1. Engel GL. The need for a new medical model: a challenge for biomedicine. Science. 1977; 196:129–36.

2. Weed LL. Medical records that guide and teach. N Engl J Med. 1968; 278:652–7.

3. Pincus T, Callahan LF. Reassessment of twelve traditional paradigms concerning the diagnosis, prevalence, morbidity and mortality of rheumatoid arthritis. Scand J Rheumatol Suppl. 1989; 79:67–96.

4. Callahan LF, Pincus T. Education, self-care, and outcomes of rheumatic diseases: further challenges to the “biomedical model” paradigm. Arthritis Care Res. 1997; 10:283–8.

5. McCollum L, Pincus T. A biopsychosocial model to complement a biomedical model: patient questionnaire data and socioeconomic status usually are more significant than laboratory tests and imaging studies in prognosis of rheumatoid arthritis. Rheum Dis Clin North Am. 2009; 35:699–712.

6. Pincus T, Castrejón I. MDHAQ/RAPID3 scores: quantitative patient history data in a standardized “scientific” format for optimal assessment of patient status and quality of care in rheumatic diseases. Bull NYU Hosp Jt Dis. 2011; 69:201–14.
7. Pincus T, Castrejón I. An evidence-based medical visit for patients with rheumatoid arthritis based on standard, quantitative scientific data from a patient MDHAQ and physician report. Bull NYU Hosp Jt Dis. 2012; 70:73–94.
8. Pincus T, Yazici Y, Castrejón I. Pragmatic and scientific advantages of MDHAQ/ RAPID3 completion by all patients at all visits in routine clinical care. Bull NYU Hosp Jt Dis. 2012; 70(Suppl 1):30–6.
9. Pincus T, Castrejón I. Are patient self-report questionnaires as “scientific” as biomarkers in “treat-to-target” and prognosis in rheumatoid arthritis? Curr Pharm Des. 2015; 21:241–56.

10. Pincus T, Sokka T, Kautiainen H. Patients seen for standard rheumatoid arthritis care have significantly better articular, radiographic, laboratory, and functional status in 2000 than in 1985. Arthritis Rheum. 2005; 52:1009–19.

11. Lichtenstein MJ, Pincus T. How useful are combinations of blood tests in “rheumatic panels” in diagnosis of rheumatic diseases? J Gen Intern Med. 1988; 3:435–42.

12. Pincus T. A pragmatic approach to cost-effective use of laboratory tests and imaging procedures in patients with musculoskeletal symptoms. Prim Care. 1993; 20:795–814.

13. Pincus T, Sokka T. Laboratory tests to assess patients with rheumatoid arthritis: advantages and limitations. Rheum Dis Clin North Am. 2009; 35:731–4.

14. Pincus T, Sokka T. Complexities in the quantitative assessment of patients with rheumatic diseases in clinical trials and clinical care. Clin Exp Rheumatol. 2005; 23(5 Suppl 39):S1–9.
15. Pincus T. Advantages and limitations of quantitative measures to assess rheumatoid arthritis: joint counts, radiographs, laboratory tests, and patient questionnaires. Bull NYU Hosp Jt Dis. 2006; 64:32–9.
16. Yazici Y, Sokka T, Pincus T. Radiographic measures to assess patients with rheumatoid arthritis: advantages and limitations. Rheum Dis Clin North Am. 2009; 35:723–9.

17. Fuchs HA, Brooks RH, Callahan LF, Pincus T. A simplified twenty-eight-joint quantitative articular index in rheumatoid arthritis. Arthritis Rheum. 1989; 32:531–7.

18. Sokka T, Pincus T. Quantitative joint assessment in rheumatoid arthritis. Clin Exp Rheumatol. 2005; 23(5 Suppl 39):S58–62.
19. Feinstein AR. An additional basic science for clinical medicine: II. The limitations of randomized trials. Ann Intern Med. 1983; 99:544–50.

20. Freireich EJ. The randomized clinical trial as an obstacle to clinical research. Varco RL, Delaney JP, editors. Controversy in surgery. Philadelphia: WB Saunders;1983. p. 5–12.
21. Ioannidis JP, Trikalinos TA. An exploratory test for an excess of significant findings. Clin Trials. 2007; 4:245–53.

22. Kaptchuk TJ. The double-blind, randomized, placebo-controlled trial: gold standard or golden calf? J Clin Epidemiol. 2001; 54:541–9.
23. Pincus T, Sokka T. Clinical trials in rheumatic diseases: designs and limitations. Rheum Dis Clin North Am. 2004; 30:701–24.

24. Pincus T, Stein CM. What is the best source of useful data on the treatment of rheumatoid arthritis: clinical trials, clinical observations, or clinical protocols? J Rheumatol. 1995; 22:1611–7.
25. Nishimura K, Sugiyama D, Kogata Y, Tsuji G, Nakazawa T, Kawano S, et al. Meta-analysis: diagnostic accuracy of anti-cyclic citrullinated peptide antibody and rheumatoid factor for rheumatoid arthritis. Ann Intern Med. 2007; 146:797–808.

26. Castrejón I, McCollum L, Tanriover MD, Pincus T. Importance of patient history and physical examination in rheumatoid arthritis compared to other chronic diseases: results of a physician survey. Arthritis Care Res (Hoboken). 2012; 64:1250–5.
27. Abelson B, Sokka T, Pincus T. Declines in erythrocyte sedimentation rates in patients with rheumatoid arthritis over the second half of the 20th century. J Rheumatol. 2009; 36:1596–9.

28. Wolfe F, Michaud K. The clinical and research significance of the erythrocyte sedimentation rate. J Rheumatol. 1994; 21:1227–37.
29. Smedstad LM, Moum T, Guillemin F, Kvien TK, Finch MB, Suurmeijer TP, et al. Correlates of functional disability in early rheumatoid arthritis: a cross-sectional study of 706 patients in four European countries. Br J Rheumatol. 1996; 35:746–51.

30. Sokka T, Pincus T. Erythrocyte sedimentation rate, C-reactive protein, or rheumatoid factor are normal at presentation in 35%-45% of patients with rheumatoid arthritis seen between 1980 and 2004: analyses from Finland and the United States. J Rheumatol. 2009; 36:1387–90.

31. Pincus T, Gibson KA, Shmerling RH. An evidence-based approach to laboratory tests in usual care of patients with rheumatoid arthritis. Clin Exp Rheumatol. 2014; 32(5 Suppl 85):S–23-8.
32. Olsen NJ, Callahan LF, Brooks RH, Nance EP, Kaye JJ, Stastny P, et al. Associations of HLA-DR4 with rheumatoid factor and radiographic severity in rheumatoid arthritis. Am J Med. 1988; 84:257–64.

33. Yelin E, Meenan R, Nevitt M, Epstein W. Work disability in rheumatoid arthritis: effects of disease, social, and work factors. Ann Intern Med. 1980; 93:551–6.

34. Pincus T, Callahan LF, Sale WG, Brooks AL, Payne LE, Vaughn WK. Severe functional declines, work disability, and increased mortality in seventy-five rheumatoid arthritis patients studied over nine years. Arthritis Rheum. 1984; 27:864–72.

35. Callahan LF, Bloch DA, Pincus T. Identification of work disability in rheumatoid arthritis: physical, radiographic and laboratory variables do not add explanatory power to demographic and functional variables. J Clin Epidemiol. 1992; 45:127–38.
36. Wolfe F, Hawley DJ. The longterm outcomes of rheumatoid arthritis: Work disability: a prospective 18 year study of 823 patients. J Rheumatol. 1998; 25:2108–17.
37. Puolakka K, Kautiainen H, Möttönen T, Hannonen P, Hakala M, Korpela M, et al. Predictors of productivity loss in early rheumatoid arthritis: a 5 year follow up study. Ann Rheum Dis. 2005; 64:130–3.

38. Wolfe F, Mitchell DM, Sibley JT, Fries JF, Bloch DA, Williams CA, et al. The mortality of rheumatoid arthritis. Arthritis Rheum. 1994; 37:481–94.

39. Aletaha D, Landewe R, Karonitsch T, Bathon J, Boers M, Bombardier C, et al. Reporting disease activity in clinical trials of patients with rheumatoid arthritis: EULAR/ACR collaborative recommendations. Arthritis Rheum. 2008; 59:1371–7.

40. Lubeck DP, Spitz PW, Fries JF, Wolfe F, Mitchell DM, Roth SH. A multicenter study of annual health service utilization and costs in rheumatoid arthritis. Arthritis Rheum. 1986; 29:488–93.

41. Michaud K, Messer J, Choi HK, Wolfe F. Direct medical costs and their predictors in patients with rheumatoid arthritis: a three-year study of 7,527 patients. Arthritis Rheum. 2003; 48:2750–62.
42. Wolfe F, Zwillich SH. The long-term outcomes of rheumatoid arthritis: a 23-year prospective, longitudinal study of total joint replacement and its predictors in 1,600 patients with rheumatoid arthritis. Arthritis Rheum. 1998; 41:1072–82.

43. Pincus T, Callahan LF. What is the natural history of rheumatoid arthritis? Rheum Dis Clin North Am. 1993; 19:123–51.

44. Smolen JS, Aletaha D, Bijlsma JW, Breedveld FC, Boumpas D, Burmester G, et al. Treating rheumatoid arthritis to target: recommendations of an international task force. Ann Rheum Dis. 2010; 69:631–7.

45. van der Heijde DM, van't Hof MA, van Riel PL, Theunisse LA, Lubberts EW, van Leeuwen MA, et al. Judging disease activity in clinical practice in rheumatoid arthritis: first step in the development of a disease activity score. Ann Rheum Dis. 1990; 49:916–20.

46. Prevoo ML, van't Hof MA, Kuper HH, van Leeuwen MA, van de Putte LB, van Riel PL. Modified disease activity scores that include twenty-eight-joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum. 1995; 38:44–8.

47. Aletaha D, Smolen J. The Simplified Disease Activity Index (SDAI) and the Clinical Disease Activity Index (CDAI): a review of their usefulness and validity in rheumatoid arthritis. Clin Exp Rheumatol. 2005; 23(5 Suppl 39):S100–8.
48. Felson DT, Smolen JS, Wells G, Zhang B, van Tuyl LH, Funovits J, et al. American College of Rheumatology/ European League Against Rheumatism provisional definition of remission in rheumatoid arthritis for clinical trials. Arthritis Rheum. 2011; 63:573–86.
49. Felson DT, Anderson JJ, Boers M, Bombardier C, Furst D, Goldsmith C, et al. American College of Rheumatology. Preliminary definition of improvement in rheumatoid arthritis. Arthritis Rheum. 1995; 38:727–35.

50. Pincus T, Yazici Y, Sokka T. Quantitative measures of rheumatic diseases for clinical research versus standard clinical care: differences, advantages and limitations. Best Pract Res Clin Rheumatol. 2007; 21:601–28.

51. Klinkhoff AV, Bellamy N, Bombardier C, Carette S, Chalmers A, Esdaile JM, et al. An experiment in reducing interobserver variability of the examination for joint tenderness. J Rheumatol. 1988; 15:492–4.
52. Uhlig T, Kvien TK, Pincus T. Test-retest reliability of disease activity core set measures and indices in rheumatoid arthritis. Ann Rheum Dis. 2009; 68:972–5.

53. Tugwell P, Wells G, Strand V, Maetzel A, Bombardier C, Crawford B, et al. Clinical improvement as reflected in measures of function and health-related quality of life following treatment with leflunomide compared with methotrexate in patients with rheumatoid arthritis: sensitivity and relative efficiency to detect a treatment effect in a twelve-month, placebo-controlled trial. Leflunomide Rheumatoid Arthritis Investigators Group. Arthritis Rheum. 2000; 43:506–14.
54. Cohen SB, Strand V, Aguilar D, Ofman JJ. Patient-versus physician-reported outcomes in rheumatoid arthritis patients treated with recombinant interleukin-1 receptor antagonist (anakinra) therapy. Rheumatology (Oxford). 2004; 43:704–11.
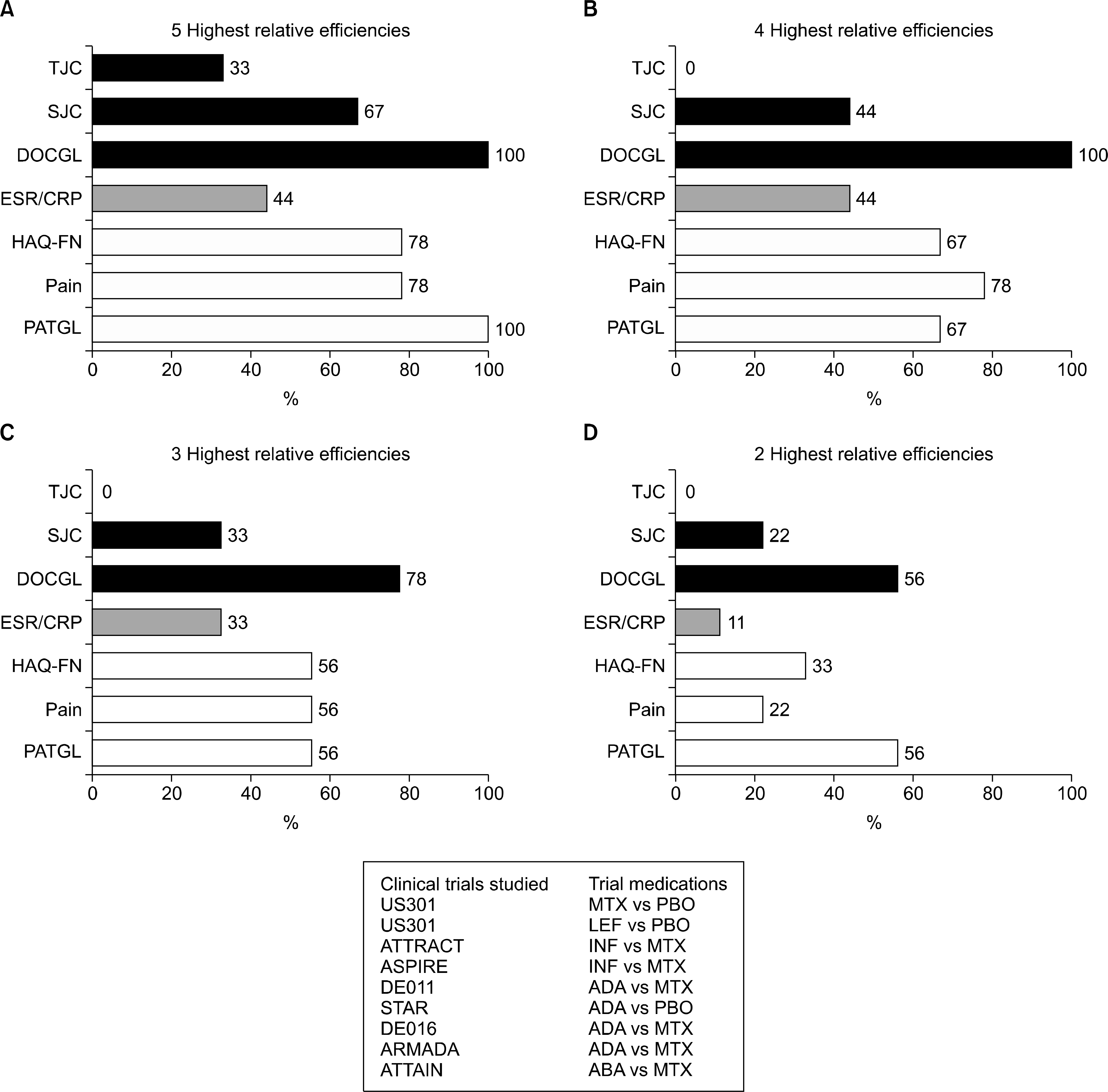
55. Pincus T, Richardson B, Strand V, Bergman MJ. Relative efficiencies of the 7 rheumatoid arthritis Core Data Set measures to distinguish active from control treatments in 9 comparisons from clinical trials of 5 agents. Clin Exp Rheumatol. 2014; 32(5 Suppl 85):S–47-54.
56. Scott DL, Grindulis KA, Struthers GR, Coulton BL, Popert AJ, Bacon PA. Progression of radiological changes in rheumatoid arthritis. Ann Rheum Dis. 1984; 43:8–17.

57. Østergaard M, Pedersen SJ, Døhn UM. Imaging in rheumatoid arthritis– status and recent advances for magnetic resonance imaging, ultrasonography, computed tomography and conventional radiography. Best Pract Res Clin Rheumatol. 2008; 22:1019–44.
58. Pincus T, Swearingen CJ, Luta G, Sokka T. Efficacy of prednisone 1-4 mg/day in patients with rheumatoid arthritis: a randomised, double-blind, placebo controlled withdrawal clinical trial. Ann Rheum Dis. 2009; 68:1715–20.

59. Anderson J, Caplan L, Yazdany J, Robbins ML, Neogi T, Michaud K, et al. Rheumatoid arthritis disease activity measures: American College of Rheumatology recommendations for use in clinical practice. Arthritis Care Res (Hoboken). 2012; 64:640–7.

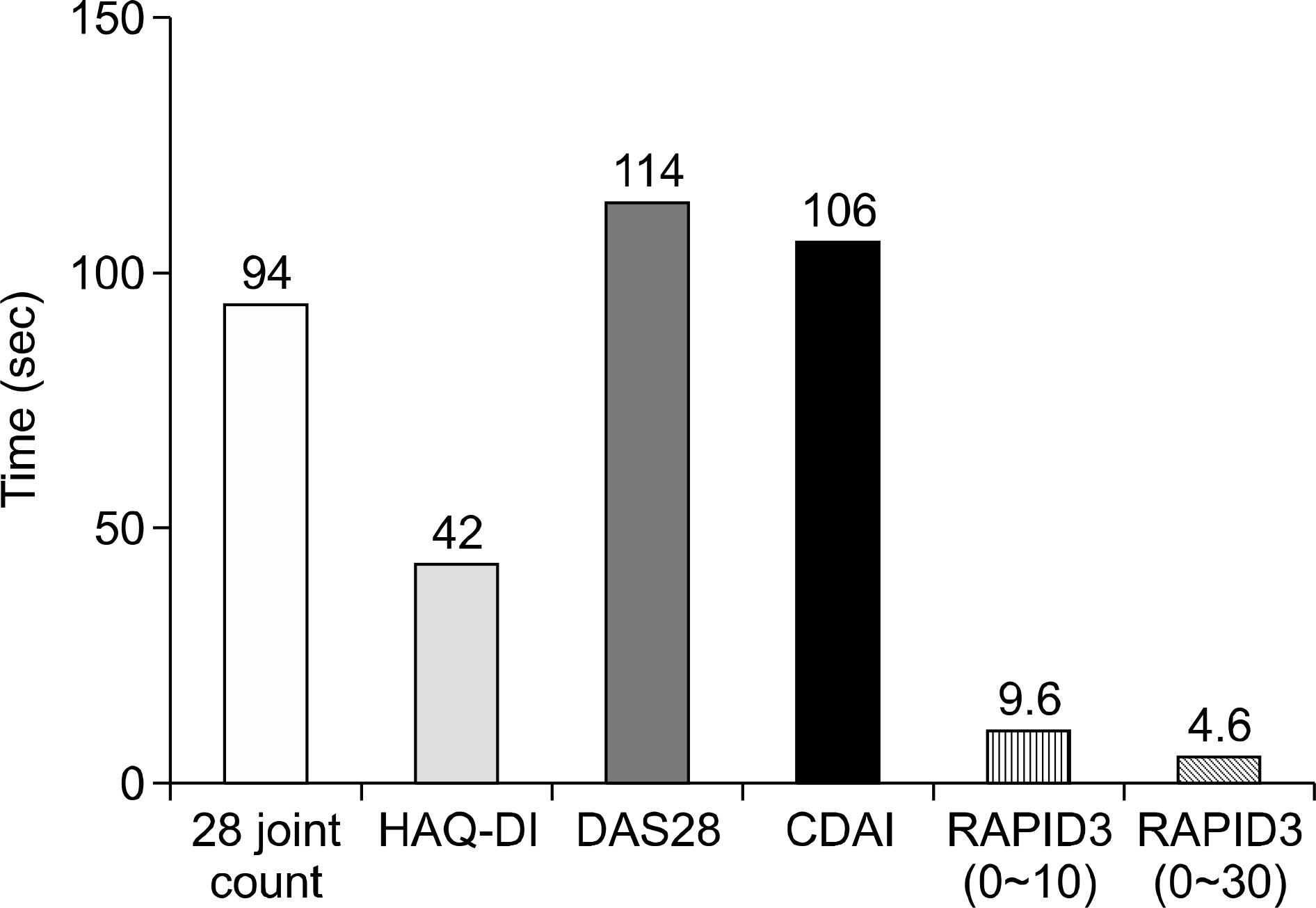
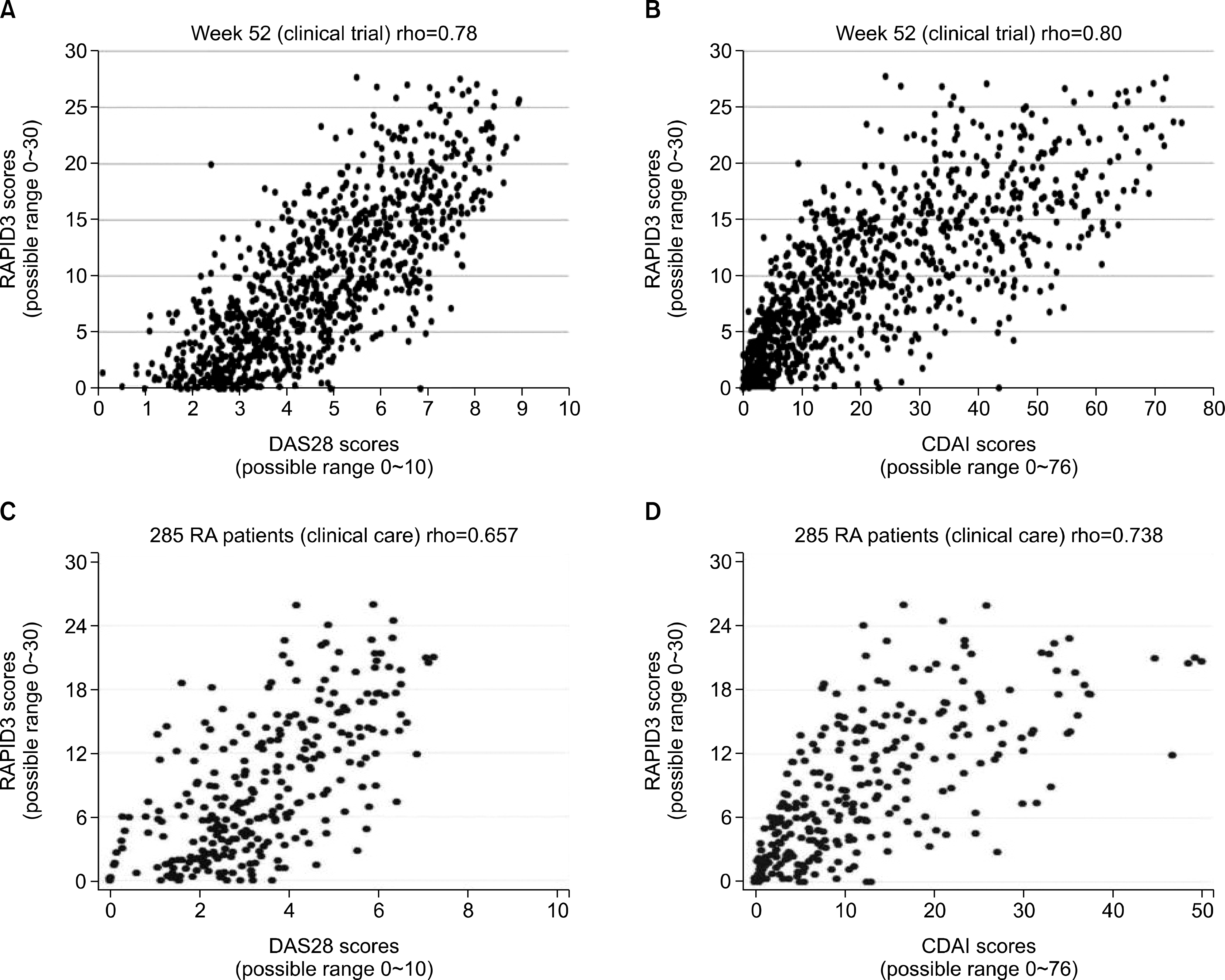
60. Pincus T, Swearingen CJ, Bergman MJ, Colglazier CL, Kaell AT, Kunath AM, et al. RAPID3 (Routine Assessment of Patient Index Data) on an MDHAQ (Multidimensional Health Assessment Questionnaire): agreement with DAS28 (Disease Activity Score) and CDAI (Clinical Disease Activity Index) activity categories, scored in five versus more than ninety seconds. Arthritis Care Res (Hoboken). 2010; 62:181–9.

61. Sackett DL, Rosenberg WM, Gray JA, Haynes RB, Richardson WS. Evidence based medicine: what it is and what it isn't. BMJ. 1996; 312:71–2.

62. Williams HJ, Ward JR, Reading JC, Brooks RH, Clegg DO, Skosey JL, et al. Comparison of auranofin, methotrexate, and the combination of both in the treatment of rheumatoid arthritis. A controlled clinical trial. Arthritis Rheum. 1992; 35:259–69.
63. Felson DT, Anderson JJ, Meenan RF. The comparative efficacy and toxicity of second-line drugs in rheumatoid arthritis. Results of two metaanalyses. Arthritis Rheum. 1990; 33:1449–61.

64. Pincus T, Marcum SB, Callahan LF. Longterm drug therapy for rheumatoid arthritis in seven rheumatology private practices: II. Second line drugs and prednisone. J Rheumatol. 1992; 19:1885–94.
65. Yazici Y, Sokka T, Kautiainen H, Swearingen C, Kulman I, Pincus T. Long term safety of methotrexate in routine clinical care: discontinuation is unusual and rarely the result of laboratory abnormalities. Ann Rheum Dis. 2005; 64:207–11.

66. Donahue KE, Gartlehner G, Jonas DE, Lux LJ, Thieda P, Jonas BL, et al. Systematic review: comparative effectiveness and harms of disease-modifying medications for rheumatoid arthritis. Ann Intern Med. 2008; 148:124–34.

67. Sokka T, Kautiainen H, Toloza S, Mäkinen H, Verstappen SM, Lund Hetland M, et al. QUEST-RA: quantitative clinical assessment of patients with rheumatoid arthritis seen in standard rheumatology care in 15 countries. Ann Rheum Dis. 2007; 66:1491–6.

68. Sokka T, Pincus T. Most patients receiving routine care for rheumatoid arthritis in 2001 did not meet inclusion criteria for most recent clinical trials or american college of rheumatology criteria for remission. J Rheumatol. 2003; 30:1138–46.
69. Sokka T, Pincus T. Eligibility of patients in routine care for major clinical trials of anti-tumor necrosis factor alpha agents in rheumatoid arthritis. Arthritis Rheum. 2003; 48:313–8.
70. Ruberman W, Weinblatt E, Goldberg JD, Chaudhary BS. Psychosocial influences on mortality after myocardial infarction. N Engl J Med. 1984; 311:552–9.

71. Pincus T. Limitations of traditional randomized controlled clinical trials in rheumatology. Yazici H, Yazici Y, Lesaffre E, editors. Understanding evidence based rheumatology – a guide to interpreting criteria, drugs, trials, registries, and ethics. New York: Springer International Publishing Switzerland;2014. p. 179–207.

72. Howick J. The philosophy of evidence-based medicine. Chichester, Wiley-Blackwell;. 2011.
73. Hampton JR, Harrison MJ, Mitchell JR, Prichard JS, Seymour C. Relative contributions of history-taking, physical examination, and laboratory investigation to diagnosis and management of medical outpatients. Br Med J. 1975; 2:486–9.

74. Sandler G. The importance of the history in the medical clinic and the cost of unnecessary tests. Am Heart J. 1980; 100:928–31.

75. Peterson MC, Holbrook JH, Von Hales D, Smith NL, Staker LV. Contributions of the history, physical examination, and laboratory investigation in making medical diagnoses. West J Med. 1992; 156:163–5.

76. Pryor DB, Shaw L, McCants CB, Lee KL, Mark DB, Harrell FE Jr, et al. Value of the history and physical in identifying patients at increased risk for coronary artery disease. Ann Intern Med. 1993; 118:81–90.

77. Fries JF, Spitz P, Kraines RG, Holman HR. Measurement of patient outcome in arthritis. Arthritis Rheum. 1980; 23:137–45.

78. Pincus T, Swearingen C, Wolfe F. Toward a multidimensional Health Assessment Questionnaire (MDHAQ): assessment of advanced activities of daily living and psychological status in the patient-friendly health assessment questionnaire format. Arthritis Rheum. 1999; 42:2220–30.

79. Pincus T, Sokka T, Kautiainen H. Further development of a physical function scale on a MDHAQ [corrected] for standard care of patients with rheumatic diseases. J Rheumatol. 2005; 32:1432–9.
80. Boers M, Tugwell P, Felson DT, van Riel PL, Kirwan JR, Edmonds JP, et al. World Health Organization and International League of Associations for Rheumatology core endpoints for symptom modifying antirheumatic drugs in rheumatoid arthritis clinical trials. J Rheumatol Suppl. 1994; 41:86–9.
81. Pincus T, Bergman M, Sokka T, Roth J, Swearingen C, Yazici Y. Visual analog scales in formats other than a 10 centimeter horizontal line to assess pain and other clinical data. J Rheumatol. 2008; 35:1550–8.
82. Pincus T, Bergman MJ, Yazici Y, Hines P, Raghupathi K, Maclean R. An index of only patient-reported outcome measures, routine assessment of patient index data 3 (RAPID3), in two abatacept clinical trials: similar results to disease activity score (DAS28) and other RAPID indices that include physician-reported measures. Rheumatology (Oxford). 2008; 47:345–9.

83. Pincus T, Swearingen CJ, Bergman M, Yazici Y. RAPID3 (Routine Assessment of Patient Index Data 3), a rheumatoid arthritis index without formal joint counts for routine care: proposed severity categories compared to disease activity score and clinical disease activity index categories. J Rheumatol. 2008; 35:2136–47.

84. Pincus T, Hines P, Bergman MJ, Yazici Y, Rosenblatt LC, MacLean R. Proposed severity and response criteria for Routine Assessment of Patient Index Data (RAPID3): results for categories of disease activity and response criteria in abatacept clinical trials. J Rheumatol. 2011; 38:2565–71.

85. Pincus T. Can RAPID3, an index without formal joint counts or laboratory tests, serve to guide rheumatologists in tight control of rheumatoid arthritis in usual clinical care? Bull NYU Hosp Jt Dis. 2009; 67:254–66.
86. DeWalt DA, Reed GW, Pincus T. Further clues to recognition of patients with fibromyalgia from a simple 2-page patient multidimensional health assessment questionnaire (MDHAQ). Clin Exp Rheumatol. 2004; 22:453–61.
87. Pincus T, Hassett AL, Callahan LF. Clues on the MDHAQ to identify patients with fibromyalgia and similar chronic pain conditions. Rheum Dis Clin North Am. 2009; 35:865–9.

88. Kirwan JR, Minnock P, Adebajo A, Bresnihan B, Choy E, de Wit M, et al. Patient perspective: fatigue as a recommended patient centered outcome measure in rheumatoid arthritis. J Rheumatol. 2007; 34:1174–7.
89. Sokka T, Pincus T. Poor physical function, pain and limited exercise: risk factors for premature mortality in the range of smoking or hypertension, identified on a simple patient self-report questionnaire for usual care. BMJ Open. 2011; 1:e000070.

90. Pincus T, Bergman MJ, Yazici Y. RAPID3-an index of physical function, pain, and global status as “vital signs” to improve care for people with chronic rheumatic diseases. Bull NYU Hosp Jt Dis. 2009; 67:211–25.
91. Hess EV, Fries JF, Klinenberg JR. A uniform database for rheumatic diseases. Arthritis Rheum. 1979; 22:1029–33.

92. Tugwell P, Boers M. Developing consensus on preliminary core efficacy endpoints for rheumatoid arthritis clinical trials. OMERACT Committee. J Rheumatol. 1993; 20:555–6.
93. Moses LE. The series of consecutive cases as a device for assessing outcomes of intervention. N Engl J Med. 1984; 311:705–10.

94. Yazici Y, Erkan D. Eligibility of rheumatoid arthritis patients seen in clinical practice for rheumatoid arthritis clinical trials: comment on the article by Sokka and Pincus. Arthritis Rheum. 2003; 48:3611. author reply 3613-5.

95. Ideker T, Galitski T, Hood L. A new approach to decoding life: systems biology. Annu Rev Genomics Hum Genet. 2001; 2:343–72.

96. Hood L, Flores M. A personal view on systems medicine and the emergence of proactive P4 medicine: predictive, preventive, personalized and participatory. N Biotechnol. 2012; 29:613–24.

97. Leigh JP, Fries JF. Mortality predictors among 263 patients with rheumatoid arthritis. J Rheumatol. 1991; 18:1307–12.
98. Wolfe F, Kleinheksel SM, Cathey MA, Hawley DJ, Spitz PW, Fries JF. The clinical value of the Stanford Health Assessment Questionnaire Functional Disability Index in patients with rheumatoid arthritis. J Rheumatol. 1988; 15:1480–8.
99. Söderlin MK, Nieminen P, Hakala M. Functional status predicts mortality in a community based rheumatoid arthritis population. J Rheumatol. 1998; 25:1895–9.
100. Sokka T, Abelson B, Pincus T. Mortality in rheumatoid arthritis: 2008 update. Clin Exp Rheumatol. 2008; 26(5 Suppl 51):S35–61.
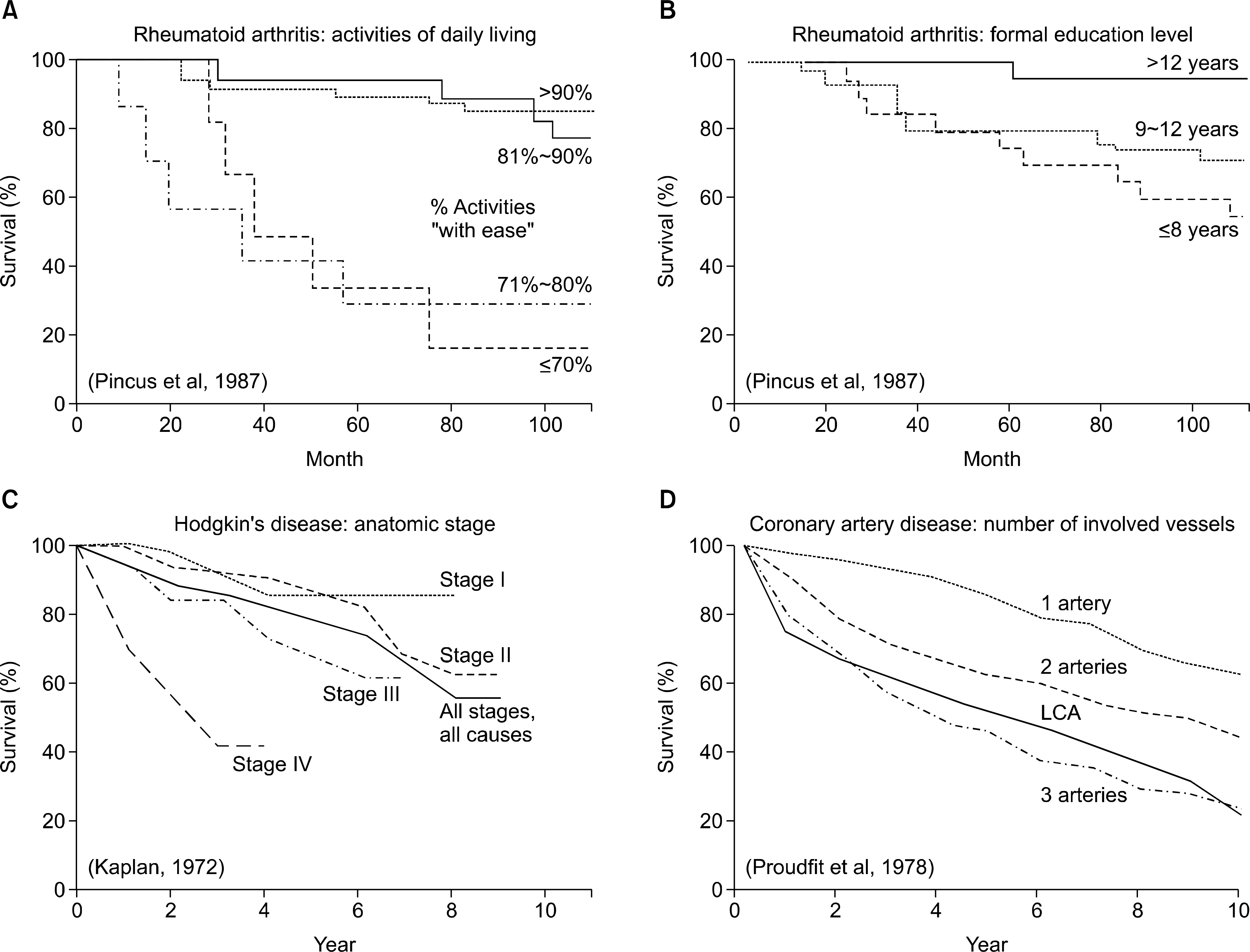
101. Pincus T, Callahan LF. Formal education as a marker for increased mortality and morbidity in rheumatoid arthritis. J Chronic Dis. 1985; 38:973–84.

102. Pincus T, Callahan LF. Taking mortality in rheumatoid arthritis seriously–predictive markers, socioeconomic status and comorbidity. J Rheumatol. 1986; 13:841–5.
103. Callahan LF, Pincus T. Formal education level as a significant marker of clinical status in rheumatoid arthritis. Arthritis Rheum. 1988; 31:1346–57.

104. Callahan LF, Smith WJ, Pincus T. Self-report questionnaires in five rheumatic diseases: comparisons of health status constructs and associations with formal education level. Arthritis Care Res. 1989; 2:122–31.

105. Kim HS, Jung UH, Lee H, Kim SK, Lee H, Choe JY, et al. Effect of formal education level on measurement of rheumatoid arthritis disease activity. J Rheum Dis. 2015; 22:231–7.

106. Pincus T, Amara I, Segurado OG, Bergman M, Koch GG. Relative efficiencies of physician/assessor global estimates and patient questionnaire measures are similar to or greater than joint counts to distinguish adalimumab from control treatments in rheumatoid arthritis clinical trials. J Rheumatol. 2008; 35:201–5.
107. Pincus T, Furer V, Keystone E, Yazici Y, Bergman MJ, Luijtens K. RAPID3 (Routine Assessment of Patient Index Data 3) severity categories and response criteria: Similar results to DAS28 (Disease Activity Score) and CDAI (Clinical Disease Activity Index) in the RAPID 1 (Rheumatoid Arthritis Prevention of Structural Damage) clinical trial of certolizumab pegol. Arthritis Care Res (Hoboken). 2011; 63:1142–9.
108. Pincus T, Strand V, Koch G, Amara I, Crawford B, Wolfe F, et al. An index of the three core data set patient questionnaire measures distinguishes efficacy of active treatment from that of placebo as effectively as the American College of Rheumatology 20% response criteria (ACR20) or the Disease Activity Score (DAS) in a rheumatoid arthritis clinical trial. Arthritis Rheum. 2003; 48:625–30.

109. Pincus T, Chung C, Segurado OG, Amara I, Koch GG. An index of patient reported outcomes (PRO-Index) discriminates effectively between active and control treatment in 4 clinical trials of adalimumab in rheumatoid arthritis. J Rheumatol. 2006; 33:2146–52.
110. Hart LE, Tugwell P, Buchanan WW, Norman GR, Grace EM, Southwell D. Grading of tenderness as a source of interrater error in the Ritchie articular index. J Rheumatol. 1985; 12:716–7.
111. Lewis PA, O'Sullivan MM, Rumfeld WR, Coles EC, Jessop JD. Significant changes in Ritchie scores. Br J Rheumatol. 1988; 27:32–6.

112. Scott DL, Choy EH, Greeves A, Isenberg D, Kassinor D, Rankin E, et al. Standardising joint assessment in rheumatoid arthritis. Clin Rheumatol. 1996; 15:579–82.

113. Pincus T, Castrejón I. Are patient self-report questionnaires as “scientific” as biomarkers in “treat-to-target” and prognosis in rheumatoid arthritis? Curr Pharm Des. 2015; 21:241–56.

114. Pincus T. RAPID3, an index of only 3 patient self-report core data set measures, but not ESR, recognizes incomplete responses to methotrexate in usual care of patients with rheumatoid arthritis. Bull Hosp Jt Dis. 2013; 71:117–20.
115. Castrejón I, Dougados M, Combe B, Guillemin F, Fautrel B, Pincus T. Can remission in rheumatoid arthritis be assessed without laboratory tests or a formal joint count? possible remission criteria based on a self-report RAPID3 score and careful joint examination in the ESPOIR cohort. J Rheumatol. 2013; 40:386–93.

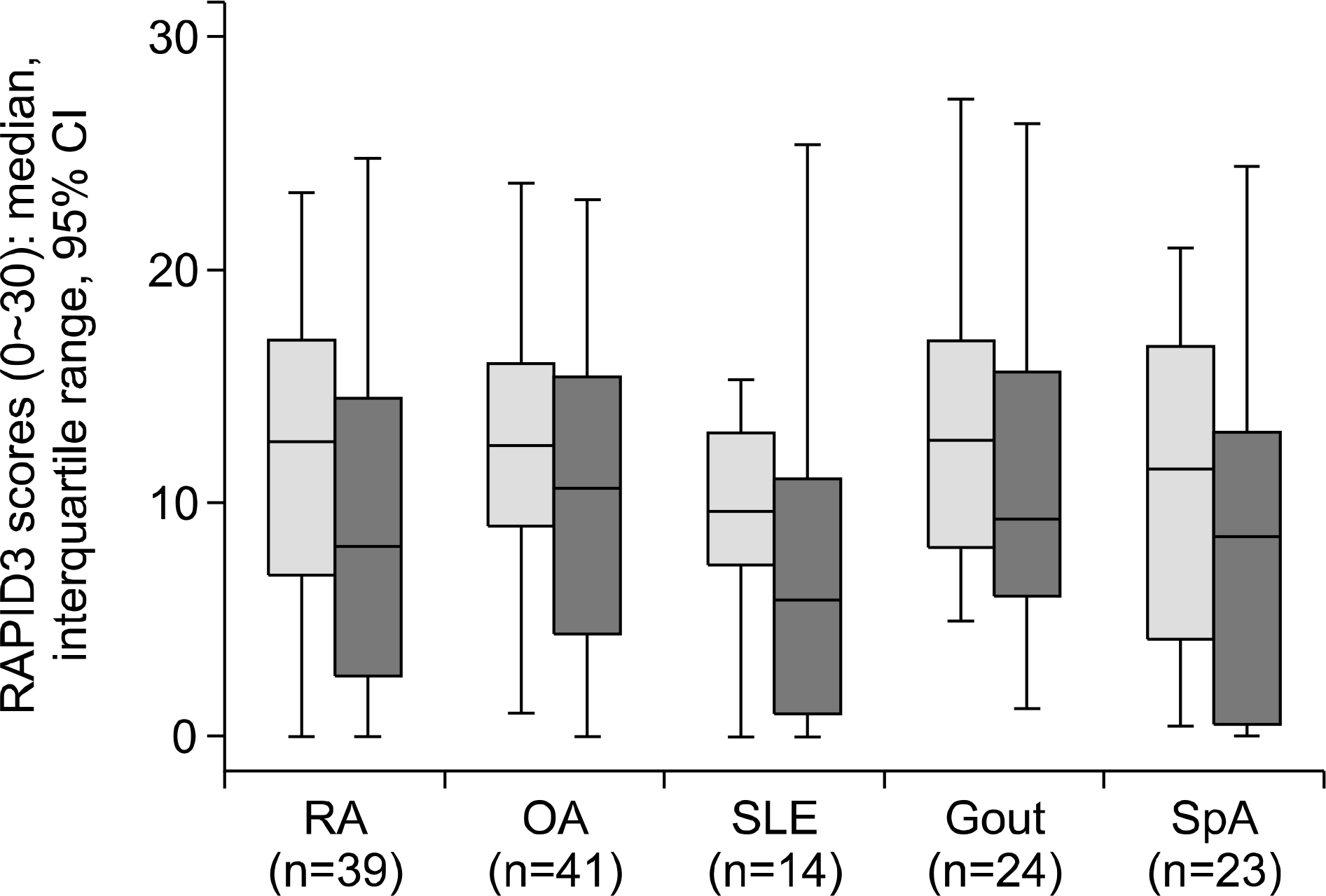
116. Castrejón I, Bergman MJ, Pincus T. MDHAQ/RAPID3 to recognize improvement over 2 months in usual care of patients with osteoarthritis, systemic lupus erythematosus, spondyloarthropathy, and gout, as well as rheumatoid arthritis. J Clin Rheumatol. 2013; 19:169–74.

117. Askanase AD, Castrejón I, Pincus T. Quantitative data for care of patients with systemic lupus erythematosus in usual clinical settings: a patient Multidimensional Health Assessment Questionnaire and physician estimate of noninflammatory symptoms. J Rheumatol. 2011; 38:1309–16.

118. Danve A, Reddy A, Vakil-Gilani K, Garg N, Dinno A, Deodhar A. Routine Assessment of Patient Index Data 3 score (RAPID3) correlates well with Bath Ankylosing Spondylitis Disease Activity index (BASDAI) in the assessment of disease activity and monitoring progression of axial spondyloarthritis. Clin Rheumatol. 2015; 34:117–24.

119. Cinar M, Yilmaz S, Cinar FI, Koca SS, Erdem H, Pay S, et al. A patient-reported outcome measures-based composite index (RAPID3) for the assessment of disease activity in ankylosing spondylitis. Rheumatol Int. 2015; 35:1575–80.

120. Michelsen B, Fiane R, Diamantopoulos AP, Soldal DM, Hansen IJ, Sokka T, et al. A comparison of disease burden in rheumatoid arthritis, psoriatic arthritis and axial spondyloarthritis. PLoS One. 2015; 10:e0123582.

121. Park SH, Choe JY, Kim SK, Lee H, Castrejón I, Pincus T. Routine Assessment of Patient Index Data (RAPID3) and Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) scores yield similar information in 85 Korean patients with ankylosing spondylitis seen in usual clinical care. J Clin Rheumatol. 2015; 21:300–4.

122. Annapureddy N, Elsallabi O, Baker J, Sreih AG. Patient-reported outcomes in ANCA-associated vasculitis. A comparison between Birmingham Vasculitis Activity Score and routine assessment of patient index data 3. Clin Rheumatol. 2016; 35:395–400.

123. Pincus T, Askanase AD, Swearingen CJ. A multi-dimensional health assessment questionnaire (MDHAQ) and routine assessment of patient index data (RAPID3) scores are informative in patients with all rheumatic diseases. Rheum Dis Clin North Am. 2009; 35:819–27.






 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download