Abstract
Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS) are severe cutaneous adverse reactions (SCAR) which are majorly caused by drugs. Though the incidence rate is low, SCAR sometimes can be life-threatening and leads to lifelong sequelae. Many pharmacogenomic associations in immune and nonimmune related genes with the development of SCAR have been discovered recently and the pharmacogenetic tests have been applied to prevent specific drug-induced SCAR. In this review, we discuss the recent advances of pharmacogenomics in SCAR.
Severe cutaneous adverse reactions (SCAR), including Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS) are known to have potential lethality and lifelong sequelae. SJS and TEN are delayed type mucocutaneous immune reactions with the involvement of widespread keratinocyte apoptosis. The clinical difference of SJS and TEN depends on their extent of epidermal detachment [1]. SJS, SJS/TEN overlap, and TEN can be classified as the degree of skin detachment involving less than 10%, 10-30%, and greater than 30% of body surface area, respectively [2, 3]. The manifestations of SJS/TEN range from mild exanthematous skin rashes to a large amount of bullae and extensive mucocutaneous sloughing [4, 5, 6, 7]. Unlike SJS/TEN, DRESS usually involves less skin manifestations but more internal organ presentation and hematological abnormalities [2, 8]. In addition to typical eosinophilia, atypical lymphocytes, and hepatitis, recent clinical histopathological findings have showed that the dyskeratosis, epidermal spongiosis, and severe interface vacuolization are significant in DRESS cases [9].
SCAR accounts for about 2% of hospital admissions [10] with varying incidence between 2 and 7 cases/million/yr in SJS/TEN [11, 12, 13, 14, 15] and 1/1,000 to 1/10,000 offending agent exposures in DRESS [16]. In spite of the low incidence, the mortality rate is 5-10% for SJS, about 30% for SJS/TEN overlap, nearly 50% for TEN [17, 18, 19, 20, 21], and 10% for DRESS [22]. Unfortunately, a consensus treatment guideline remains unavailable. Intravenous immunoglobulin (IVIG) [23, 24], cyclosporine [25, 26, 27] and systemic corticosteroids [28, 29] have been used to treat patients with SJS/TEN, yet a lack of consensus on the outcome is observed. Supportive care remains the most frequent monotherapy with proven benefit on mortality [30], although patients with SJS/TEN usually receive the combination treatment of IVIG, corticosteroids [31] or cyclosporine [27]. Recently, studies on the immunological mechanism of SJS/TEN have suggested that immunosuppressive agents, such as anti-tumor necrolysis factor-alpha (TNF-α) [23], anti-Fas ligand (FasL) monoclonal antibody, or perforin/granzyme B inhibitors [32] might be useful for treating SJS/TEN; however, the real beneficial effects are not known due to lack of controlled clinical trial.
Previously, we found that the secretory granulysin acts as a primary cytotoxic molecule that leads to the disseminate keratinocyte apoptosis in SJS/TEN [33]. Granulysin is produced by cytotoxic T lymphocytes (CTLs), natural killer, and natural killer T cells, and released into the extracellular space alone with other immune mediators (e.g., soluble FasL, granzymeB, and perforin) [33, 34, 35]. We found that depletion of granulysin by antigranulysin antibodies markedly decreased apoptosis of keratinocytes induced by blister fluids from patients with SJS or TEN [33], suggesting a potential of antigranulysin for therapeutic purposes in the future.
In this article, we primarily review the current advances in exploring the genetic predisposing factors and pathogenic mechanism of SCAR induced by drugs.
Although several proposed hypotheses have been reported to explain how small molecule synthetic compounds are recognized by T cells in a major histocompatibility complex-dependent fashion, the pharmaco-immune concept remains the one that has been widely recognized. It illustrates a direct pharmacological interaction of a drug with immune receptors. Upon the noncovalent binding of an offending drug presented by a human leukocyte antigen (HLA) molecule to the CD8+ T-cell receptor (TCR), the HLA-drug-TCR may initiate a series of reactions, resulting in an expansion of CTLs [36, 37], the releases of cytotoxic proteins [24, 33, 38], and induction of keratinocyte apoptosis in SJS/TEN. Recently, we also identified shared and restricted TCR usage in carbamazepine (CBZ)-induced SJS/TEN patients [39] and demonstrated that the endogenous peptide-loaded HLA-B*15:02 molecule presented CBZ to CTLs without the involvement of intracellular drug metabolism or antigen processing [40]. More importantly, we found that granulysin acts as a major "killer" and is responsible for keratinocyte death [33] (Fig. 1).
In the ensuing decade, a number of important pharmacogenomic associations with SCAR have been found in different populations. In 1987, the genetic susceptibility was first being connected by the finding of HLA B12 and oxicam- and sulfonamide-related TEN [41]. Since then, many of the genetic associations have been discovered, such as HLA-B*57:01 and abacavir-induced hypersensitivity in European [42], and Thai and Cambodian children [43], HLA-B*58:01 and allopurinol-induced SJS/TEN in Han Chinese [44], Thai [45], Japanese [46], Korean [47], and European [48], HLA-B*15:02 and CBZ-SJS/TEN in Han Chinese [49, 50], Thai [51], Indian [52], and Malaysian [53], HLA-B*15:11 and CBZ-SJS/TEN in Japanese [54], Korean [55], and Han Chinese [56, 57], HLA-B*59:01 and CBZ-SJS/TEN in Japanese [58], HLA-A*31:01 and CBZ-SJS/TEN/drug-induced hypersensitivity syndrome in Japanese [59, 60], and CBZ-maculopapular exanthema (MPE)/DRESS in Han Chinese [61, 62], European [62, 63], HLA-B*13:01 and dapsone induced hypersensitivty in Han Chinese [64], HLA-B*15:02 and lamotrigine-SJS/TEN in Han Chinese [65, 66], HLA-B*15:02 and phenytoin (PHT)-SJS/TEN in Thai [51], HLA-B*59:01 and methazolamide-SJS/TEN in Korean and Japanese [67], HLA-DRB1*01:01 and nevirapine hypersensitivity in Australian [68] and French [69], HLA-B*14:02 (or HLA-B*14:02-Cw*08:02 haplotype)-nevirapine hypersensitivity in Sardinian [70], HLA-B*35:05 and nevirapine-MPE/DRESS in Thai [71], HLA-Cw*08:01 or 08:02 and navirapine hypersensitivity in Sardinian [70] and Japanese [72], HLA-B*73 and oxicam-SJS/TEN [48], HLA-A2 and B12 and oxicam-TEN [41], HLA-A29, B12, and DR7 and oxicam-TEN sulfonamide-TEN [41], HLA-B*15:02 and oxcarbazepine-SJS/TEN [66].
The genomic associations in HLA with drug hypersensitivity can be restricted to specific phenotypes. Our recent studies demonstrated a variable strength of association between the HLA genotypes and a wide spectrum of phenotypes of the CBZ hypersensitivity in Han Chinese. The most powerful CBZ-HLA association was the B*15:02 in SJS/TEN, and the A*31:01 and B*51:01 in MPE/DRESS [57]. In addition, we also found a correlation between the strength of HLA-B*15:02 association and degree of skin detachment in CBZ-SJS/TEN [57]. With international collaboration with RegiSCAR consortium containing European and Chinese populations, we further clarified that the HLA-A*31:01 is a genetic predictor for CBZ-DRESS only, but not for CBZ-SJS/TEN [62].
Moreover, the associations of HLA and drugs-induced SCAR can be also different significantly in different ethnic populations. For example, studies did not show that patients with CBZ-SJS/TEN significantly associated with HLA-B*15:02 in Europeans; however, this connection was still present in Han Chinese ancestry of Europeans. The ethnic difference can be tracked by the historical evolution and we did found an interesting fact in Asia. Although a general and historical cognition interprets that the Korean and Japanese ancestors originated from China, the HLA-B*15:02 appears to be present at a low frequency (<1%) there [46, 55, 73]. U.S. Food and Drug Administration as well as many Asian health administrations have recommended HLA-B*15:02 screening for CBZ new users of Asian ancestry since 2007 [74].
Different from the specificity of HLA-B*15:02 in Han Chinese, B*15:11 and A*31:01 had been reported to be predominantly associated with CBZ-SJS and HSS/SCAR respectively in Korean population [55]. Various HLA allele frequencies in worldwide populations decide the specificity of the ethnicity-related association. The frequency of HLA-B*15:02 allele is about 1-8% in the countries of Southeast Asia, whereas it is rare or absent in Northeast Asian countries [55] or Caucasian [75]. The worldwide distribution of the associations between the causative drugs and HLA is illustrated in Fig. 2.
Other than the genetic association of SCAR with HLA, recent evidence also showed that the death of keratinocytes in SCAR may be contributed by the annexin A1-formyl peptide receptor 1 (FPR1). This study demonstrated that conditioned media from peripheral blood mononuclear cells (PBMCs) that had been exposed to the causative drug from patients with SJS/TEN induced the death of SJS/TEN keratinocytes, whereas that from PBMCs of patients with ordinary drug skin reactions (ODSRs) exposed to the same drug did not. Keratinocytes from ODSR patients or from healthy controls were unaffected by the conditioned media from SJS/TEN or ODSR PBMCs. Further investigations identified that the annexin A1 was associated with the cytotoxic level via the binding with its receptor FPR1 in keratinocytes, leading to the keratinocyte necroptosis in SJS/TEN [76].
Though the HLA predisposition plays a critical role in druginduced SCAR, other factors like individual differences in drug clearance or metabolism may also contribute to SCAR development, recovery or prognosis. As knowing that the drug clearance is important for preventing further damage from the retention of drug toxicity in the body, drug metabolism becomes another dependent key for the SCAR development.
Recent evidences show that impaired drug metabolism may also involve in the pathogenesis of SCAR. Our recent study identified that a variant of CYP2C locus is related to PHT induced SCAR. From a genome-wide association study encompassing 105 cases with PHT-related SCAR, we identified 16 significant singlenucleotide polymorphisms in CYP2C genes at 10q23.33. Further direct sequencing showed that CYP2C9*3 which reduce CYP2C9 enzymatic activity was significantly associated with PHT-induced SCAR. PHT-induced SCAR and patients who carried CYP2C9*3 showed a delayed clearance of plasma PHT [77]. CYP2C9*3 has been known to be related to the drug metabolism, and it can attenuate the clearance of PHT [78, 79]. Previous study also showed an association of CYP2C9*3 with PHT-induced MPE in Korean [80]. Another evidence showing genetic variability in a metabolizing enzyme can also contribute to SCAR was nevirapine-induced SJS/TEN that CYP2B6 G516T and T983C single nucleotide polymorphisms were found to be associated with SJS/TEN susceptibility [81] (Table 1).
Allopurinol is a widely used drug for treating goat and hyperuricaemia and is notorious for its potential risk to induce SCAR. Although HLA-B*5801 is strongly associated with allopurinol-induced SCAR, but the low positive predictive value of HLA-B*5801 suggesting that other factors are also involved in the pathogenesis of allopurinol-induced SCAR [44, 82, 83]. We recently revealed that the renal insufficiency directly affected the excretion of plasma oxypurinol, an active metabolite of allopurinol that is majorly eliminated through the kidney, and sustained high levels of oxypurinol in the plasma of patients with allopurinol-induced SCAR. An analysis between the allopurinol-SCAR cases and the tolerant controls showed that the impaired renal function was also significantly associated with allopurinol-SCAR. In addition, we also found that the high plasma levels of granulysin and oxypurinol after allopurinol discontinuation correlated with the high mortality in allopurinol-SJS/TEN and prolonged cutaneous reactions in allopurinol-DRESS [84]. We interpreted that the sustained and high levels of plasma oxypurinol may provoke CTLs and subsequently induce stronger immune reactions, therefore causing the prolonged disease remission, poor outcomes, and higher mortality in SCAR patients.
Recent advance in pharmacogenomic researches has improved our understanding of SCAR in recent years and provides physicians and scientists a direction to deepen the mechanisms from aspects of pathology, immunology, and genetics. The findings of pharmacogenomic markers for SCAR have successfully implicated for clinical practice in many countries and prevented high-risk patients from SCAR-induced by some risk medicines, such as CBZ and allopurinol.
Figures and Tables
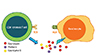 | Fig. 1Mechanisms of keratinocyte apoptosis in stevens-Johnson syndrome/toxic epidermal necrolysis. The complex of drug antigen/metabolite-human leukocyte antigen (HLA)-T cell receptor (TCR) initiates immune reactions. Specific HLA allele predisposition and interaction with culprit drug allow the presentation of drug-bound HLA to the TCR which further triggers the activation of CD8+ cytotoxic lymphocytes and a series of specific immune reactions. |
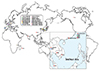 | Fig. 2Worldwide distribution of causative drugs and human leukocyte antigen (HLA) association in Stevens-Johnson syndrome/toxic epidermal necrolysis. ALL, allopurinol; CBZ, carbamazepine; LMT, lamotrigine; MTZ, methazolamide; NVP, nevirapine; OXC, oxcarbazepine; PHT, phenytoin; SMX, sulfamethoxazole. |
References
1. Bastuji-Garin S, Zahedi M, Guillaume JC, Roujeau JC. Toxic epidermal necrolysis (Lyell syndrome) in 77 elderly patients. Age Ageing. 1993; 22:450–456.

2. Roujeau JC, Stern RS. Severe adverse cutaneous reactions to drugs. N Engl J Med. 1994; 331:1272–1285.

3. Bastuji-Garin S, Rzany B, Stern RS, Shear NH, Naldi L, Roujeau JC. Clinical classification of cases of toxic epidermal necrolysis, Stevens-Johnson syndrome, and erythema multiforme. Arch Dermatol. 1993; 129:92–96.

4. Wong KC, Kennedy PJ, Lee S. Clinical manifestations and outcomes in 17 cases of Stevens-Johnson syndrome and toxic epidermal necrolysis. Australas J Dermatol. 1999; 40:131–134.

5. Roujeau JC. The spectrum of Stevens-Johnson syndrome and toxic epidermal necrolysis: a clinical classification. J Invest Dermatol. 1994; 102:28S–30S.

6. Auquier-Dunant A, Mockenhaupt M, Naldi L, Correia O, Schroder W, Roujeau JC. SCAR Study Group. Severe Cutaneous Adverse Reactions. Correlations between clinical patterns and causes of erythema multiforme majus, Stevens-Johnson syndrome, and toxic epidermal necrolysis: results of an international prospective study. Arch Dermatol. 2002; 138:1019–1024.

7. Kardaun SH, Sidoroff A, Valeyrie-Allanore L, Halevy S, Davidovici BB, Mockenhaupt M, Roujeau JC. Variability in the clinical pattern of cutaneous side-effects of drugs with systemic symptoms: does a DRESS syndrome really exist? Br J Dermatol. 2007; 156:609–611.

8. Bocquet H, Bagot M, Roujeau JC. Drug-induced pseudolymphoma and drug hypersensitivity syndrome (Drug Rash with Eosinophilia and Systemic Symptoms: DRESS). Semin Cutan Med Surg. 1996; 15:250–257.

9. Chi MH, Hui RC, Yang CH, Lin JY, Lin YT, Ho HC, Chung WH, Kuo TT. Histopathological analysis and clinical correlation of drug reaction with eosinophilia and systemic symptoms (DRESS). Br J Dermatol. 2014; 170:866–873.

10. Valeyrie-Allanore L, Sassolas B, Roujeau JC. Drug-induced skin, nail and hair disorders. Drug Saf. 2007; 30:1011–1030.

11. Sekula P, Dunant A, Mockenhaupt M, Naldi L, Bouwes Bavinck JN, Halevy S, Kardaun S, Sidoroff A, Liss Y, Schumacher M, Roujeau JC. RegiSCAR study group. Comprehensive survival analysis of a cohort of patients with Stevens-Johnson syndrome and toxic epidermal necrolysis. J Invest Dermatol. 2013; 133:1197–1204.

12. Sassolas B, Haddad C, Mockenhaupt M, Dunant A, Liss Y, Bork K, Haustein UF, Vieluf D, Roujeau JC, Le Louet H. ALDEN, an algorithm for assessment of drug causality in Stevens-Johnson Syndrome and toxic epidermal necrolysis: comparison with case-control analysis. Clin Pharmacol Ther. 2010; 88:60–68.

13. Mockenhaupt M, Viboud C, Dunant A, Naldi L, Halevy S, Bouwes Bavinck JN, Sidoroff A, Schneck J, Roujeau JC, Flahault A. Stevens-Johnson syndrome and toxic epidermal necrolysis: assessment of medication risks with emphasis on recently marketed drugs. The EuroSCAR-study. J Invest Dermatol. 2008; 128:35–44.

14. Halevy S, Ghislain PD, Mockenhaupt M, Fagot JP, Bouwes Bavinck JN, Sidoroff A, Naldi L, Dunant A, Viboud C, Roujeau JC. EuroSCAR Study Group. Allopurinol is the most common cause of Stevens-Johnson syndrome and toxic epidermal necrolysis in Europe and Israel. J Am Acad Dermatol. 2008; 58:25–32.

15. Levi N, Bastuji-Garin S, Mockenhaupt M, Roujeau JC, Flahault A, Kelly JP, Martin E, Kaufman DW, Maison P. Medications as risk factors of Stevens-Johnson syndrome and toxic epidermal necrolysis in children: a pooled analysis. Pediatrics. 2009; 123:e297–e304.

16. Amante MF, Filippini AV, Cejas N, Lendoire J, Imventarza O, Parisi C. Dress syndrome and fulminant hepatic failure induced by lamotrigine. Ann Hepatol. 2009; 8:75–77.

17. Pereira FA, Mudgil AV, Rosmarin DM. Toxic epidermal necrolysis. J Am Acad Dermatol. 2007; 56:181–200.

18. Borchers AT, Lee JL, Naguwa SM, Cheema GS, Gershwin ME. Stevens-Johnson syndrome and toxic epidermal necrolysis. Autoimmun Rev. 2008; 7:598–605.

19. Trent J, Halem M, French LE, Kerdel F. Toxic epidermal necrolysis and intravenous immunoglobulin: a review. Semin Cutan Med Surg. 2006; 25:91–93.

20. Revuz J, Penso D, Roujeau JC, Guillaume JC, Payne CR, Wechsler J, Touraine R. Toxic epidermal necrolysis. Clinical findings and prognosis factors in 87 patients. Arch Dermatol. 1987; 123:1160–1165.

21. Schopf E, Stuhmer A, Rzany B, Victor N, Zentgraf R, Kapp JF. Toxic epidermal necrolysis and Stevens-Johnson syndrome. An epidemiologic study from West Germany. Arch Dermatol. 1991; 127:839–842.

22. Cacoub P, Musette P, Descamps V, Meyer O, Speirs C, Finzi L, Roujeau JC. The DRESS syndrome: a literature review. Am J Med. 2011; 124:588–597.

23. Paquet P, Pierard GE. New insights in toxic epidermal necrolysis (Lyell's syndrome): clinical considerations, pathobiology and targeted treatments revisited. Drug Saf. 2010; 33:189–212.
24. Viard I, Wehrli P, Bullani R, Schneider P, Holler N, Salomon D, Hunziker T, Saurat JH, Tschopp J, French LE. Inhibition of toxic epidermal necrolysis by blockade of CD95 with human intravenous immunoglobulin. Science. 1998; 282:490–493.

25. Sullivan JR, Watson A. Lamotrigine-induced toxic epidermal necrolysis treated with intravenous cyclosporin: a discussion of pathogenesis and immunosuppressive management. Australas J Dermatol. 1996; 37:208–212.

26. Rai R, Srinivas CR. Suprapharmacologic doses of intravenous dexamethasone followed by cyclosporine in the treatment of toxic epidermal necrolysis. Indian J Dermatol Venereol Leprol. 2008; 74:263–265.

27. Singh GK, Chatterjee M, Verma R. Cyclosporine in Stevens Johnson syndrome and toxic epidermal necrolysis and retrospective comparison with systemic corticosteroid. Indian J Dermatol Venereol Leprol. 2013; 79:686–692.

28. Lissia M, Mulas P, Bulla A, Rubino C. Toxic epidermal necrolysis (Lyell's disease). Burns. 2010; 36:152–163.

29. Fromowitz JS, Ramos-Caro FA, Flowers FP. University of Florida. Practical guidelines for the management of toxic epidermal necrolysis and Stevens-Johnson syndrome. Int J Dermatol. 2007; 46:1092–1094.

30. Schneck J, Fagot JP, Sekula P, Sassolas B, Roujeau JC, Mockenhaupt M. Effects of treatments on the mortality of Stevens-Johnson syndrome and toxic epidermal necrolysis: a retrospective study on patients included in the prospective EuroSCAR Study. J Am Acad Dermatol. 2008; 58:33–40.

31. Mittmann N, Chan B, Knowles S, Cosentino L, Shear N. Intravenous immunoglobulin use in patients with toxic epidermal necrolysis and Stevens-Johnson syndrome. Am J Clin Dermatol. 2006; 7:359–368.

32. Murata J, Abe R, Shimizu H. Increased soluble Fas ligand levels in patients with Stevens-Johnson syndrome and toxic epidermal necrolysis preceding skin detachment. J Allergy Clin Immunol. 2008; 122:992–1000.

33. Chung WH, Hung SI, Yang JY, Su SC, Huang SP, Wei CY, Chin SW, Chiou CC, Chu SC, Ho HC, Yang CH, Lu CF, Wu JY, Liao YD, Chen YT. Granulysin is a key mediator for disseminated keratinocyte death in Stevens-Johnson syndrome and toxic epidermal necrolysis. Nat Med. 2008; 14:1343–1350.

34. Gamen S, Hanson DA, Kaspar A, Naval J, Krensky AM, Anel A. Granulysin-induced apoptosis. I. Involvement of at least two distinct pathways. J Immunol. 1998; 161:1758–1764.
35. Deng A, Chen S, Li Q, Lyu SC, Clayberger C, Krensky AM. Granulysin, a cytolytic molecule, is also a chemoattractant and proinflammatory activator. J Immunol. 2005; 174:5243–5248.

36. Pichler WJ. Pharmacological interaction of drugs with antigenspecific immune receptors: the p-i concept. Curr Opin Allergy Clin Immunol. 2002; 2:301–305.

37. Naisbitt DJ, Britschgi M, Wong G, Farrell J, Depta JP, Chadwick DW, Pichler WJ, Pirmohamed M, Park BK. Hypersensitivity reactions to carbamazepine: characterization of the specificity, phenotype, and cytokine profile of drug-specific T cell clones. Mol Pharmacol. 2003; 63:732–741.

38. David V, Bourge JF, Guglielmi P, Mathieu-Mahul D, Degos L, Bensussan A. Human T cell clones use a CD3-associated surface antigen recognition structure to exhibit both NK-like and allogeneic cytotoxic reactivity. J Immunol. 1987; 138:2831–2836.
39. Ko TM, Chung WH, Wei CY, Shih HY, Chen JK, Lin CH, Chen YT, Hung SI. Shared and restricted T-cell receptor use is crucial for carbamazepine-induced Stevens-Johnson syndrome. J Allergy Clin Immunol. 2011; 128:1266–1276.e11.

40. Wei CY, Chung WH, Huang HW, Chen YT, Hung SI. Direct interaction between HLA-B and carbamazepine activates T cells in patients with Stevens-Johnson syndrome. J Allergy Clin Immunol. 2012; 129:1562–1569.e5.

41. Roujeau JC, Huynh TN, Bracq C, Guillaume JC, Revuz J, Touraine R. Genetic susceptibility to toxic epidermal necrolysis. Arch Dermatol. 1987; 123:1171–1173.

42. Mallal S, Phillips E, Carosi G, Molina JM, Workman C, Tomazic J, Jagel-Guedes E, Rugina S, Kozyrev O, Cid JF, Hay P, Nolan D, Hughes S, Hughes A, Ryan S, Fitch N, Thorborn D, Benbow A. PREDICT-1 Study Team. HLA-B*5701 screening for hypersensitivity to abacavir. N Engl J Med. 2008; 358:568–579.
43. Puthanakit T, Bunupuradah T, Kosalaraksa P, Vibol U, Hansudewechakul R, Ubolyam S, Suwanlerk T, Kanjanavanit S, Ngampiyaskul C, Wongsawat J, Luesomboon W, Vonthanak S, Ananworanich J, Ruxrungtham K. PREDICT Study Group. Prevalence of human leukocyte antigen-B*5701 among HIV-infected children in Thailand and Cambodia: implications for abacavir use. Pediatr Infect Dis J. 2013; 32:252–253.
44. Hung SI, Chung WH, Liou LB, Chu CC, Lin M, Huang HP, Lin YL, Lan JL, Yang LC, Hong HS, Chen MJ, Lai PC, Wu MS, Chu CY, Wang KH, Chen CH, Fann CS, Wu JY, Chen YT. HLA-B*5801 allele as a genetic marker for severe cutaneous adverse reactions caused by allopurinol. Proc Natl Acad Sci U S A. 2005; 102:4134–4139.
45. Tassaneeyakul W, Jantararoungtong T, Chen P, Lin PY, Tiamkao S, Khunarkornsiri U, Chucherd P, Konyoung P, Vannaprasaht S, Choonhakarn C, Pisuttimarn P, Sangviroon A, Tassaneeyakul W. Strong association between HLA-B*5801 and allopurinol-induced Stevens-Johnson syndrome and toxic epidermal necrolysis in a Thai population. Pharmacogenet Genomics. 2009; 19:704–709.
46. Kaniwa N, Saito Y, Aihara M, Matsunaga K, Tohkin M, Kurose K, Sawada J, Furuya H, Takahashi Y, Muramatsu M, Kinoshita S, Abe M, Ikeda H, Kashiwagi M, Song Y, Ueta M, Sotozono C, Ikezawa Z, Hasegawa R. JSAR research group. HLA-B locus in Japanese patients with antiepileptics and allopurinol-related Stevens-Johnson syndrome and toxic epidermal necrolysis. Pharmacogenomics. 2008; 9:1617–1622.

47. Kang HR, Jee YK, Kim YS, Lee CH, Jung JW, Kim SH, Park HW, Chang YS, Jang IJ, Cho SH, Min KU, Kim SH, Lee KW. Adverse Drug Reaction Research Group in Korea. Positive and negative associations of HLA class I alleles with allopurinol-induced SCARs in Koreans. Pharmacogenet Genomics. 2011; 21:303–307.

48. Lonjou C, Borot N, Sekula P, Ledger N, Thomas L, Halevy S, Naldi L, Bouwes-Bavinck JN, Sidoroff A, de Toma C, Schumacher M, Roujeau JC, Hovnanian A, Mockenhaupt M. RegiSCAR study group. A European study of HLA-B in Stevens-Johnson syndrome and toxic epidermal necrolysis related to five high-risk drugs. Pharmacogenet Genomics. 2008; 18:99–107.

49. Chung WH, Hung SI, Hong HS, Hsih MS, Yang LC, Ho HC, Wu JY, Chen YT. Medical genetics: a marker for Stevens-Johnson syndrome. Nature. 2004; 428:486.
50. Man CB, Kwan P, Baum L, Yu E, Lau KM, Cheng AS, Ng MH. Association between HLA-B*1502 allele and antiepileptic drug-induced cutaneous reactions in Han Chinese. Epilepsia. 2007; 48:1015–1018.
51. Locharernkul C, Loplumlert J, Limotai C, Korkij W, Desudchit T, Tongkobpetch S, Kangwanshiratada O, Hirankarn N, Suphapeetiporn K, Shotelersuk V. Carbamazepine and phenytoin induced Stevens-Johnson syndrome is associated with HLA-B*1502 allele in Thai population. Epilepsia. 2008; 49:2087–2091.
52. Mehta TY, Prajapati LM, Mittal B, Joshi CG, Sheth JJ, Patel DB, Dave DM, Goyal RK. Association of HLA-B*1502 allele and carbamazepineinduced Stevens-Johnson syndrome among Indians. Indian J Dermatol Venereol Leprol. 2009; 75:579–582.
53. Tangamornsuksan W, Chaiyakunapruk N, Somkrua R, Lohitnavy M, Tassaneeyakul W. Relationship between the HLA-B*1502 allele and carbamazepine-induced Stevens-Johnson syndrome and toxic epidermal necrolysis: a systematic review and meta-analysis. JAMA Dermatol. 2013; 149:1025–1032.
54. Kaniwa N, Saito Y, Aihara M, Matsunaga K, Tohkin M, Kurose K, Furuya H, Takahashi Y, Muramatsu M, Kinoshita S, Abe M, Ikeda H, Kashiwagi M, Song Y, Ueta M, Sotozono C, Ikezawa Z, Hasegawa R. JSAR research group. HLA-B*1511 is a risk factor for carbamazepine-induced Stevens-Johnson syndrome and toxic epidermal necrolysis in Japanese patients. Epilepsia. 2010; 51:2461–2465.
55. Kim SH, Lee KW, Song WJ, Kim SH, Jee YK, Lee SM, Kang HR, Park HW, Cho SH, Park SH, Min KU, Chang YS. Adverse Drug Reaction Research Group in Korea. Carbamazepine-induced severe cutaneous adverse reactions and HLA genotypes in Koreans. Epilepsy Res. 2011; 97:190–197.

56. Shi YW, Min FL, Qin B, Zou X, Liu XR, Gao MM, Wang Q, Zhou JQ, Liao WP. Association between HLA and Stevens-Johnson syndrome induced by carbamazepine in Southern Han Chinese: genetic markers besides B*1502? Basic Clin Pharmacol Toxicol. 2012; 111:58–64.
57. Hsiao YH, Hui RC, Wu T, Chang WC, Hsih MS, Yang CH, Ho HC, Chang YG, Chen MJ, Lin JY, Chen DP, Chang PY, Wu TL, Hung SI, Chung WH. Genotype-phenotype association between HLA and carbamazepineinduced hypersensitivity reactions: strength and clinical correlations. J Dermatol Sci. 2014; 73:101–109.

58. Ikeda H, Takahashi Y, Yamazaki E, Fujiwara T, Kaniwa N, Saito Y, Aihara M, Kashiwagi M, Muramatsu M. HLA class I markers in Japanese patients with carbamazepine-induced cutaneous adverse reactions. Epilepsia. 2010; 51:297–300.

59. Ozeki T, Mushiroda T, Yowang A, Takahashi A, Kubo M, Shirakata Y, Ikezawa Z, Iijima M, Shiohara T, Hashimoto K, Kamatani N, Nakamura Y. Genome-wide association study identifies HLA-A*3101 allele as a genetic risk factor for carbamazepine-induced cutaneous adverse drug reactions in Japanese population. Hum Mol Genet. 2011; 20:1034–1041.
60. Niihara H, Kakamu T, Fujita Y, Kaneko S, Morita E. HLA-A31 strongly associates with carbamazepine-induced adverse drug reactions but not with carbamazepine-induced lymphocyte proliferation in a Japanese population. J Dermatol. 2012; 39:594–601.

61. Hung SI, Chung WH, Jee SH, Chen WC, Chang YT, Lee WR, Hu SL, Wu MT, Chen GS, Wong TW, Hsiao PF, Chen WH, Shih HY, Fang WH, Wei CY, Lou YH, Huang YL, Lin JJ, Chen YT. Genetic susceptibility to carbamazepine-induced cutaneous adverse drug reactions. Pharmacogenet Genomics. 2006; 16:297–306.

62. Genin E, Chen DP, Hung SI, Sekula P, Schumacher M, Chang PY, Tsai SH, Wu TL, Bellon T, Tamouza R, Fortier C, Toubert A, Charron D, Hovnanian A, Wolkenstein P, Chung WH, Mockenhaupt M, Roujeau JC. HLA-A*31:01 and different types of carbamazepine-induced severe cutaneous adverse reactions: an international study and metaanalysis. Pharmacogenomics J. 2014; 14:281–288.
63. McCormack M, Alfirevic A, Bourgeois S, Farrell JJ, Kasperaviciutė D, Carrington M, Sills GJ, Marson T, Jia X, de Bakker PI, Chinthapalli K, Molokhia M, Johnson MR, O'Connor GD, Chaila E, Alhusaini S, Shianna KV, Radtke RA, Heinzen EL, Walley N, Pandolfo M, Pichler W, Park BK, Depondt C, Sisodiya SM, Goldstein DB, Deloukas P, Delanty N, Cavalleri GL, Pirmohamed M. HLA-A*3101 and carbamazepine-induced hypersensitivity reactions in Europeans. N Engl J Med. 2011; 364:1134–1143.
64. Zhang FR, Liu H, Irwanto A, Fu XA, Li Y, Yu GQ, Yu YX, Chen MF, Low HQ, Li JH, Bao FF, Foo JN, Bei JX, Jia XM, Liu J, Liany H, Wang N, Niu GY, Wang ZZ, Shi BQ, Tian HQ, Liu HX, Ma SS, Zhou Y, You JB, Yang Q, Wang C, Chu TS, Liu DC, Yu XL, Sun YH, Ning Y, Wei ZH, Chen SL, Chen XC, Zhang ZX, Liu YX, Pulit SL, Wu WB, Zheng ZY, Yang RD, Long H, Liu ZS, Wang JQ, Li M, Zhang LH, Wang H, Wang LM, Xiao P, Li JL, Huang ZM, Huang JX, Li Z, Liu J, Xiong L, Yang J, Wang XD, Yu DB, Lu XM, Zhou GZ, Yan LB, Shen JP, Zhang GC, Zeng YX, de Bakker PI, Chen SM, Liu JJ. HLA-B*13:01 and the dapsone hypersensitivity syndrome. N Engl J Med. 2013; 369:1620–1628.
65. Cheung YK, Cheng SH, Chan EJ, Lo SV, Ng MH, Kwan P. HLA-B alleles associated with severe cutaneous reactions to antiepileptic drugs in Han Chinese. Epilepsia. 2013; 54:1307–1314.

66. Hung SI, Chung WH, Liu ZS, Chen CH, Hsih MS, Hui RC, Chu CY, Chen YT. Common risk allele in aromatic antiepileptic-drug induced Stevens-Johnson syndrome and toxic epidermal necrolysis in Han Chinese. Pharmacogenomics. 2010; 11:349–356.

67. Kim SH, Kim M, Lee KW, Kim SH, Kang HR, Park HW, Jee YK. HLAB* 5901 is strongly associated with methazolamide-induced Stevens-Johnson syndrome/toxic epidermal necrolysis. Pharmacogenomics. 2010; 11:879–884.
68. Martin AM, Nolan D, James I, Cameron P, Keller J, Moore C, Phillips E, Christiansen FT, Mallal S. Predisposition to nevirapine hypersensitivity associated with HLA-DRB1*0101 and abrogated by low CD4 T-cell counts. AIDS. 2005; 19:97–99.
69. Vitezica ZG, Milpied B, Lonjou C, Borot N, Ledger TN, Lefebvre A, Hovnanian A. HLA-DRB1*01 associated with cutaneous hypersensitivity induced by nevirapine and efavirenz. AIDS. 2008; 22:540–541.
70. Littera R, Carcassi C, Masala A, Piano P, Serra P, Ortu F, Corso N, Casula B, La Nasa G, Contu L, Manconi PE. HLA-dependent hypersensitivity to nevirapine in Sardinian HIV patients. AIDS. 2006; 20:1621–1626.

71. Chantarangsu S, Mushiroda T, Mahasirimongkol S, Kiertiburanakul S, Sungkanuparph S, Manosuthi W, Tantisiriwat W, Charoenyingwattana A, Sura T, Chantratita W, Nakamura Y. HLA-B*3505 allele is a strong predictor for nevirapine-induced skin adverse drug reactions in HIVinfected Thai patients. Pharmacogenet Genomics. 2009; 19:139–146.
72. Gatanaga H, Yazaki H, Tanuma J, Honda M, Genka I, Teruya K, Tachikawa N, Kikuchi Y, Oka S. HLA-Cw8 primarily associated with hypersensitivity to nevirapine. AIDS. 2007; 21:264–265.

73. Song JS, Kang ES, Joo EY, Hong SB, Seo DW, Lee SY. Absence of HLAB* 1502 and HLA-A*3101 alleles in 9 Korean patients with antiepileptic drug-induced skin rash: a preliminary study. Ann Lab Med. 2014; 34:372–375.
74. Ferrell PB Jr, McLeod HL. Carbamazepine, HLA-B*1502 and risk of Stevens-Johnson syndrome and toxic epidermal necrolysis: US FDA recommendations. Pharmacogenomics. 2008; 9:1543–1546.
75. Chung WH, Hung SI, Chen YT. Genetic predisposition of life-threatening antiepileptic-induced skin reactions. Expert Opin Drug Saf. 2010; 9:15–21.

76. Saito N, Qiao H, Yanagi T, Shinkuma S, Nishimura K, Suto A, Fujita Y, Suzuki S, Nomura T, Nakamura H, Nagao K, Obuse C, Shimizu H, Abe R. An annexin A1-FPR1 interaction contributes to necroptosis of keratinocytes in severe cutaneous adverse drug reactions. Sci Transl Med. 2014; 6:245ra95.

77. Chung WH, Chang WC, Lee YS, Wu YY, Yang CH, Ho HC, Chen MJ, Lin JY, Hui RC, Ho JC, Wu WM, Chen TJ, Wu T, Wu YR, Hsih MS, Tu PH, Chang CN, Hsu CN, Wu TL, Choon SE, Hsu CK, Chen DY, Liu CS, Lin CY, Kaniwa N, Saito Y, Takahashi Y, Nakamura R, Azukizawa H, Shi Y, Wang TH, Chuang SS, Tsai SF, Chang CJ, Chang YS, Hung SI. Taiwan Severe Cutaneous Adverse Reaction Consortium. Japan Pharmacogenomics Data Science Consortium. Genetic variants associated with phenytoin-related severe cutaneous adverse reactions. JAMA. 2014; 312:525–534.

78. Kesavan R, Narayan SK, Adithan C. Influence of CYP2C9 and CYP2C19 genetic polymorphisms on phenytoin-induced neurological toxicity in Indian epileptic patients. Eur J Clin Pharmacol. 2010; 66:689–696.

79. Depondt C, Godard P, Espel RS, Da Cruz AL, Lienard P, Pandolfo M. A candidate gene study of antiepileptic drug tolerability and efficacy identifies an association of CYP2C9 variants with phenytoin toxicity. Eur J Neurol. 2011; 18:1159–1164.
80. Lee AY, Kim MJ, Chey WY, Choi J, Kim BG. Genetic polymorphism of cytochrome P450 2C9 in diphenylhydantoin-induced cutaneous adverse drug reactions. Eur J Clin Pharmacol. 2004; 60:155–159.
81. Ciccacci C, Di Fusco D, Marazzi MC, Zimba I, Erba F, Novelli G, Palombi L, Borgiani P, Liotta G. Association between CYP2B6 polymorphisms and Nevirapine-induced SJS/TEN: a pharmacogenetics study. Eur J Clin Pharmacol. 2013; 69:1909–1916.

83. Nuki G. An appraisal of the 2012 American College of Rheumatology guidelines for the management of gout. Curr Opin Rheumatol. 2014; 26:152–161.

84. Chung WH, Chang WC, Stocker SL, Juo CG, Graham GG, Lee MH, Williams KM, Tian YC, Juan KC, Jan Wu YJ, Yang CH, Chang CJ, Lin YJ, Day RO, Hung SI. Insights into the poor prognosis of allopurinol-induced severe cutaneous adverse reactions: the impact of renal insufficiency, high plasma levels of oxypurinol and granulysin. Ann Rheum Dis. 2014; 08. 12. [Epub] http://dx.doi.org/10.1136/annrheumdis-2014-205577.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download