Abstract
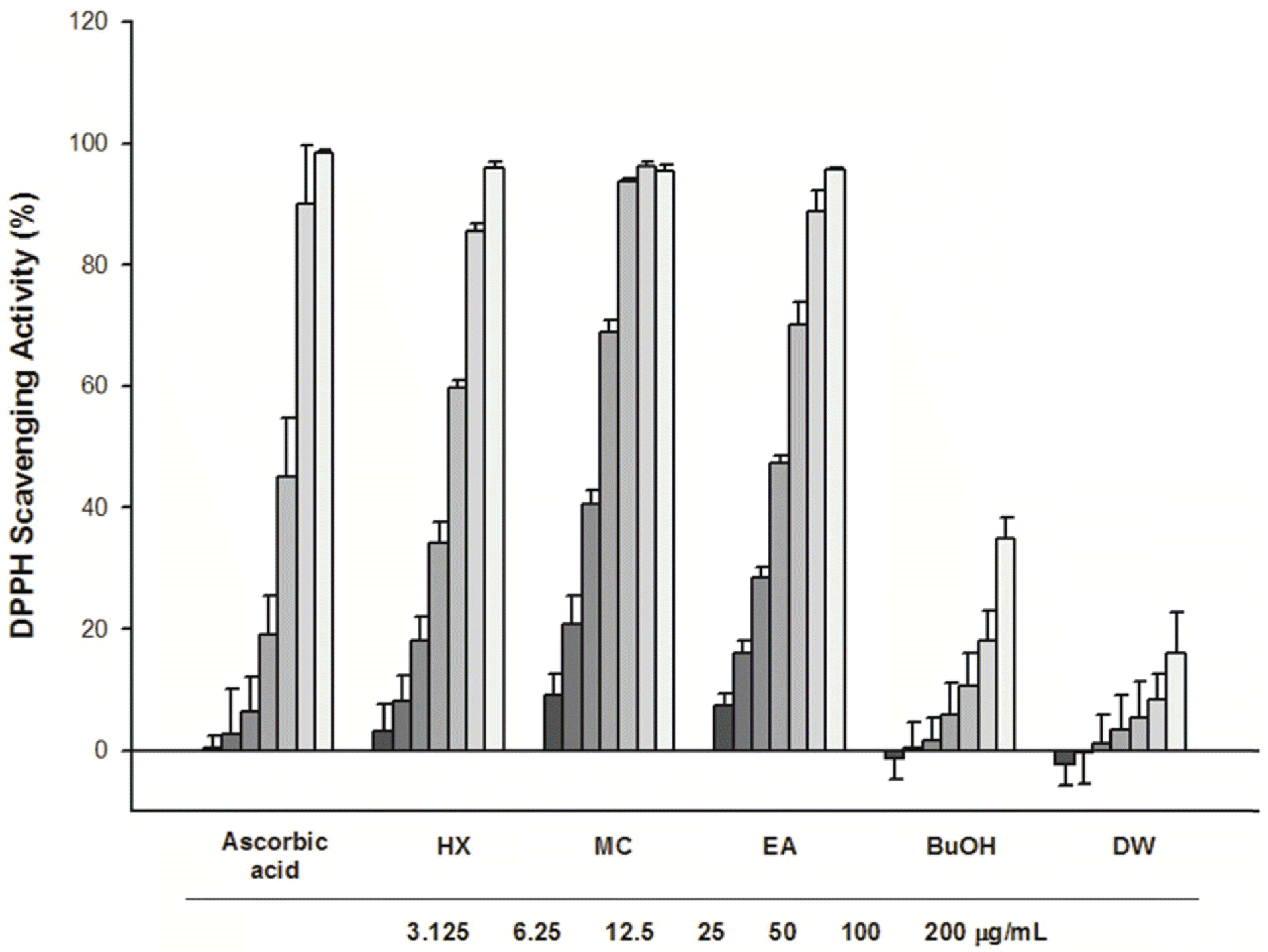
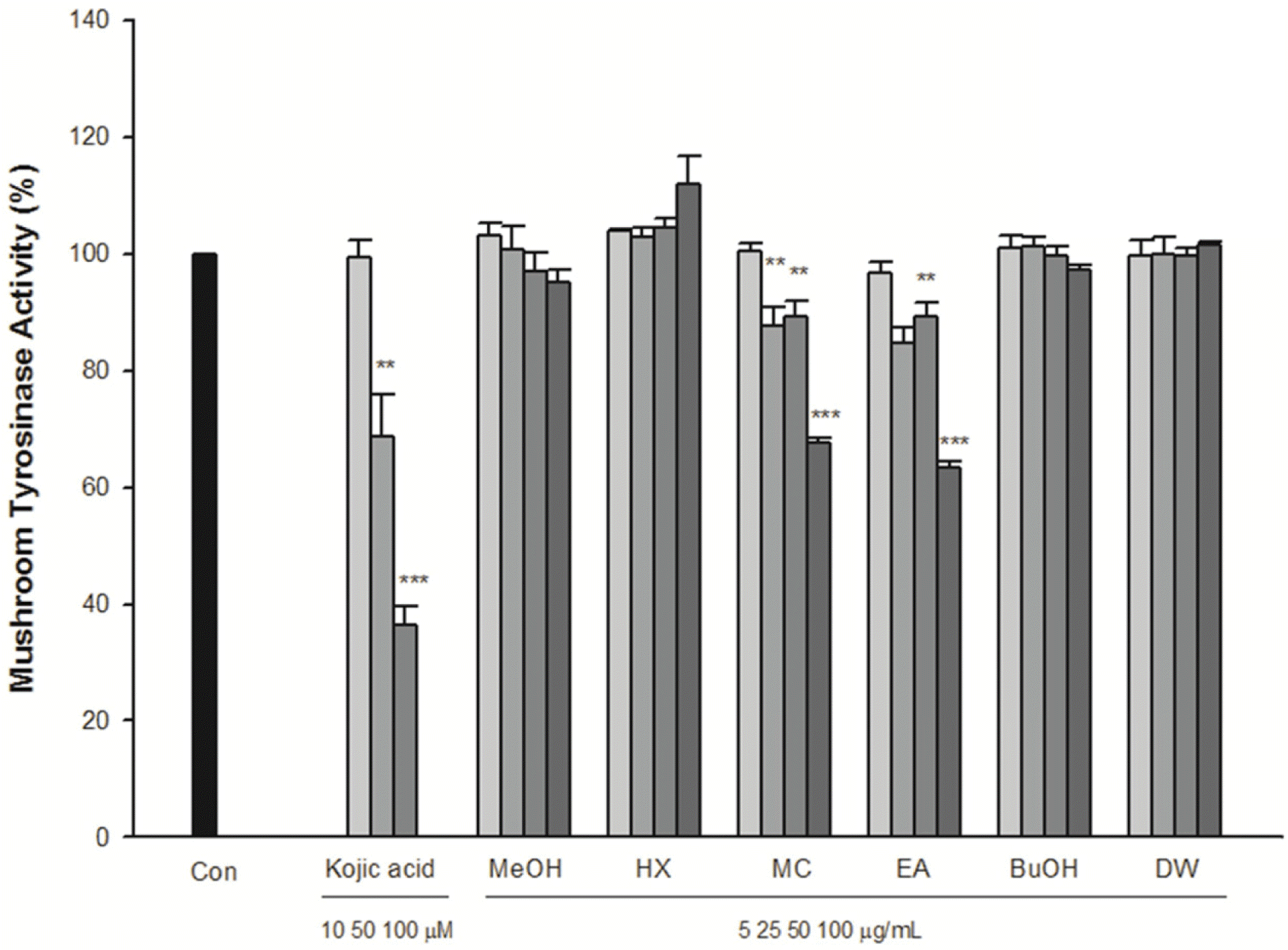
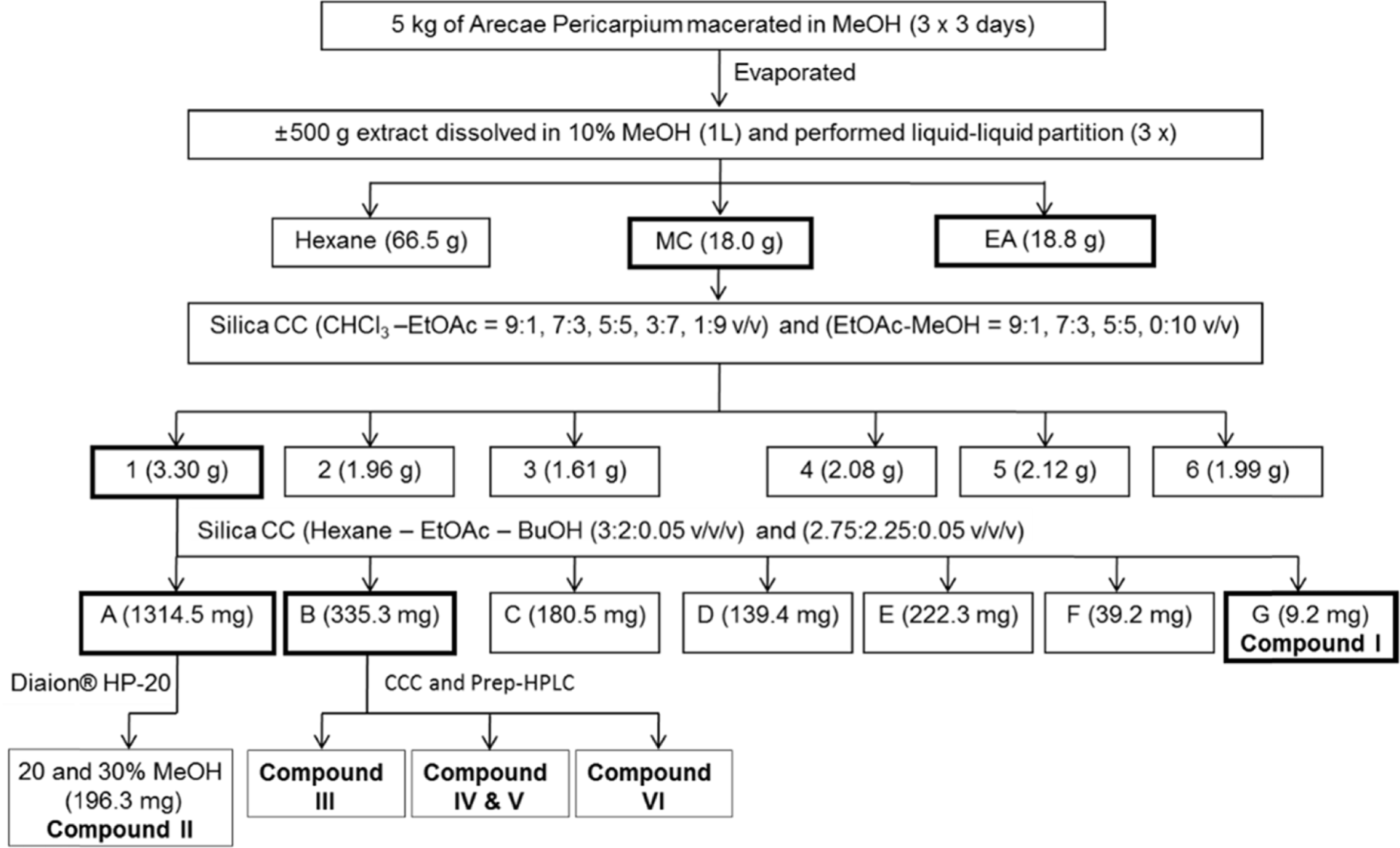
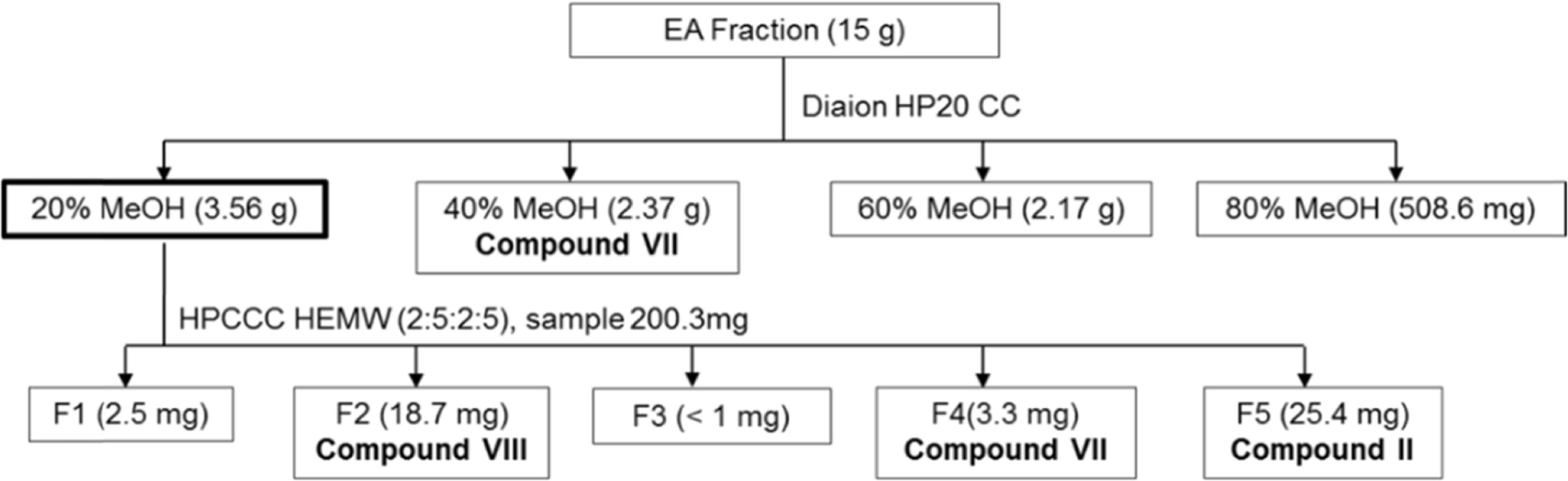
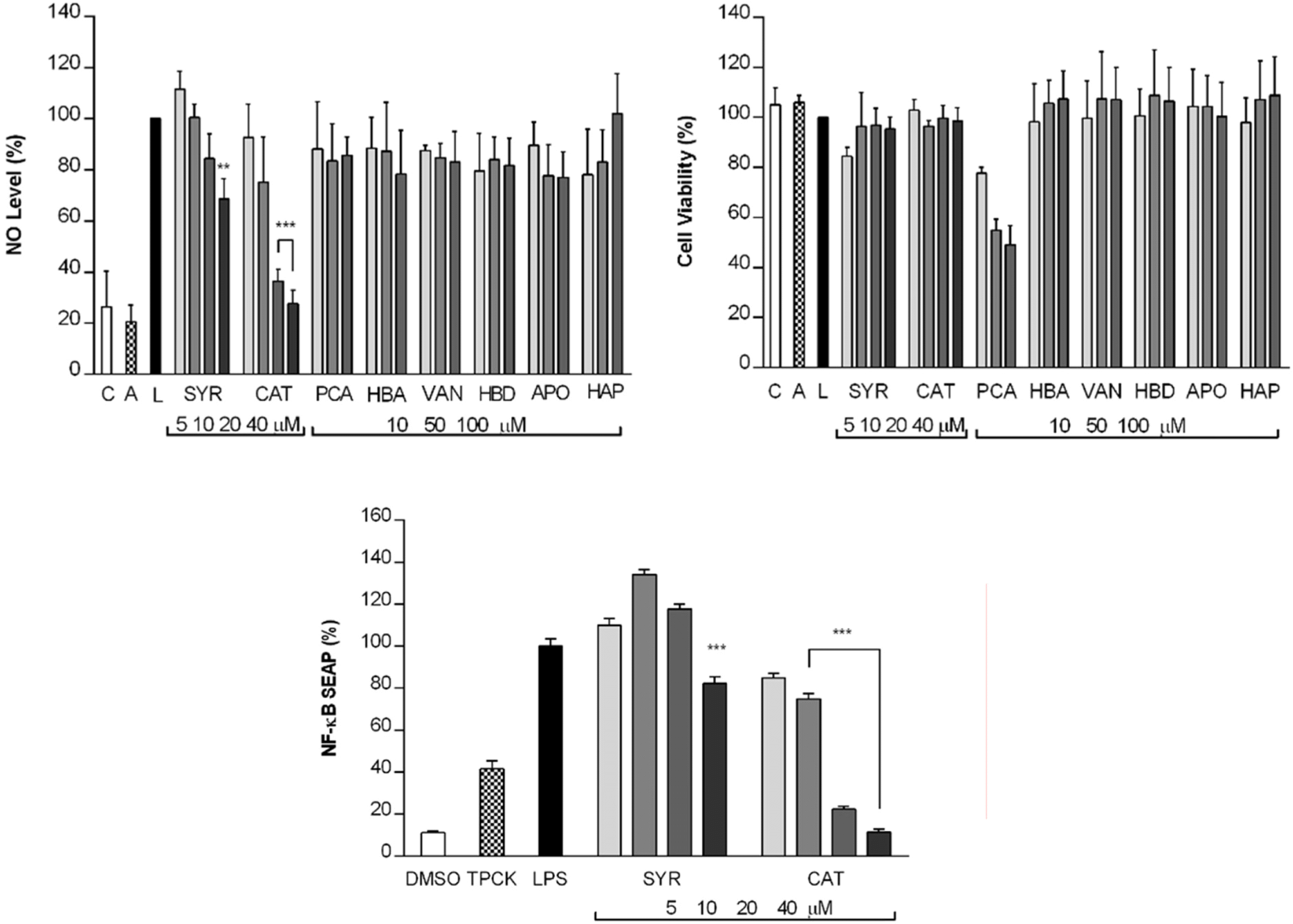
This study describes the anti-inflammatory, anti-oxidant, and melanogenesis inhibition activities of methanol extract and various organic solvent fractions of Arecae Pericarpium. We examined the inhibition of lipopolysaccharide (LPS)-induced nitric oxide (NO) production in RAW 264.7 cells, 1,1-diphenyl-2-picrylhydrazine (DPPH) scavenging activity, mushroom tyrosinase inhibition activity and melanin contents. The study showed that, among all tested fractions, methylene chloride fraction showed the strongest inhibition of LPS-induced NO production in RAW 264.7 cells (IC50 value 8.89µg/mL) and DPPH radical scavenging activity (EC50 value 21.39µg/ mL). Methylene chloride and ethyl acetate fractions similarly inhibited mushroom tyrosinase activity. Methanol extract exhibited strongest reduction of melanin content in B16F10 melanoma cells. Based on the bioactivity assay results, methylene chloride and ethyl acetate fractions were further separated. Eight phenolic compounds were isolated, which are dimeric syringol (1), catechol (2), 4-hydroxybenzaldehyde (3), vanillin (4), 4-hydroxya-cetophenone (5), apocynin (6), protocatechuic acid (7) and 4-hydroxybenzoic acid (8). Among the isolated compounds tested, catechol showed the strongest inhibition of LPS-induced NO production in RAW 264.7 cells. Catechol also showed the concentration-dependent NF-κ B inhibition activity. Arecae Pericarpium might have potentials to be developed as anti-inflammatory agent or dermatological product for skin-whitening agent.
Go to : 
REFERENCES
(1). Peng W., Liu Y. J., Wu N., Sun T., He X. Y., Gao Y. X., Wu C. J. J.Ethnopharmacol. 2015; 164:340–356.
(2). Yenjit P., Issarakraisila M., Intana W., Chantrapromma Kan. Postharvest Biol Technol. 2010; 55:129–132.
(3). Phaechamud T., Toprasri P., Chinpaisal C.Pharm. Biol. 2009; 47:242–247.
(4). Zamora R., Vodovotz Y., Billiar T. R.Mol Med. 2000; 6:347–373.
(5). Korhonen R., Lahti A., Kankaanranta H., Moilanen E.Curr. Drug Targets Inflamm. Allergy. 2005; 4:471–479.
(6). Sun J., Zhang X., Broderick M., Fein H.Sensors. 2003; 3:276–284.
(7). Kim Y. J., Uyama H.Cell Mol. Life Sci. 2005; 62:1707–1723.
(8). Briganti S., Camera E., Picardo M.Pigment Cell Res. 2003; 16:101–110.
(9). Lee K. K., Choi J. D. J.Cosmet. Sci. 1998; 49:351–359.
(10). Moon K. Y., Hahn B. S., Lee J., Kim Y. S.Anal. Biochem. 2001; 292:17–21.
(11). Prieto J. M.Procedure: Preparation of DPPH Radical, and antioxidant scavenging assay. 2012; https://www.researchgate.net/file. as of Jan. 10, 2016.
(12). Zhang W. M., Li B., Han L., Zhang H. D. Afr. J.Biotechnol. 2009; 8:3887–3892.
(13). Tsukamoto K., Jackson I. J., Urabe K., Montague P. M., Hearing V. J.EMBO J. 1992; 11:519–526.
Go to : 
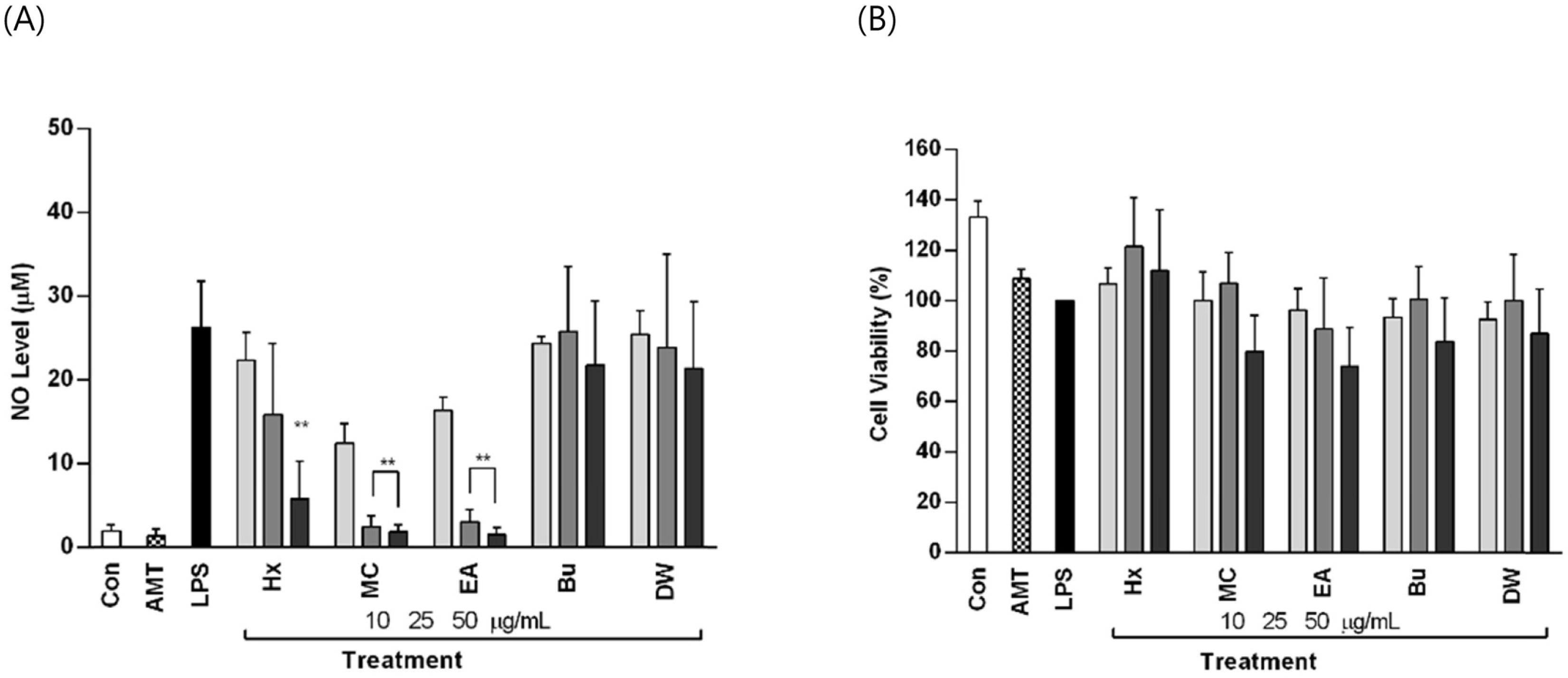 | Fig. 1.Effect of Arecae Pericarpium fractions on (A) LPS-induced NO production and (B) Cell viability in RAW 264.7 cells. |
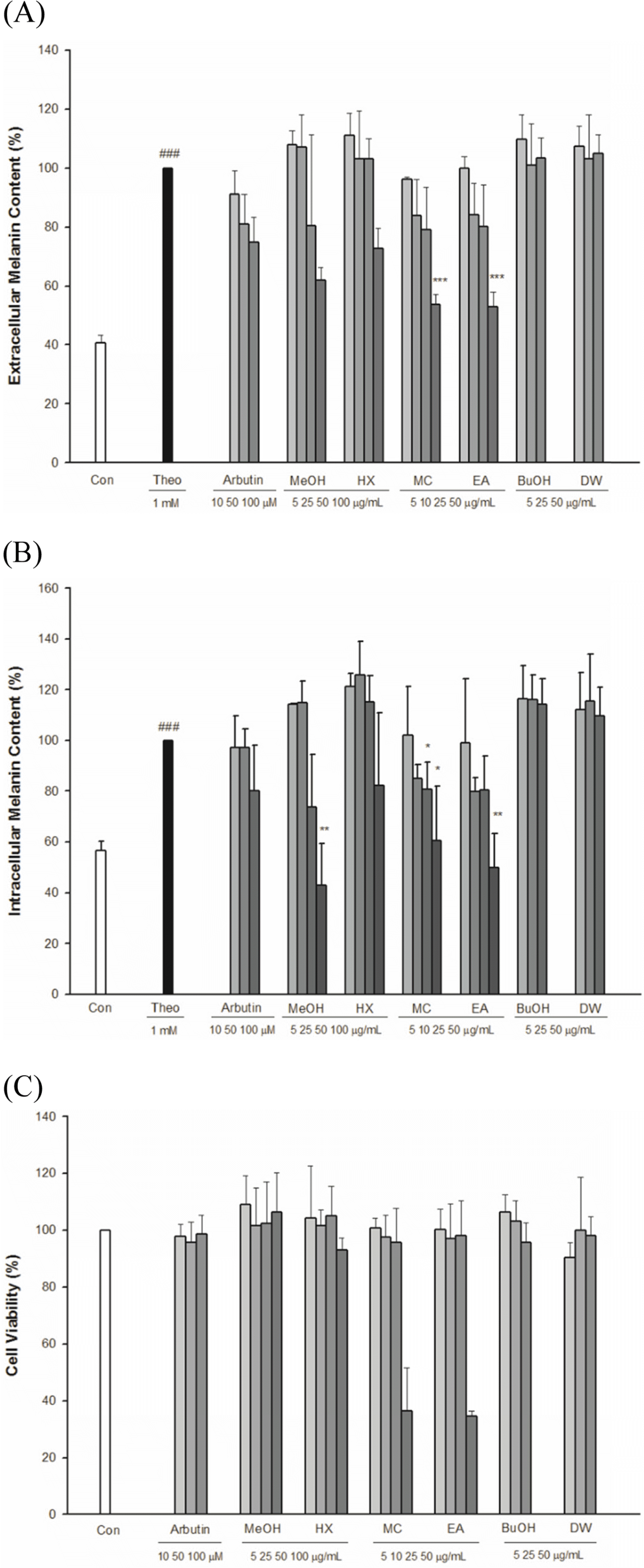 | Fig. 4.Effect of Arecae Pericarpium extract and fractions on (A) Extracellular melanin content (B) Intracellular melanin content (C) Cell viability in B1F10 melanoma cells. |




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download