Abstract
Kigelia africana (Lam.) Benth. (Bignoniaceae) is a flowering plants in South, Central and West Africa and commonly known as the sausage tree (Eng.); worsboom (Afr.); umVunguta, umFongothi (Zulu); Modukguhlu (North Sotho); Muvevha (Venda). The dried, powdered fruits are used as dressing for wounds and ulcers, haemorrhoids, rheumatism, purgative, skin-firming, lactation in breastfeeding mothers. The aim of this study is to investigate the cytotoxic and apoptotic potentials of 70% ethanolic extracts of Kigelia africana fruits in HCT116 human colon cancer cells. Treatment of Kigelia africana fruits with various concentrations resulted in a sequence of characteristic of apoptosis, including loss of cell viability and morphological changes. Flow cytometry analysis showed Kigelia africana fruits increased the sub-G1 phase (apoptosis) population. Apoptosis confirmed by annexin V-fluorescein isothiocyanate and propidium iodide double staining in HCT116 human colon cancer cell lines. Moreover, analysis of the mechanism indicated that Kigelia africana fruits showed an increased Bax and Bcl-2 expressions in a dose-dependent manner, resulting in activation of hallmarks of apoptotic events, caspase-3, caspase-9 and cleaved poly-ADP-ribose polymerase. This is the first report to demonstrate the cytotoxicity of Kigelia africana fruits on HCT116 human colon cancer cells.
REFERENCES
(1). Jemal A., Bray F., Center M. M., Ferlay J., Ward E., Forman D. CA. Cancer J. Clin. 2011; 61:69–90.
(2). Line-Edwige M., Raymond F. G., Francois E., François E., Edouard N. E. Afr. J.Tradit. Complement. Altern. Med. 2009; 6:112–117.
(4). Thornberry N. A., Rano T. A., Peterson E. P., Rasper D. M., Timkey T., Garcia-Calvo M., Houtzager V. M., Nordstrom P. A., Roy S., Vaillancourt J. P., Chapman K. T., Nicholson D. W. J.Biol. Chem. 1997; 272:17907–17911.
(5). Wang X.Genes Dev. 2001; 15:2922–2933.
(6). Reuter S., Eifes S., Dicato M., Aggarwal B. B., Diederich M.Biochem. Pharmacol. 2008; 76:1340–1351.
(7). Balaban R. S., Nemoto S., Finket T.Cell. 2005; 120:483–495.
(8). Madesh M., Antonsson B., Srinivasula S. M., Alnemri E. S., Hajnóczky G. J.Biol. Chem. 2002; 277:5651–5659.
(9). Fernandes-Alnemri T., Litwack G., Alnemri E. S. J.Biol. Chem. 1994; 269:30761–30764.
(10). Saini S., Kaur H., Verma B., Ripudaman Singh S. K.Nat. Prod. Rad. 2009; 8:190–197.
(11). Higgins C. A., Bell T., Delbederi Z., Feutren-Burton S., McClean B., O'Dowd C., Watters W., Armstrong P., Waugh D., van den Berg H.Planta Med. 2010; 76:1840–1846.
(12). Agyare C., Dwobeng A. S., Agyepong N., Boakye Y. D., Mensah K. B., Ayande P. G., Adarkwa-Yiadom M.See comment in PubMed Commons below Adv. Pharmacol. Sci. 2013; Article ID 692613. 10.
(13). Picerno P., Autore G., Marzocco S., Meloni M., Sanogo R., Aquino R. P. J.Nat. Prod. 2005; 68:1610–1614.
(14). Carmichael J., DeGraff W. G., Gazdar A. F., Minna J. D., Mitchell J. B.Cancer Res. 1987; 47:936–942.
(15). Lizard G., Fournel S., Genestier L., Dhedin N., Chaput C., Flacher M., Mutin M., Panaye G., Revillard J. P.Cytometry. 1995; 21:275–283.
(16). Nicoletti I., Migliorati G., Paqliacci M. C., Grignani F., Riccardi C. J.Immunol. Methods. 1991; 139:271–279.
(17). Ryu M. J., Kim A. D., Kang K. A., Chung H. S., Kim H. S., Suh I. S., Chang W. Y., Hyun J. W.In Vitro Cell. Dev. Biol. Anim. 2013; 49:74–81.
(18). Strickland L., Letson G. D., Muro-Cacho C. A.Cancer Control. 2001; 8:252–261.
(19). Ryu M. J., Chung H. S.In Vitro Cell. Dev. Biol. Anim. 2015; 51:92–101.
(20). Bold R. J., Termuhlen P. M., McConkey D. J.Surg. Oncol. 1997; 6:133–142.
(21). Xu Y., Ge R., Du J., Xin H., Yi T., Sheng J., Wang Y., Ling C.Cancer Lett. 2009; 284:229–237.
(22). Novák B., Tyson J. J.Biochem. Soc. Trans. 2003; 31:1526–1529.
(23). Han C. R., Jun D. Y., Woo H. J., Jeong S. Y., Woo M. H., Kim Y. H.Bioorg. Med. Chem. Lett. 2012; 22:945–953.
(24). Cory S., Adams J. M.Nat. Rev. Cancer. 2002; 2:647–656.
(25). Nagappan A., Park K. I., Park H. S., Kim J. A., Hong G. E., Kang S. R., Lee D. H., Kim E. H., Lee W. S., Won C. K., Kim G. S.Food Chem. 2012; 135:1920–1928.
(26). Kim K. N., Ham Y. M., Moon J. Y., Kim M. J., Jung Y. H., Jeon Y. J., Lee N. H., Kang N., Yang H. M., Kim D., Hyun C. G.Food Chem. 2012; 135:2112–2117.
(27). Oliver F. J., de la Rubia G., Rolli V., Ruiz-Ruiz M. C., de Murcia G., Murcia J. M. J.Biol. Chem. 1998; 273:33533–33539.
(28). Cuadrado A., Nebreda A. R.Biochem. J. 2010; 429:403–417.
(29). Ahmed-Choudhury J., Williams K. T., Young L. S., Adams D. H., Afford S. C.Cell Signal. 2006; 18:456–468.
Fig. 1.
Cytotoxic effect of KAF in (A) normal human colon cells and (B) human colon cancer cells line. Cell viability at the indicated concentrations of KAF in HT-29 and HCT116 cells at 72 hr was assessed by MTT assay. ∗p < 0.05, significantly different from control cells.
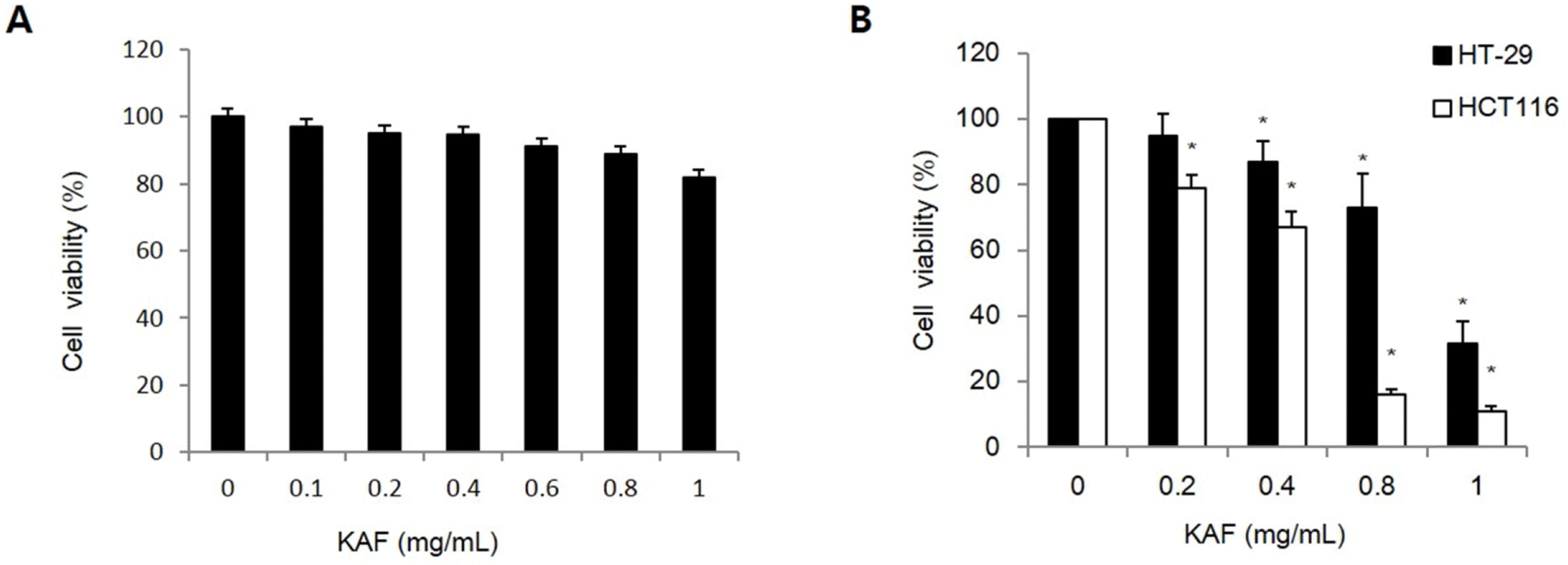
Fig. 2.
Induction of apoptosis by KAF in HCT116 human colon cancer cells. The formation of apoptotic bodies (arrows) in Hoechst-33258-stained cells observed by fluorescent microscopy.
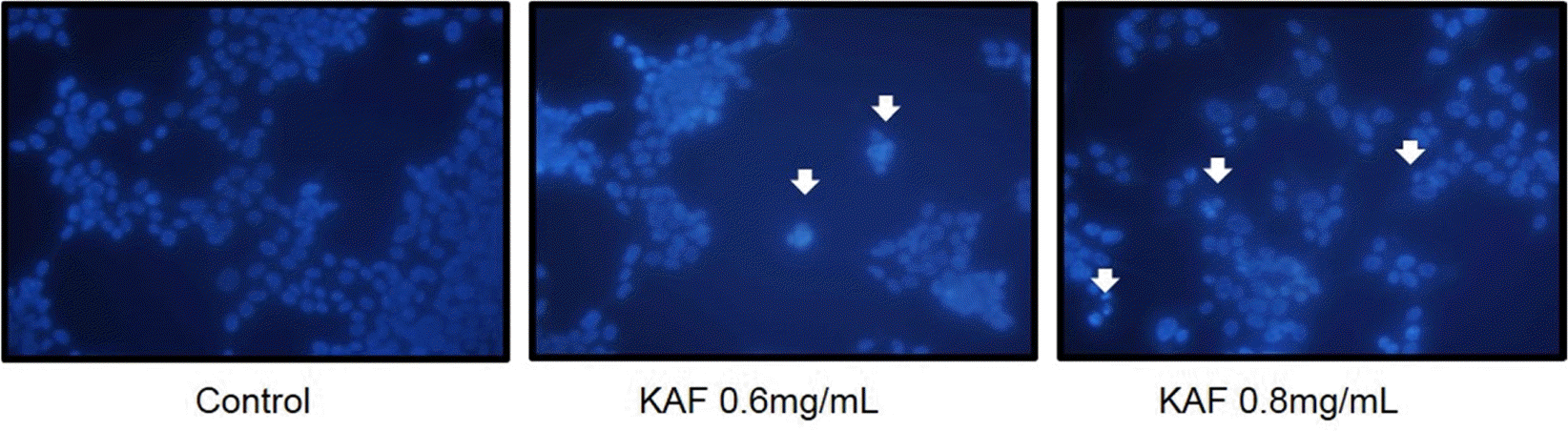
Fig. 3.
Effects of apoptosis by KAF in HCT116 human colon cancer cells. (A) Flow cytometric analysis of HCT116 human colon cancer cells incubated with KAF for 72 h. The right bottom quadrant represents Annexin V-stained cells (early-phase apoptotic cells). The top right quadrant represents PI- and Annexin V-stained cells (late-phase apoptotic cells). (B) Statistical analysis of apoptosis. ∗p < 0.05, significantly different from control cells.
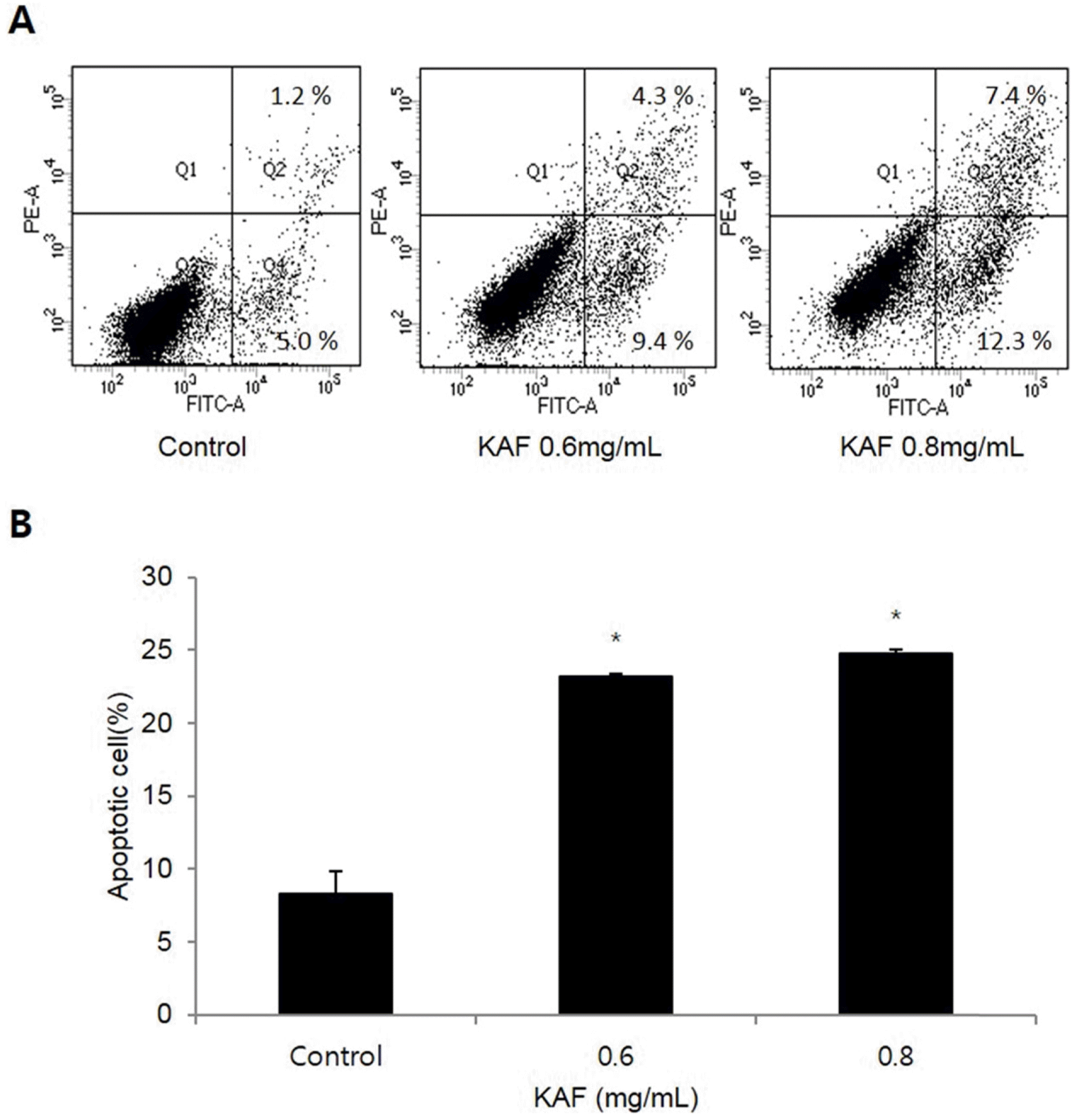
Fig. 4.
Effects of KAF on cell cycle phase distribution in HCT116 cells. After incubation with various concentrations of KAF for 72 hr, sub-G1 cells were detected by flow cytometry after propidium iodide stainning.∗p < 0.05, significantly different from control cells.
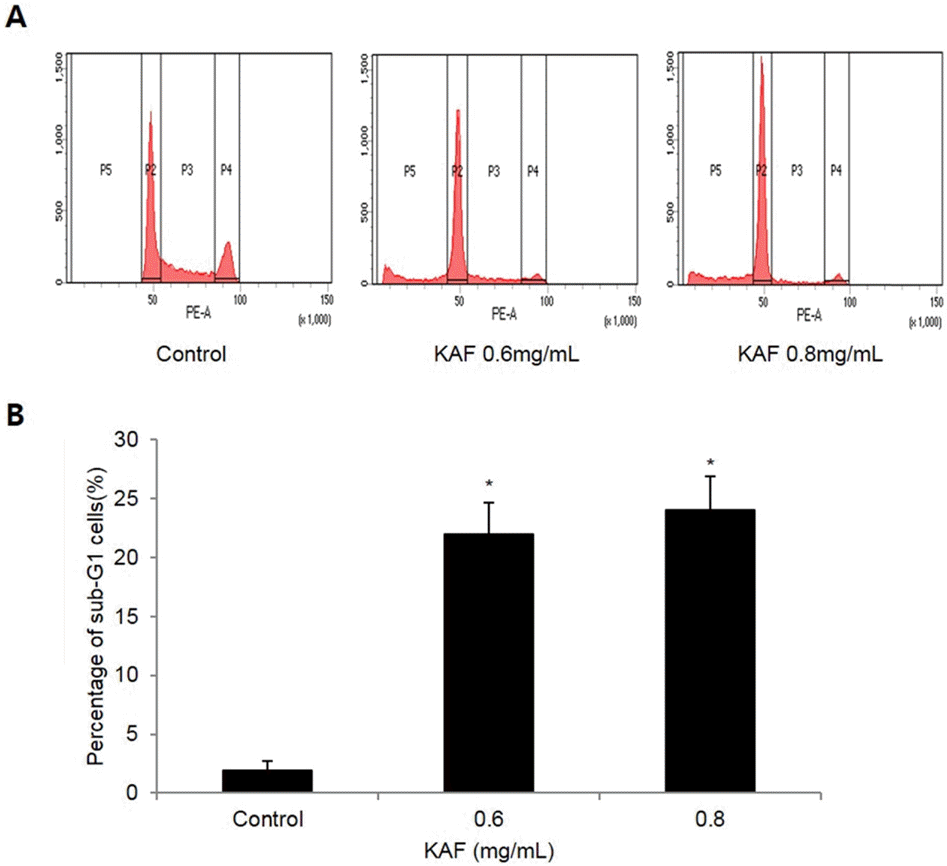
Fig. 5.
Effects of KAF on the expression of Bcl-2 family and caspase proteins in HCT116 cells. Cell lysates were electrophoresed Bcl-2, Bax, caspase-3, caspase-8, caspase-9, and cleaved PARP were detected by Western blotting analysis with the corresponding antibodies.

Fig. 6.
Effects of KAF on the expression of MAPK in HCT116 cells. Cell lysates were electrophoresed and JNK, ERK, and p38 were detected by Western blotting analysis with the corresponding antibodies.
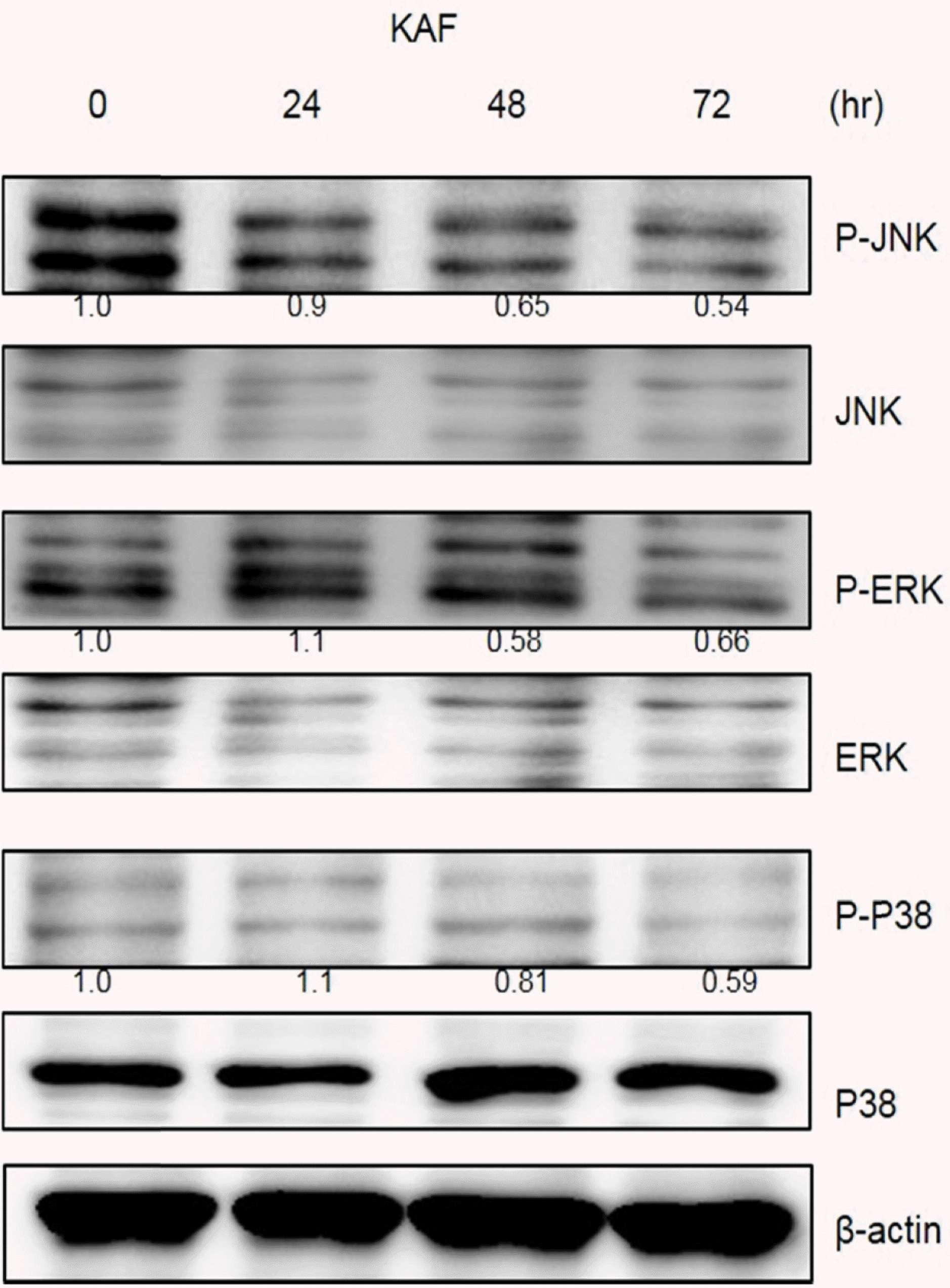
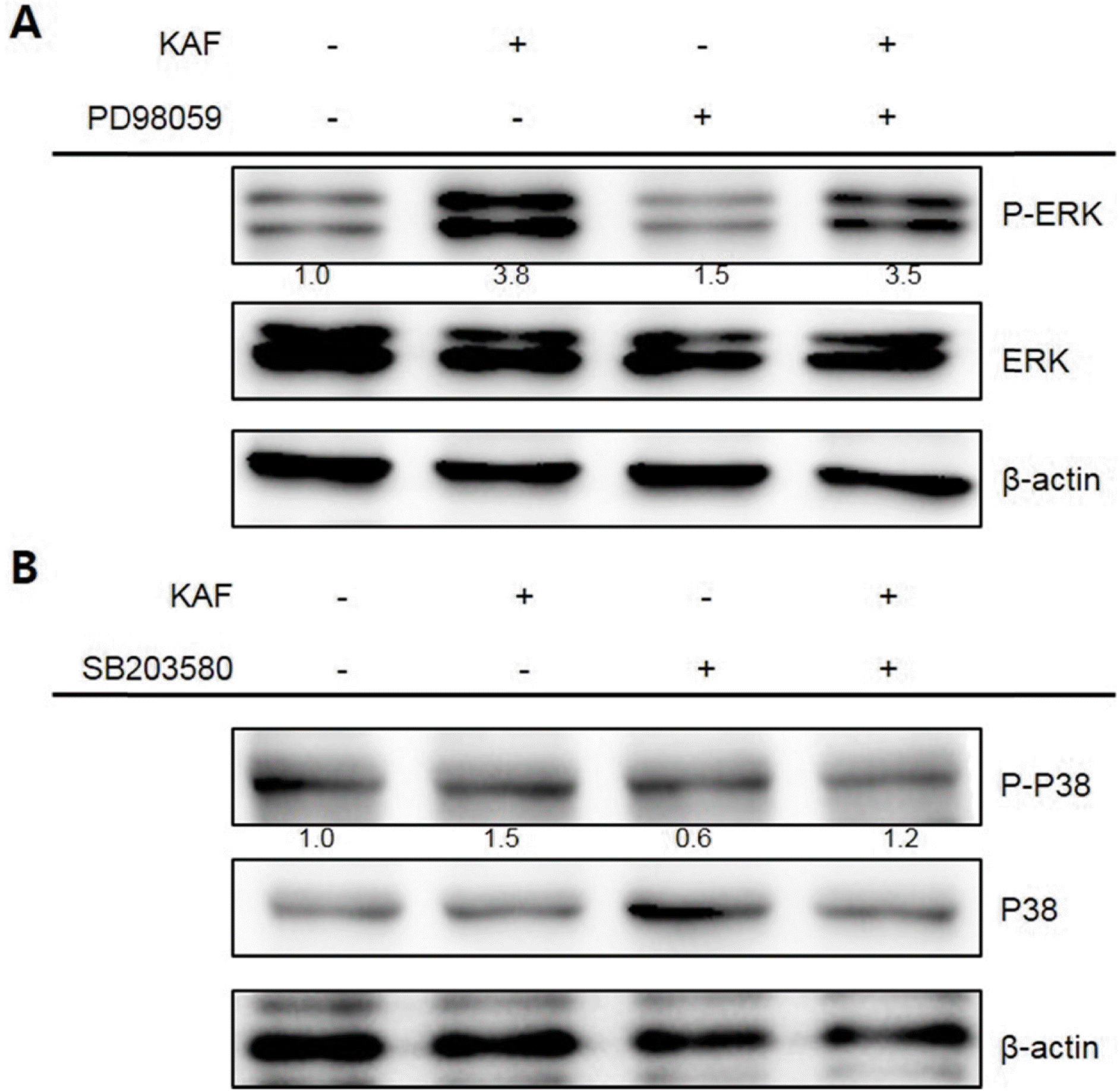
Fig. 7.
Role of ERK (A) and p38 (B) in KAF-induced apoptosis in HCT116 cells. The cells were stimulated with 0.8 mg/mL of KAF after pre-treatment with PD98059 and SB202190, respectively. Equal amounts of cell lysates were electrophoresed and MAPK-related protein expression form were detected by Western blotting analysis with corresponding antibodies.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download