A. Binding selectivity profiles for ligands of multiple receptor types: focus on opioid receptors. Trends Pharmacol Sci. 1987. 8:456–459.
31. Slotkin TA. Perinatal exposure to methadone: how do early biochemical alterations cause neurofunctional disturbances? Prog Brain Res. 1988. 73:265–279.
32. Thomas JD, Goodlett CR, West JR. Alcohol-induced Purkinje cell loss depends on developmental timing of alcohol exposure and correlates with motor performance. Brain Res Dev Brain Res. 1998. 105:159–166.
33. Cheng WH, Quimby FW, Lei XG. Impacts of glutathione peroxidase-1 knockout on the protection by injected selenium against the pro-oxidant-induced liver aponecrosis and signaling in selenium-deficient mice. Free Radic Biol Med. 2003. 34:918–927.
34. Pretorius E, Bornman MS. Calcium-mediated aponecrosis plays a central role in the pathogenesis of estrogenic chemical-induced neurotoxicity. Med Hypotheses. 2005. 65:893–904.
35. van Tonder JJ. Effect of the cardiac glycoside, digoxin, on neuronal viability, serotonin production and brain development in the embryo. 2007. Pretoria: School of Medicine, Faculty of Health Science, University of Pretoria;MSc Thesis.
36. Garcia MM, Gilster J, Harlan RE. Chronic morphine decreases calbindin D28k immunoreactivity in a subset of cerebellar Purkinje neurons of rat brain. Brain Res. 1996. 734:123–134.
37. Kim JS, Kim JM, Son JA, Han SY, Kim CT, Lee NS, Jeong YG. Decreased calbindin-immunoreactive Renshaw cells (RCs) in the lumbar spinal cord of the ataxic pogo mice. Korean J Anat. 2008. 41:255–263.
38. Farber NB, Olney JW. Drugs of abuse that cause developing neurons to commit suicide. Brain Res Dev Brain Res. 2003. 147:37–45.
39. Darmanto W, Inouye M, Takagishi Y, Ogawa M, Mikoshiba K, Murata Y. Derangement of Purkinje cells in the rat cerebellum following prenatal exposure to X-irradiation: decreased Reelin level is a possible cause. J Neuropathol Exp Neurol. 2000. 59:251–262.
40. Fam A. Depressant effects of morphine on cellular composition of lymphoid tissues and cerebellum in rats. J Egypt Ger Soc Zool. 2002. 39:411–438.
41. Demeri O, Celik I, Seker M, Sur E, Ozkan Y, Aydin MF, Salbacak A. Histological and morphometric studies of the cerebellar cortex and nucleolus organiser region (Ag-NORs) in Purkinje neurons of chronic morphine treated rats. Proceedings of the XVII International Symposium on Morphological Science. 2002. Sep 11-15; Timisoara, Romania.
42. Simat M, Parpan F, Fritschy JM. Heterogeneity of glycinergic and gabaergic interneurons in the granule cell layer of mouse cerebellum. J Comp Neurol. 2007. 500:71–83.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download