Abstract
Allergic Aspergillus sinusitis (AAS) is a three decade old clinicopathologic entity in which mucoid impaction akin to that of allergic bronchopulmonary aspergillosis (ABPA) occurs in the paranasal sinuses. Features such as radiographic evidence of pansinusitis, passage of nasal plugs and recurrent nasal polyposis in patients with an atopic background is suggestive of AAS. Histopathlogic confirmation from the inspissated mucus is a sine qua non for the diagnosis. Heterogeneous densities on computed tomography of the paranasal sinuses are caused by the 'allergic mucin' in the sinuses. Many patients give a history of having undergone multiple surgical procedures for symptomatic relief. The current approach to treatment appears to include an initial surgical debridement followed by postoperative oral corticosteroids for long durations. Although both ABPA and AAS are classified as Aspergillus-related hypersensitivity respiratory disorders, their co-occurrence appears to be an infrequently recognised phenomenon. This could perhaps be attributed to the fact that these two diseases are often treated by two different specialties. A high index of suspicion is required to establish the diagnoses of ABPA and AAS. All patients with asthma and/or rhinosinusitis along with sensitisation to Aspergillus antigens are at an increased risk of developing ABPA and/or AAS. ABPA must be excluded in all patients with AAS and vice versa. Early diagnosis and initiation of appropriate therapy could plausibly alter the course of the disease processes and prevent the possible development of long term sequelae.
Aspergillus, a genus of spore-forming ubiquitous fungi, affects both the upper and the lower respiratory tracts [1, 2] and has protean manifestations (Table 1). The clinical spectrum of hypersensitivity manifestations in the respiratory tract includes Aspergillus induced asthma, allergic bronchopulmonary aspergillosis (ABPA) and allergic Aspergillus sinusitis (AAS). Allergic bronchopulmonary aspergillosis, predominantly a disease of the asthmatics, is well established worldwide for nearly six decades with reports from around the globe [3], while AAS is comparatively a more recently identified entity that is now known for the last 30 years. We present a brief review on AAS and its association with ABPA.
Hinson and colleagues [4] were the first to recognise ABPA, a potentially destructive lung disease, in England in 1952. Almost three decades later, in 1981, Millar and colleagues [5], also from England, presented five patients with chronic sinusitis, all of whom had pathologic resemblance to the mucoid impaction of ABPA. They stated this as the first description of allergic aspergillosis of the paranasal sinuses. In 1983, Katzenstein and coworkers [6] retrospectively reviewed 119 surgically obtained specimens from the paranasal sinuses and found that seven patients had pathologic findings characteristic of the mucoid impaction of ABPA. These investigators coined the term 'allergic mucin' to describe the nasal material comprising cellular debris, eosinophils, Charcot-Leyden crystals and scattered fungal hyphae. The authors designated this newly recognised clinicopathologic disease as 'allergic Aspergillus sinusitis'.
Once resemblance to ABPA was observed, a set of criteria was enunciated for the diagnosis of AAS [7, 8]. While radiologic and histopathologic features are key components, demonstration of fungal elements/culture obtained from the sinus material is the mainstay for the diagnosis (Table 2). Diabetics, immunocompromised subjects and patients with invasive fungal disease are excluded from the diagnosis. Other features, akin to those in ABPA, support the diagnosis of AAS. These include peripheral blood eosinophilia, cutaneous hypersensitivity to Aspergillus antigens, elevated total IgE levels, Aspergillus-specific IgE and IgG antibodies, and precipitating antibodies to Aspergillus antigens [9]. We have elicited type-I hypersensitivity by skin testing in all our patients with AAS [8]. This was complemented by elevated total IgE levels in all patients and the presence of Aspergillus-specific IgE antibodies in most of the patients.
Fungi other than Aspergillus may also cause allergic sinusitis and hence a comprehensive term allergic fungal sinusitis (AFS) was introduced [10]. A retrospective case review [11] of 67 consecutive patients with AFS from the United States, all of whom met the histopathologic criteria for the diagnosis, showed that the mould Bipolaris spicifera was cultured at sinus surgery in more than two-thirds of the patients. This was followed by Aspergillus, which was seen in 9%. Exserohilum, Alternaria and Curvularia were grown in 3% each while Torula was isolated in 2% of the patients. The sinus material was sterile in 13%.
With the availability of easier diagnostic surgeries and improved fungal culture techniques, AFS, including AAS, has now been widely recognised. In the Asia-Pacific region, cases have been described from Australia [10], India [8, 12-14], Thailand [15], Malaysia [16] and Saudi Arabia [17]. We reported the first patient of AAS from India [12], and also described the first instance of concomitant occurrence of ABPA and AAS from Asia [18].
This allergic sinus disorder of fungal origin is characterised by mucoid impaction in the paranasal sinuses, which is akin to that of ABPA. This too occurs in atopic individuals and, as in ABPA, it is likely that release of antigenic material from the fungi sets into motion a chain of immunologic reactions culminating in the development of AAS. We have earlier shown that a fourth of the patients with perennial rhinitis had positive skin reactivity to Aspergillus antigens [19]. Recently this was further confirmed by us in 131 patients with allergic rhinitis where type-I hypersensitivity to Aspergillus was demonstrated in 24.4% of the patients [20]. Two of these 131 subjects were diagnosed as AAS. Since rhinitis is an important predisposing factor for sinusitis, it appears that these patients may be at a greater risk for developing AAS. It has been suggested that 5 to 10% of patients with chronic rhinosinusitis go on to develop AFS [13, 21-23]. Human leukocyte antigen gene association, particularly with major histocompatibility class II regions, were observed in two-thirds of 44 patients with AFS from the United States [24]. This association has also been found in ABPA, a disease with similar immunopathologic mechanisms [25].
In the series of 119 patients studied by Katzenstein et al. [6], there were another two patients who had similar histopathologic findings of the characteristic eosinophilic mucin plugs as seen in AAS but Aspergillus could not be cultured from the sinus material. When more reports of patients with allergic mucin without fungal elements were published [26-29], these were termed as having 'eosinophilic mucin rhinosinusitis' (EMRS) rather than AFS. It was retrospectively observed that patients with EMRS, when compared to those with AFS, had a higher incidence of aspirin sensitive asthma and were older in age at presentation [29]. In addition, a significantly greater number were asthmatics.
In order to differentiate such patients, Pant et al. [30] studied the demographic, clinical and immunologic characteristics of patients who were divided into four different subgroups based on the presence/absence of fungi in the eosinophilic mucin and the presence/absence of type-I hypersensitivity to fungi. Although the patients did not differ significantly among each other, those with IgE-mediated fungal allergy were almost a decade younger than the nonallergic patients. However, as a single group, these patients had more severe sinus disease when compared to those with chronic rhinosinusitis without eosinophilic mucin.
Patients with AFS belong to a fairly younger age group, which, given an atopic background, is not surprising. In the retrospective case review [11] of 67 patients, the mean age was 33.3 years with a female preponderance (58%). The presentation is generally insidious with a history of rhinorrhoea, sneezing, nasal blockage, and passage of nasal plugs. History of asthma may also be elicited, and some of these may also complain of passage of golden brown sputum plugs. On physical examination, nasal polyps and crusts can be found which cause gradual obstruction of the ostia leading to accumulation of allergic mucin in the closed spaces of the sinuses. The expanding mass may gradually cause distortion of facial features, which may not be easily noticed. It can erode into the sinus walls to extend into the adjacent spaces such as the orbit, causing proptosis, and sometimes even loss of vision [31, 32]. A history of previous surgeries for recurrent sinusitis is also evident. Superimposed acute bacterial infection may lead to pain [33].
The demonstration of sinusitis on plain sinus roentgenograms in one or more paranasal sinuses is one of the diagnostic criteria for AAS [7, 8, 12]. Haziness or clouding of the sinus is the most common feature. More often than not, more than one sinus is opacified. This is in contrast to a fungal ball where almost always a single sinus is affected. Bilateral involvement was observed in up to 83% of cases reported [8, 34]. Furthermore, Waxman and colleagues [34] state, "when sequential time related studies were performed, progressive involvement of multiple sinuses was the rule."
Other manifestations include mucosal thickening and varying degrees of bony sclerosis and/or destruction. However, these findings are non-specific as they may also be seen in chronic and/or infective sinusitis or malignancies of the paranasal sinuses. Currently, computed tomography (CT) has emerged as the imaging modality of choice.
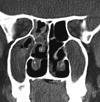
A more reliable and accurate radiological diagnosis can be made with the help of CT of the paranasal sinuses (CT-PNS). The characteristic feature is the occurrence of heterogeneous densities, signifying opacification of the sinuses; with serpiginous areas of increased attenuation on non-contrast scans [35, 36]. These hyperdense areas (Fig. 1) are due to the presence of 'allergic mucin'. The serpiginous pattern of the central area of high attenuation has also been described as 'star-filled sky' or 'ground glass' patterns [37]. Presence of ferromagnetic elements (iron and manganese) that are produced by the fungi is said to be responsible for the heterogeneous densities [35].
The extent of disease is better visualised on CT-PNS. In a study of 44 patients with AFS [26], CT scans showed involvement of three or more sinuses in all patients. Both unilateral [35, 38] and bilateral [8, 34, 39] predominance of the disease was noticed in different series. Mukherji et al. [36] retrospectively reviewed CT scans, along with surgical and histologic reports, in 45 patients with AFS and found an almost equal preponderence of unilateral (49%) and bilateral involvement (51%). All 45 patients had increased intrasinus attenuation with mucosal thickening on non-contrast scans. The ethmoid sinus complex was most frequently involved (96%), followed by the maxillary (93%), frontal (71%) and sphenoid (67%) sinuses. While complete opacification of at least one sinus was seen in all but one (98%) patient, multiple sinus involvement was noted in 96%. The CT-PNS in most of these patients demonstrated expansion of the opacified sinus (98%), remodeling of the bony sinus walls (95%) and erosion of a sinus wall (93%). A fifth of the patients also had evidence of involvement of adjacent soft tissue structures.
The exact mechanism of the limited bony destruction, which has been noted in up to 80% of patients with AFS, is not clear [27, 35, 40]. This is either due to pressure necrosis causing reabsorption of the bone or bone destroying enzymes produced by the fungus. Inflammatory mediators such as major basic protein produced by the allergic response could also be responsible [41, 42]. This is in contrast to invasive fungal aspergillosis wherein bone destruction is frankly evident [2].
Prior to definitive surgery, it is important to determine whether there is any intracranial involvement or not [43-45]. In a retrospective review of 25 patients with AFS [44], extensive erosion of the skull base with intracranial extension of the disease was seen in four patients. However, histologic evidence of tissue invasion was absent in all 25 patients. Leakage of sinus contents into the orbit may also occur, and this is more clearly delineated on CT scanning [31, 45, 46]. A case of pansinusitis with orbital involvement associated with an allergic response to the fungus Curvularia lunata has been described [31]. An analysis of 21 patients with AFS revealed intracranial extension in eight and erosion of the lamina papyracea in six, with four of them having both intracranial and intraorbital involvement [45]. Orbital involvement may perhaps be more common in children due to the presence of incompletely opacified cartilaginous sinus walls [47].
Computed tomography also helps in distinguishing AFS from other forms of fungal sinusitis, which include acute (fulminant) invasive fungal sinusitis, chronic and/or indolent fungal sinusitis and fungal balls in the sinuses [2, 48, 49]. Aggressive bone erosion with extension of disease into the adjacent soft tissues, viz. dura mater and orbit, is a feature of acute invasive fungal sinusitis. Unlike in the allergic form of the disease, sinus expansion and sinus wall modeling is uncommon in acute invasive fungal sinusitis. Sometimes, a patient may present during the early stages of invasive fungal sinusitis well before bony erosion and tissue necrosis are evident on CT, which cannot be differentiated from noninvasive sinusitis by CT alone [50].
Chronic disease of the sinuses with desiccated secretions within mucocoeles and polyps often result in thickening and sclerosis of the adjacent sinus walls, while the presence of expansion and thinning of the sinus walls is suggestive of AFS [36]. Fungal balls appear as focal round areas of increased attenuation that are usually centered within a diseased maxillary sinus. This may sometimes be difficult to differentiate from AFS but the increased attenuation of the saprophytic colonisation in the fungal ball is more homogeneous than that of the allergic disease process.
Although not the primary diagnostic procedure, magnetic resonance imaging (MRI) is more sensitive than CT in differentiating various forms of invasive and noninvasive fungal diseases of the paranasal sinuses [51]. On MRI, AFS is characterised by areas of decreased T1 weighted signal intensities [52]. These correspond to the hyperattenuated lesions seen on CT. On T1 weighted images, high signal intensity, representing inflammation, is also seen along the periphery of the affected sinuses.
A central area of signal void and a narrow peripheral rim of increased intensity, due to the inflamed mucosa of the sinuses involved, is visualised on T2 weighted images. The hypodense regions are due to the presence of ferromagnetic elements within the fungal concretions [42, 51, 52]. It has also been postulated that the low intensity on T1 and T2 weighted images could be due to the high protein and low water concentration of the 'allergic mucin', while the high water content within the surrounding oedematous mucosa may be responsible for increased attenuation [45].
MRI also provides information about areas of critical importance, which include the cavernous sinuses and the brain. Furthermore, AFS can also be differentiated from other mycotic diseases of the paranasal sinuses. Although fungal balls of the sinuses cast a similar appearance of low intensity on T1 weighted images and signal voids on T2 weighted images, peripheral hyperintense areas are absent. Malignancies appear as moderately hyperintense areas while bacterial infections are even more hyperintense [52].
The clinical categories of Aspergillus-related respiratory disorders seem to remain mutually exclusive. Tenacious secretions within the bronchi of some patients, usually asthmatics, provide a hospitable environment for the growth of fungi. The subsequent release of antigenic fungal material sets into motion a host of immunological reactions culminating in pulmonary damage [53]. Usually the disease process manifests as a type I (IgE mediated) hypersensitivity response, but type III (IgG mediated immune complexes) and type IV (cell mediated) reactions have also been noted. Tissue invasion, however, does not occur [54]. A similar syndrome can also be caused by fungi other than Aspergillus, which is collectively classified as allergic bronchopulmonary mycosis (ABPM) [55].
In spite of similar histopathological features between the allergic mucin of AAS and the mucous plugs of ABPA, concomitant occurrence of ABPA and AAS has not often been reported. However, our analysis of 95 patients with ABPA suggests that AAS may not be all that uncommon in patients with ABPA [8]. In 1976, Safirstein [56] reported a patient with ABPA who gave a history of passage of hard nasal plugs and had evidence of upper airways obstruction. This, in retrospect, could quite likely be a case of concomitant ABPA and AAS. Till date, 22 such patients have been published [8, 18, 57-66], 11 of whom were documented by us [8, 18, 59, 60, 62, 66]. Four of these subjects had associated familial occurrence [18, 66]. These included some unique presentations such as associated familial occurrence of ABPA [18], middle lobe syndrome [59], collapsed lung with pleural effusion and prior operated aspergilloma [60] and a case where coexisting aspergilloma was also detected [62].
In our review of 95 patients with ABPA, 22 had radiological evidence of sinusitis [8]. Nine of these patients consented to undergo the surgical procedures required for procuring sinus material for histopathologic examination. Seven patients were confirmed as having concomitant AAS. Nasal symptoms preceded chest symptoms in two patients, vice versa in one and occurred simultaneously in four. In the remaining 13 patients the possibility of AAS could not be ruled out as they refused to undergo the invasive procedures needed to establish the diagnosis. Since then, the availability of minimally invasive methods has helped in establishing the diagnosis more easily.
To highlight the coexistence of ABPM and AFS, Venarske and deShazo [61], in 2002, coined the term 'sinobronchial allergic mycosis' (the SAM syndrome) to describe the expression of fungal hypersensitivity in both upper and lower airways. This was on the basis of five patients, three of whom were earlier reported by us for their rare presentations [18, 59, 60]. The authors further state in the addendum that four other cases of concomitant ABPA and AAS reported by us [8] also fulfill the criteria for SAM syndrome enunciated by them. Very recently, we retrospectively reviewed the records of 164 patients with ABPA diagnosed over a period of 22 years, and found familial occurrence in four pairs. Concomitant AAS was present in one patient each in three of these four pairs [66].
The co-occurrence of both these diseases is yet to receive the recognition it deserves. In the Mayo Clinic series of 44 patients with AFS [27], there was no mention of concomitant ABPA. In another series of 67 consecutive cases of AFS predominantly caused by Bipolaris spicifera, concomitant AFS and ABPM were seen in only two patients [11]. Both these patients had been previously diagnosed with ABPM after undergoing partial lobectomy. A study on 126 patients of ABPA [67] from India does not mention any patient with nasal symptoms, or any attempt to diagnose concomitant AAS. Recently, a study on 20 children with fungal sinusitis [68] identified 11 of them with asthma, but these were not investigated further for the presence of ABPA.
A standardised protocol for the treatment of AFS has not yet evolved. Till date, no prospective randomised controlled studies are available. A combined approach with surgical debridement along with oral corticosteroids would most probably be required to manage the ongoing allergic inflammation [8, 69]. Surgery alone has seldom proved to be curative, and more often than not patients have had to undergo repeat procedures as the recurrence rate is high. However, during surgery, patients benefit from improved drainage and aeration to the sinus mucosa. Removal of the inspissated allergic mucin and resection of the hypertrophic sinus mucosa also helps in reducing fungal load.
Based on the immuno-pathological anology of AAS with ABPA, treatment recommendations for the former have been formulated on existing guidelines for the latter. Oral prednisolone, given for long durations, helps not only in improving the symptomatology but also in prolonging recurrence after sinus surgery [70]. Addition of oral corticosteroids postoperatively currently remains the best bet for patients with AFS. We have assessed the feasibility of a bi-weekly regimen of oral prednisolone in 26 patients with ABPA, two of whom had concurrent AAS as well. We found that this regimen was as effective as the conventional alternate day oral prednisolone therapy in the management of patients with ABPA and AAS [71].
The optimal duration of treatment is yet to be determined. Topical corticosteroids alone, the mainstay of therapy for allergic rhinosinusitis, are not sufficient to bring symptomatic relief. While oral antifungals do not seem to have a clear cut benefit, the role of topical antifungal agents is still unknown. It was presumed that by decreasing the fungal load using antifungal drugs, antigenic stimulation would be reduced thereby decreasing the inflammatory response. A randomised placebo controlled trial with 32 weeks of oral itraconazole in patients with severe asthma and fungal sensitisation demonstrated significant improvements in quality of life and modest improvements in some of the clinical endpoints of asthma and rhinitis [72]. The Cochrane Database review [73] on azoles for ABPA inferred that itraconazole, in addition to modifying the immunologic activation associated with ABPA, improved clinical outcome. However, such studies have yet to be replicated in patients with AFS/AAS. The role of allergen immunotherapy in AFS too has been investigated [74].
Allergic Aspergillus sinusitis, a chronic indolent disease, is characterised by the presence of 'allergic mucin' in the sinonasal material obtained for histopathology. This must be sought for in all patients with rhinosinusitis with a positive skin allergy test to Aspergillus or other fungal antigens. The association of ABPA and AAS is not as uncommon as it appears. These entities share similar immunopathologic mechanisms, and the mainstay of treatment for both these diseases is oral prednisolone. Since asthma and sinusitis are two diseases often treated by two different specialties, the occurrence of AAS in ABPA and of ABPA in AAS may easily be overlooked [8]. Emphasis on early recognition of chest symptoms in patients with AAS and nasal symptoms in patients with ABPA could increase the diagnosis of contemporaneous involvement, and possibly reduce morbidity.
Figures and Tables
References
1. Shah A. Allergic bronchopulmonary aspergillosis. Indian J Chest Dis Allied Sci. 1998. 40:41–54.
3. Shah A, Panjabi C. Allergic bronchopulmonary aspergillosis: a review of a disease with a worldwide distribution. J Asthma. 2002. 39:273–289.

4. Hinson KF, Moon AJ, Plummer NS. Broncho-pulmonary aspergillosis: a review and a report of eight new cases. Thorax. 1952. 7:317–333.
5. Millar JW, Johnston A, Lamb D. Allergic aspergillosis of the maxillary sinuses [Abstract]. Thorax. 1981. 36:710.
6. Katzenstein AL, Sale SR, Greenberger PA. Allergic Aspergillus sinusitis: a newly recognized form of sinusitis. J Allergy Clin Immunol. 1983. 72:89–93.
7. deShazo RD, Swain RE. Diagnostic criteria for allergic fungal sinusitis. J Allergy Clin Immunol. 1995. 96:24–35.

8. Shah A, Panchal N, Agarwal AK. Concomitant allergic bronchopulmonary aspergillosis and allergic Aspergillus sinusitis: a review of an uncommon association. Clin Exp Allergy. 2001. 31:1896–1905.
9. Shah A. Allergic bronchopulmonary and sinus aspergillosis: the co-occurrence. Chest (India). 2001. 2:234–235.
10. Robson JM, Hogan PG, Benn RA, Gatenby PA. Allergic fungal sinusitis presenting as a paranasal sinus tumour. Aust N Z J Med. 1989. 19:351–353.

11. Schubert MS, Goetz DW. Evaluation and treatment of allergic fungal sinusitis. I. Demographics and diagnosis. J Allergy Clin Immunol. 1998. 102:387–394.

12. Shah A, Khan ZU, Sircar M, Chaturvedi S, Malik GB, Randhawa HS. Allergic Aspergillus sinusitis: an Indian report. Respir Med. 1990. 84:249–251.
13. Saravanan K, Panda NK, Chakrabarti A, Das A, Bapuraj RJ. Allergic fungal rhinosinusitis: an attempt to resolve the diagnostic dilemma. Arch Otolaryngol Head Neck Surg. 2006. 132:173–178.
14. Michael RC, Michael JS, Ashbee RH, Mathews MS. Mycological profile of fungal sinusitis: An audit of specimens over a 7-year period in a tertiary care hospital in Tamil Nadu. Indian J Pathol Microbiol. 2008. 51:493–496.

15. Aeumjaturapat S, Saengpanich S, Isipradit P, Keelawat S. Eosinophilic mucin rhinosinusitis: terminology and clinicopathological presentation. J Med Assoc Thai. 2003. 86:420–424.
16. Goh BS, Gendeh BS, Rose IM, Pit S, Samad SA. Prevalence of allergic fungal sinusitis in refractory chronic rhinosinusitis in adult Malaysians. Otolaryngol Head Neck Surg. 2005. 133:27–31.

17. Al-Swiahb JN, Al-Ammar A, Al-Dousary SH. Allergic fungal sinusitis in children in Saudi Arabia. Saudi Med J. 2007. 28:1711–1714.
18. Shah A, Khan ZU, Chaturvedi S, Malik GB, Randhawa HS. Concomitant allergic Aspergillus sinusitis and allergic bronchopulmonary aspergillosis associated with familial occurrence of allergic bronchopulmonary aspergillosis. Ann Allergy. 1990. 64:507–512.
19. Shah A, Sircar M. Sensitization to Aspergillus antigens in perennial rhinitis. Asian Pac J Allergy Immunol. 1991. 9:137–139.
20. Sahay S, Shah A. Allergic rhinitis: Aspergillus sensitisation increases the severity of sinusitis in "blockers" as compared to "sneezers and runners" [Abstract]. Allergy. 2008. 63:Suppl 88. S73.
21. Ence BK, Gourley DS, Jorgensen NL. Allergic fungsal sinusitis. Am J Rhinol. 1990. 4:169–178.
22. Collins MM, Nair SB, Wormald PJ. Prevalence of noninvasive fungal sinusitis in South Australia. Am J Rhinol. 2003. 17:127–132.

23. Thrasher RD, Kingdom TT. Fungal infections of the head and neck: an update. Otolaryngol Clin North Am. 2003. 36:577–594.

24. Schubert MS, Hutcheson PS, Graff RJ, Santiago L, Slavin RG. HLA-DQB1*03 in allergic fungal sinusitis and other chronic hypertrophic rhinosinusitis disorders. J Allergy Clin Immunol. 2004. 114:1376–1383.

25. Chauhan B, Santiago L, Hutcheson PS, Schwartz HJ, Spitznagel E, Castro M, Slavin RG, Bellone CJ. Evidence for the involvement of two different MHC class II regions in susceptibility or protection in allergic bronchopulmonary aspergillosis. J Allergy Clin Immunol. 2000. 106:723–729.

26. Allphin AL, Strauss M, Abdul-Karim FW. Allergic fungal sinusitis: problems in diagnosis and treatment. Laryngoscope. 1991. 101:815–820.
27. Cody DT, Neel HB, Ferreiro JA, Roberts GD. Allergic fungal sinusitis: the Mayo Clinic experience. Laryngoscope. 1994. 104:1074–1079.
28. Ramadan HH, Quraishi HA. Allergic mucin sinusitis without fungus. Am J Rhinol. 1997. 11:145–147.

29. Ferguson BJ. Eosinophilic mucin rhinosinusitis: a distinct clinicopathological entity. Laryngoscope. 2000. 110:799–813.

30. Pant H, Kette FE, Smith WB, Macardle PJ, Wormald PJ. Eosinophilic mucus chronic rhinosinusitis: clinical subgroups or a homogeneous pathogenic entity? Laryngoscope. 2006. 116:1241–1247.

31. Brummund W, Kurup VP, Harris GJ, Duncavage JA, Arkins JA. Allergic sino-orbital mycosis. A clinical and immunologic study. JAMA. 1986. 256:3249–3253.

32. Marple BF, Gibbs SR, Newcomer MT, Mabry RL. Allergic fungal sinusitis-induced visual loss. Am J Rhinol. 1999. 13:191–195.

34. Waxman JE, Spector JG, Sale SR, Katzenstein AL. Allergic Aspergillus sinusitis: concepts in diagnosis and treatment of a new clinical entity. Laryngoscope. 1987. 97:261–266.
35. Bent JP 3rd, Kuhn FA. Diagnosis of allergic fungal sinusitis. Otolaryngol Head Neck Surg. 1994. 111:580–588.

36. Mukherji SK, Figueroa RE, Ginsberg LE, Zeifer BA, Marple BF, Alley JG, Cooper LL, Nemzek WR, Yousem DM, Jones KR, Kupferberg SB, Castillo M. Allergic fungal sinusitis: CT findings. Radiology. 1998. 207:417–422.

37. Houser SM, Corey JP. Allergic fungal rhinosinusitis: pathophysiology, epidemiology, and diagnosis. Otolaryngol Clin North Am. 2000. 33:399–408.
38. Sohail MA, Al Khabori MJ, Hyder J, Verma A. Allergic fungal sinusitis: can we predict the recurrence? Otolaryngol Head Neck Surg. 2004. 131:704–710.

40. Manning SC, Vuitch F, Weinberg AG, Brown OE. Allergic aspergillosis: a newly recognized form of sinusitis in the pediatric population. Laryngoscope. 1989. 99:681–685.
41. Corey JP, Delsupehe KG, Ferguson BJ. Allergic fungal sinusitis: allergic, infectious, or both? Otolaryngol Head Neck Surg. 1995. 113:110–119.

42. Nussenbaum B, Marple BF, Schwade ND. Characteristics of bony erosion in allergic fungal rhinosinusitis. Otolaryngol Head Neck Surg. 2001. 124:150–154.

43. Tsimikas S, Hollingsworth HM, Nash G. Aspergillus brain abscess complicating allergic Aspergillus sinusitis. J Allergy Clin Immunol. 1994. 94:264–267.
44. Kinsella JB, Bradfield JJ, Gourley WK, Calhoun KH, Rassekh CH. Allergic fungal sinusitis. Clin Otolaryngol Allied Sci. 1996. 21:389–392.

45. Liu JK, Schaefer SD, Moscatello AL, Couldwell WT. Neurosurgical implications of allergic fungal sinusitis. J Neurosurg. 2004. 100:883–890.

46. Shah TS, Sundaram P, Rege JD, Joshi JM. Proptosis in an asthmatic patient. Postgrad Med J. 2003. 79:710.

48. Goldstein MF, Atkins PC, Cogen FC, Kornstein MJ, Levine RS, Zweiman B. Allergic Aspergillus sinusitis. J Allergy Clin Immunol. 1985. 76:515–524.
49. Morpeth JF, Rupp NT, Dolen WK, Bent JP III, Kuhn FA. Fungal sinusitis: an update. Ann Allergy Asthma Immunol. 1996. 76:128–140.

50. Oğretmenoğlu O. Shortfalls of imaging diagnostics for allergic fungal sinusitis. Arch Otolaryngol Head Neck Surg. 2003. 129:1351.
51. Manning SC, Merkel M, Kriesel K, Vuitch F, Marple B. Computed tomography and magnetic resonance diagnosis of allergic fungal sinusitis. Laryngoscope. 1997. 107:170–176.

52. Zinreich SJ, Kennedy DW, Malat J, Curtin HD, Epstein JI, Huff LC, Kumar AJ, Johns ME, Rosenbaum AE. Fungal sinusitis: diagnosis with CT and MR imaging. Radiology. 1988. 169:439–444.

53. Diwakar A, Panjabi C, Shah A. Allergic bronchopulmonary aspergillosis, allergic Aspergillus sinusitis and their co-occurrence. Open Allergy J. 2008. 1:52–61.
54. Patterson R. Fishman AP, Elias JA, Fishman JA, Grippi MA, Kaiser CR, Senior RM, editors. Allergic bronchopulmonary aspergillosis and hypersensitivity reactions to fungi. Fishman's pulmonary diseases and disorders. 1998. Vol. 1:3rd ed. New York: McGraw-Hill;777–782.
55. Patterson R. Allergic bronchopulmonary aspergillosis: a historical perspective. Immunol Allergy Clin North Am. 1998. 18:471–478.
56. Safirstein BH. Allergic bronchopulmonary aspergillosis with obstruction of the upper respiratory tract. Chest. 1976. 70:788–790.

57. Sher TH, Schwartz HJ. Allergic Aspergillus sinusitis with concurrent allergic bronchopulmonary Aspergillus: report of a case. J Allergy Clin Immunol. 1988. 81:844–846.
58. Schwartz HJ, Witt WJ, Sher TH. Allergic bronchopulmonary aspergillosis and allergic aspergillus sinusitis: case report. Ann Allergy. 1992. 69:447–448.
59. Shah A, Bhagat R, Panchal N, Jaggi OP, Khan ZU. Allergic bronchopulmonary aspergillosis with middle lobe syndrome and allergic Aspergillus sinusitis. Eur Respir J. 1993. 6:917–918.
60. Bhagat R, Shah A, Jaggi OP, Khan ZU. Concomitant allergic bronchopulmonary aspergillosis and allergic Aspergillus sinusitis with an operated aspergilloma. J Allergy Clin Immunol. 1993. 91:1094–1096.
61. Venarske DL, deShazo RD. Sinobronchial allergic mycosis: the SAM syndrome. Chest. 2002. 121:1670–1676.
62. Shah A, Panjabi C. Contemporaneous occurrence of allergic bronchopulmonary aspergillosis, allergic Aspergillus sinusitis, and aspergilloma. Ann Allergy Asthma Immunol. 2006. 96:874–878.
63. Erwin GE, Fitzgerald JE. Case report: allergic bronchopulmonary aspergillosis and allergic fungal sinusitis successfully treated with voriconazole. J Asthma. 2007. 44:891–895.

64. Braun JJ, Pauli G, Schultz P, Gentine A, Ebbo D, de Blay F. Allergic fungal sinusitis associated with allergic bronchopulmonary aspergillosis: an uncommon sinobronchial allergic mycosis. Am J Rhinol. 2007. 21:412–416.

65. Prasad R, Garg R, Sanjay , Dua R. Concomitant allergic bronchopulmonary aspergillosis and allergic fungal sinusitis. Indian J Med Sci. 2007. 61:532–534.

66. Shah A, Kala J, Sahay S, Panjabi C. Frequency of familial occurrence in 164 patients with allergic bronchopulmonary aspergillosis. Ann Allergy Asthma Immunol. 2008. 101:363–369.

67. Agarwal R, Gupta D, Aggarwal AN, Behera D, Jindal SK. Allergic bronchopulmonary aspergillosis: lessons from 126 patients attending a chest clinic in north India. Chest. 2006. 130:442–448.
68. Campbell JM, Graham M, Gray HC, Bower C, Blaiss MS, Jones SM. Allergic fungal sinusitis in children. Ann Allergy Asthma Immunol. 2006. 96:286–290.

69. Schubert MS. Allergic fungal sinusitis: pathogenesis and management strategies. Drugs. 2004. 64:363–374.
70. Schubert MS, Goetz DW. Evaluation and treatment of allergic fungal sinusitis. II. Treatment and follow-up. J Allergy Clin Immunol. 1998. 102:395–402.

71. Shah A, Panjabi C. Biweekly therapy with prednisolone is effective in the management of allergic bronchopulmonary aspergillosis [Abstract]. Allergy Clin Immunol Int - J World Allergy Org. 2005. Suppl 1. 113.
72. Denning DW, O'Driscoll BR, Powell G, Chew F, Atherton GT, Vyas A, Miles J, Morris J, Niven RM. Randomized controlled trial of oral antifungal treatment for severe asthma with fungal sensitization: The Fungal Asthma Sensitization Trial (FAST) study. Am J Respir Crit Care Med. 2009. 179:11–18.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download