Abstract
Purpose:
The objective of this study was to evaluate whether the effect on afferent c-fiber activity is the underlying mechanism of intravesical electrical stimulation (IVES) in spinal cord injured rats.
Materials and Methods:
Thirty five female Sprague-Dawley rats weighting 200-300g each were divided into the normal and spinalized groups. For the spinalized rats, we observed the c-fos expression, and we compared this in the non-IVES group with that in the IVES group. Cystometrograms were performed for all the groups via a suprapubic catheter.
Results:
After performing IVES in the normal and spinalized rats, the abnormal increases of the intercontraction interval (ICI) and the voiding pressure (VP) were reduced close to the normal range. In the spinalized rats, the number of c-fos positive cells in the dorsal commissure (DCM) decreased in the group that had IVES performed when compared with the non-IVES group.
Go to : 
REFERENCES
1.Kaplan WE., Richards I. Intravesical bladder stimulation in myelodysplasia. J Urol. 1988. 140:1282–4.

2.Cheng EY., Richards I., Balcom A., Steinhardt G., Diamond M., Rich M, et al. Bladder stimulation therapy improves bladder compliance: results from a multi-institutional trial. J Urol. 1996. 156:761–4.

3.Boone TB., Roehrbom CG., Hurt G. Transurediral intravesical electrotherapy for neurogenic bladder dysfunction in children widi myelodysplasia: a prospective, randomized clinical trial. J Urol. 1992. 148:550–4.
4.Ebner A., Jiang C., Lindstorm S. Intravesical electrical stimulation-an experimental analysis of the mechanism of action. J Urol. 1992. 148:920–4.

5.Cheng EY., Richards I., Kaplan WE. Use of bladder stimulation in high risk patients. J Urol. 1996. 156:749–52.

6.Lyne CJ., Bellinger MF. Early experience with transurethral electrical bladder stimulation. J Urol. 1993. 150:697–9.

7.Han SW., Kim MJ., Kim JH., Hong CH., Kim JW., Noh JY. Intravesical electrical stimulation improves neurogenic bowel dysfunction in children with spina bifida. J Urol. 2004. 171:2648–50.

8.Jiang CH., Lindstrom S. Intravesical electrical stimulation induces a prolonged decrease in micturition threshold volume in the rat. J Urol. 1996. 155:1477–81.

9.Matsumoto G., Hisamitsu T., de Groat WC. Role of glutamate and NMDA receptors in the descending limb of the spino-bulbospinal micturition reflex pathway of the rat. Neurosci Lett. 1995. 183:58–61.

10.Jiang CH. Modulation of the micturition reflex pathway by intravesical electrical stimulation: an experimental study in die rat. Neururol Urodyn. 1995. 17:543–53.
11.Vizzard MA. Increased expression of spinal cord Fos protein induced by bladder stimulation after spinal cord injury. Am J Physiol Regul Integr Comp Physiol. 2000. 279:R295–305.

12.Katona F. Stages of vegetative afferentation in reorganization of bladder control during intravesical electrotherapy. Urol Int. 1975. 30:192–203.

13.Cheng CL., Ma CP., de Groat WC. E伴ect of capsaicin on micturition and associated reflexes in chronic spinal rats. Brain Res. 1995. 678:40–8.
14.Mallory B., Steers WD., De Groat WC. Electrophysiological study of micturition reflexes in rats. Am J Physiol. 1989. 257:R410–21.

15.Wang Y., Hassouna MM. Neuromodulation reduces c-fos gene expression in spinalized rats: a double-blind randomized study. J Urol. 2000. 163:1966–70.

16.Birder LA., de Groat WC. Induction of c-fos expression in spinal neurons by nociceptive and nonnociceptive stimulation of LUT. Am J Physiol. 1993. 265:R326–33.

17.Birder LA., de Groat WC. Increased c-fos expression in spinal neurons after irritation of the lower urinary tract in the rat. J Neurosci. 1992. 12:4878–89.

18.Birder LA., Roppolo JR., Iadarola MJ., de Groat WC. C-fos as a marker for subsets of vesceral second order neurons in the rat lumbosacral spinal cord. Soc Neurosci. 1990. 16:(abstr): 703A.
Go to : 
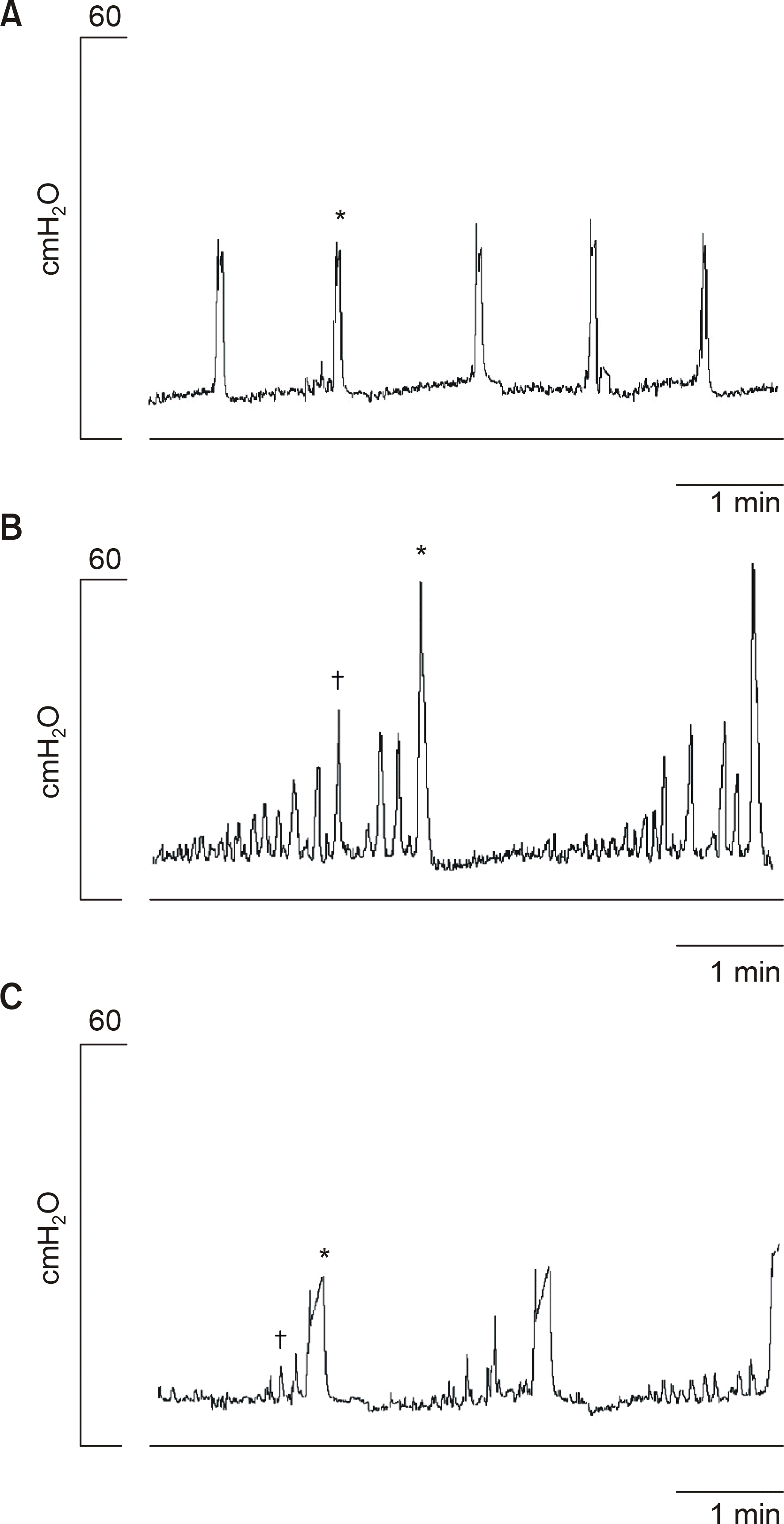 | Fig. 1.Effects of intravesical electrical stimulation. Cystometro- gram after the intravesical instillation of saline in the normal group (A) and the spinalized group (B). Intravesical electrical stimulation improved the urodynamic parameters in the spinalized group (C). Note the decrease in the number and the pressure of the nonvoiding contractions. ∗: voiding contraction (VC), : nonvoiding contraction (NVC). |
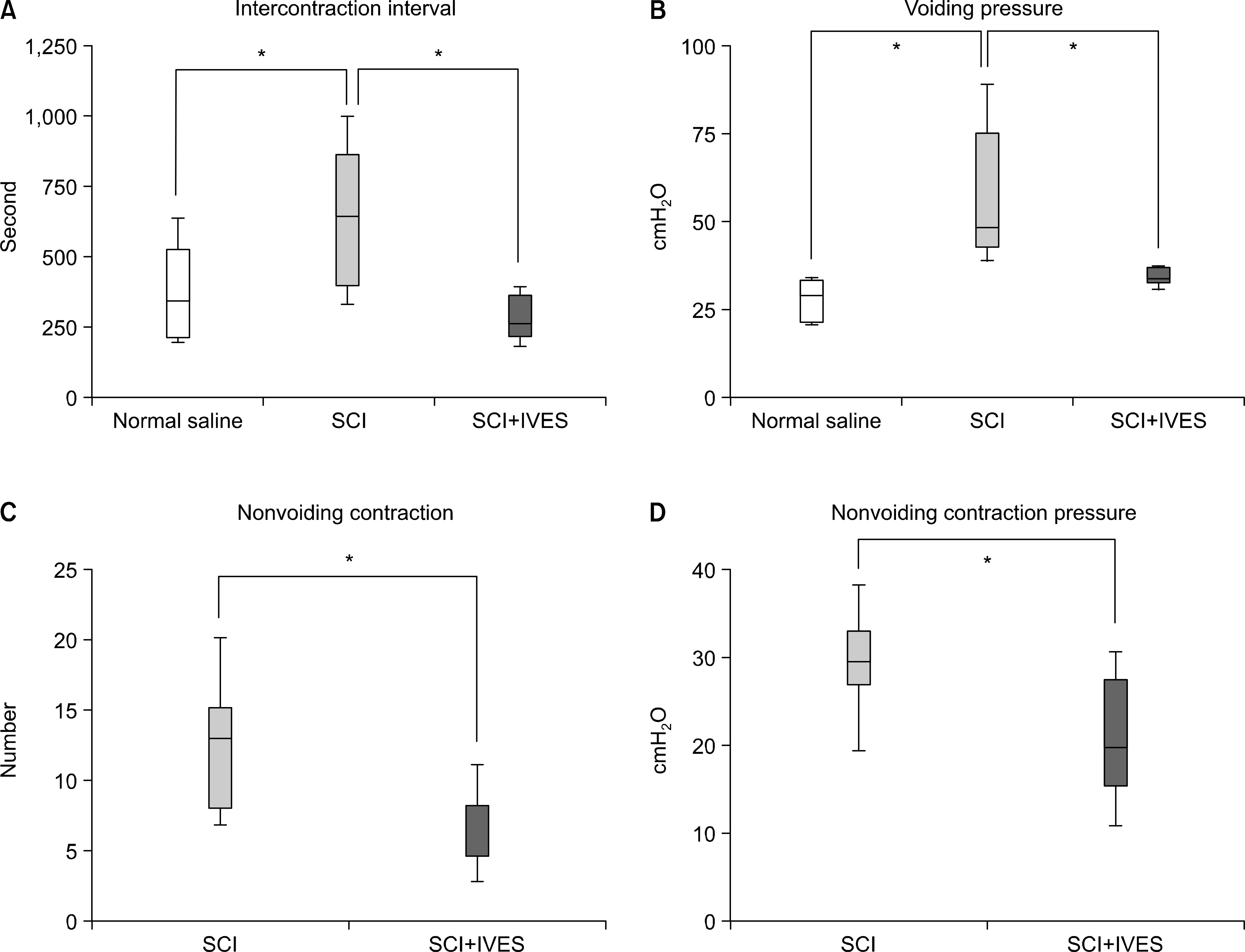 | Fig. 2.Intravesical electrical stimulation decreased the intercontraction interval and voiding pressure in the spinalized group (A, B). The number and pressure of the nonvoiding contractions decreased after intravesical electrical stimulation (C, D). SCI: spinal cord injury, IVES: intravesical electrical stimulation. ∗: statistical analysis by Student's t-test (unpaired) (p<0.05). |
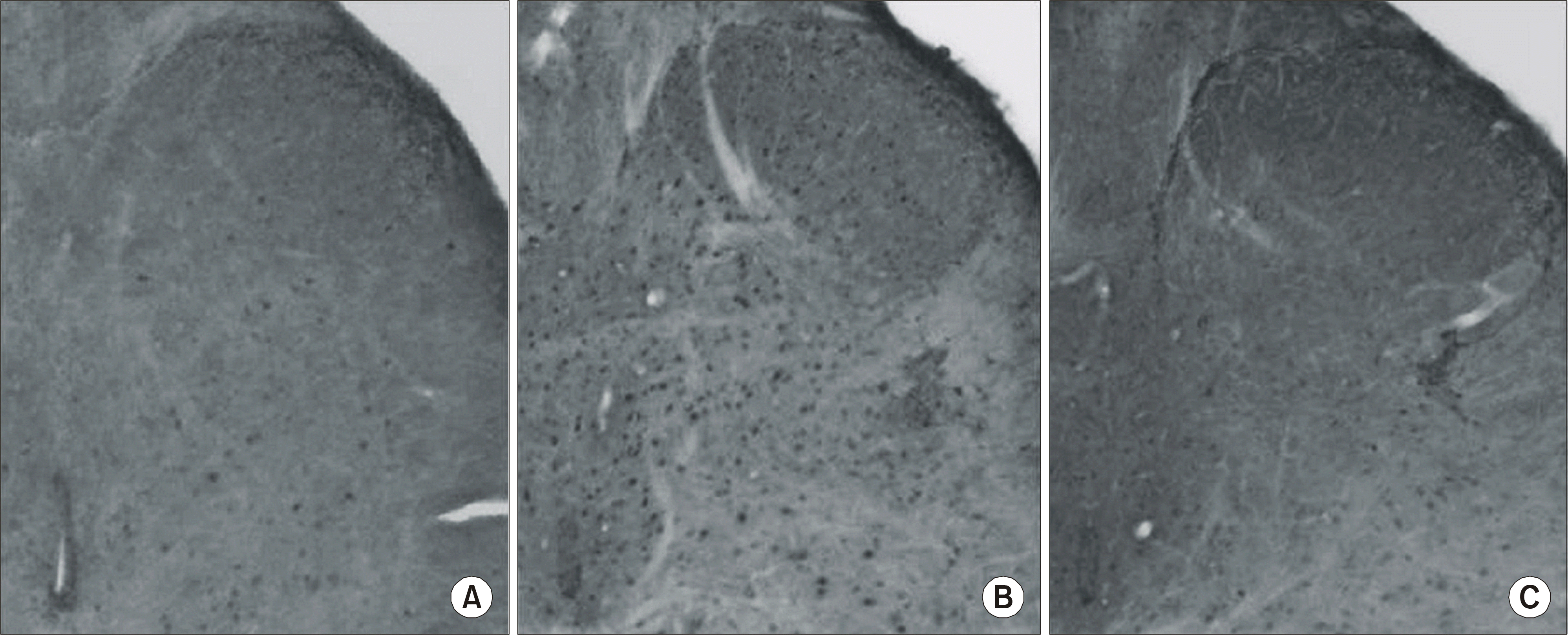 | Fig. 3.Immunohistochemical staining. The normal group (A), the saline group (SCI+NS) (B), and the IVES groups (SCI+IVES) (C). SCI: spinal cord injury, NS: normal saline, IVES: intravesical electrical stimulation. |
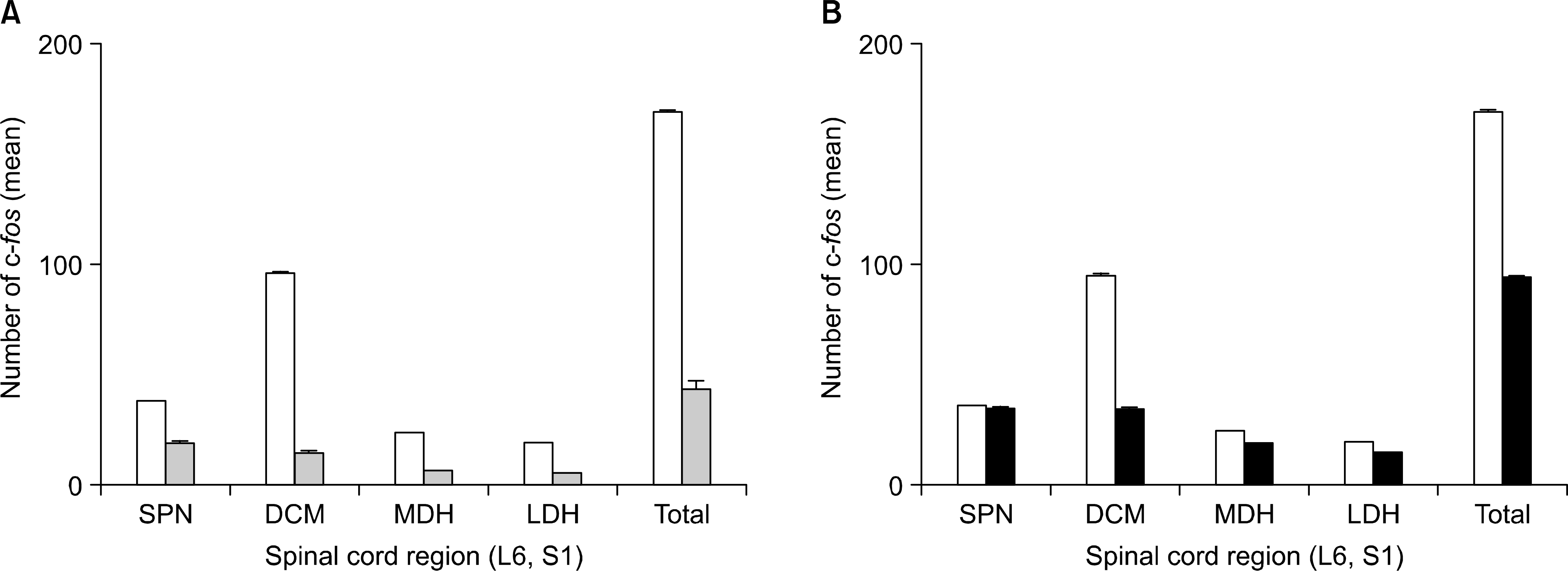 | Fig. 4.Topographical distribution according to the c-fos expression in the L6-S1 spinal cord of the spinalized rat. Saline group (□) and capsaicin group (□) (A), saline group (□) and IVES group (■) (B). SPN: sacral parasympathetic nucleus, DCM: dorsal commissure, MDH: medial dorsal horn, LDH: lateral dorsal horn. |
Table 1.
The number of Fos immunoreactive (IR) cells induced in each group (means 土 SE)
∗ : p-value indicates the comparison beween saline group (SCI+NS) and capsaicin group (SCI+Caps) (p<0.05)
† : p-value indicates the comparison between saline group (SCI+NS) and IVES group (SCI+IVES)(p<0.05), SCI: spinal cord injury, NS: normal saline, Caps: capsai in, IVES: intravesical electrical stimulation, MDH: medial dorsal horn, LDH: lateral dorsal horn, DCM: dorsal commissure, SPN: sacral parasympathetic nucleus




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download