1. Van Thuyne W, Van Eenoo P, Delbeke F. Nutritional supplements: prevalence of use and contamination with doping agents. Nutr Res Rev. 2006; 19:147–158. PMID:
19079882.

2. Eisenberg DM, Davis RB, Ettner SL, Appel S, Wilkey S, Van Rompay M, Kessler RC. Trends in alternative medicine use in the United States, 1990–1997: results of a follow-up national survey. JAMA. 1998; 280:1569–1575. PMID:
9820257.
3. Kobayashi MS, Han D, Packer L. Antioxidants and herbal extracts protect HT-4 neuronal cells against glutamate-induced cytotoxicity. Free Radic Res. 2000; 32:115–124. PMID:
10653482.

4. Rohdewald P. A review of the French maritime pine bark extract (Pycnogenol), a herbal medication with a diverse clinical pharmacology. Int J Clin Pharmacol Ther. 2002; 40:158–168. PMID:
11996210.

5. Packer L, Rimbach G, Virgili F. Antioxidant activity and biologic properties of a procyanidin-rich extract from pine (Pinus maritima) bark, pycnogenol. Free Radic Biol Med. 1999; 27:704–724. PMID:
10490291.

6. Dragendorff G. Die heilpflanzen der verschiedenen völker und zeiten: ihre anwendung, wesentlichen bestandtheile und geschichte. Ein handbuch für ärzte, apotheker, botaniker und droguisten. Stuttgart: Ferdinand Enke;1898.
7. Blazso G, Gábor M, Rohdewald P. Antiinflammatory activities of procyanidin-containing extracts from Pinus pinaster Ait. after oral and cutaneous application. Pharmazie. 1997; 52:380–382. PMID:
9183789.
8. Devaraj S, Vega-López S, Kaul N, Schönlau F, Rohdewald P, Jialal I. Supplementation with a pine bark extract rich in polyphenols increases plasma antioxidant capacity and alters the plasma lipoprotein profile. Lipids. 2002; 37:931–934. PMID:
12530550.

9. Majno G, Joris I. Cells, tissues, and disease: principles of general pathology. 2nd ed. New York (NY): Oxford University Press;2004.
10. Medzhitov R. Origin and physiological roles of inflammation. Nature. 2008; 454:428–435. PMID:
18650913.

11. Nehring SM, Bhimji SS. C reactive protein (CRP). Treasure Island (FL): StatPearls Publishing;2017.
12. Yamashita H, Shimada K, Seki E, Mokuno H, Daida H. Concentrations of interleukins, interferon, and C-reactive protein in stable and unstable angina pectoris. Am J Cardiol. 2003; 91:133–136. PMID:
12521622.

13. Ticinesi A, Lauretani F, Nouvenne A, Porro E, Fanelli G, Maggio M, Meschi T. C-reactive protein (CRP) measurement in geriatric patients hospitalized for acute infection. Eur J Intern Med. 2017; 37:7–12. PMID:
27594414.

14. Peng Q, Wei Z, Lau BH. Pycnogenol inhibits tumor necrosis factor-α-induced nuclear factor kappa B activation and adhesion molecule expression in human vascular endothelial cells. Cell Mol Life Sci. 2000; 57:834–841. PMID:
10892347.
15. Belcaro G, Cornelli U, Luzzi R, Cesarone MR, Dugall M, Feragalli B, Errichi S, Ippolito E, Grossi MG, Hosoi M, Cornelli M, Gizzi G. Pycnogenol® supplementation improves health risk factors in subjects with metabolic syndrome. Phytother Res. 2013; 27:1572–1578. PMID:
23359520.

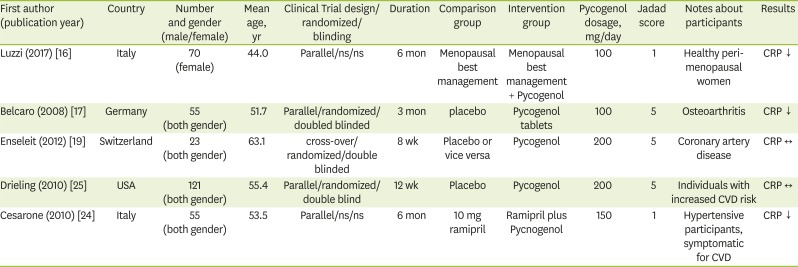
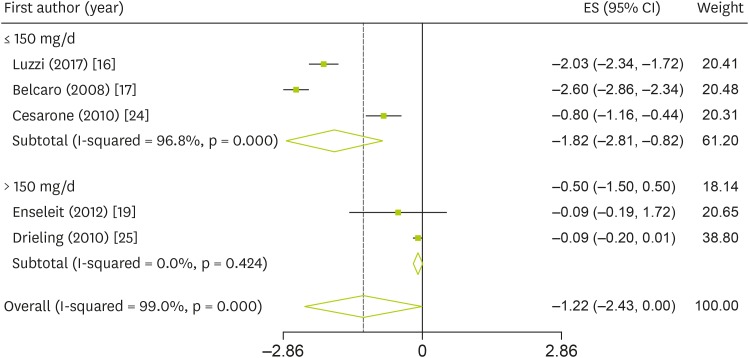
16. Luzzi R, Belcaro G, Hosoi M, Feragalli B, Cornelli U, Dugall M, Ledda A. Normalization of cardiovascular risk factors in peri-menopausal women with Pycnogenol®. Minerva Ginecol. 2017; 69:29–34. PMID:
28116886.

17. Belcaro G, Cesarone MR, Errichi S, Zulli C, Errichi BM, Vinciguerra G, Ledda A, Di Renzo A, Stuard S, Dugall M, Pellegrini L, Gizzi G, Ippolito E, Ricci A, Cacchio M, Cipollone G, Ruffini I, Fano F, Hosoi M, Rohdewald P. Variations in C-reactive protein, plasma free radicals and fibrinogen values in patients with osteoarthritis treated with Pycnogenol. Redox Rep. 2008; 13:271–276. PMID:
19017467.

18. Schäfer A, Chovanová Z, Muchová J, Sumegová K, Liptáková A, Duracková Z, Högger P. Inhibition of COX-1 and COX-2 activity by plasma of human volunteers after ingestion of French maritime pine bark extract (Pycnogenol). Biomed Pharmacother. 2006; 60:5–9. PMID:
16330178.
19. Enseleit F, Sudano I, Périat D, Winnik S, Wolfrum M, Flammer AJ, Fröhlich GM, Kaiser P, Hirt A, Haile SR, Krasniqi N, Matter CM, Uhlenhut K, Högger P, Neidhart M, Lüscher TF, Ruschitzka F, Noll G. Effects of Pycnogenol on endothelial function in patients with stable coronary artery disease: a double-blind, randomized, placebo-controlled, cross-over study. Eur Heart J. 2012; 33:1589–1597. PMID:
22240497.

20. Tang Y, Fung E, Xu A, Lan HY. C-reactive protein and ageing. Clin Exp Pharmacol Physiol. 2017; 44(Suppl 1):9–14. PMID:
28378496.

21. Picot J, Hartwell D, Harris P. The preferred reporting items for systematic reviews and meta-analyses checklist. Southampton: NIHR Journals Library;2012.
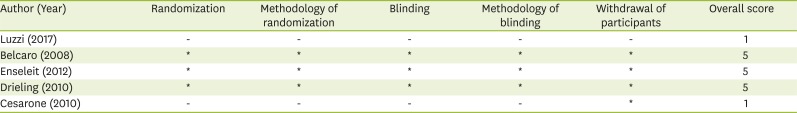
22. Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJ, Gavaghan DJ, McQuay HJ. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials. 1996; 17:1–12. PMID:
8721797.

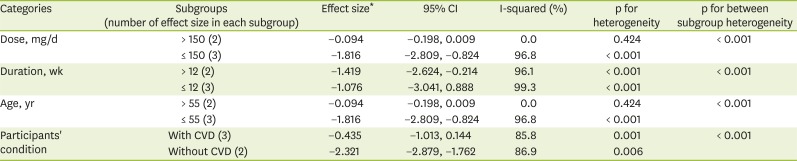
23. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003; 327:557–560. PMID:
12958120.

24. Cesarone MR, Belcaro G, Stuard S, Schönlau F, Di Renzo A, Grossi MG, Dugall M, Cornelli U, Cacchio M, Gizzi G, Pellegrini L. Kidney flow and function in hypertension: protective effects of Pycnogenol in hypertensive participants--a controlled study. J Cardiovasc Pharmacol Ther. 2010; 15:41–46. PMID:
20097689.

25. Drieling RL, Gardner CD, Ma J, Ahn DK, Stafford RS. No beneficial effects of pine bark extract on cardiovascular disease risk factors. Arch Intern Med. 2010; 170:1541–1547. PMID:
20876405.

26. Hayden MS, Ghosh S. Regulation of NF-κB by TNF family cytokines. Semin Immunol. 2014; 26:253–266. PMID:
24958609.

27. Jin X, Chen M, Yi L, Chang H, Zhang T, Wang L, Ma W, Peng X, Zhou Y, Mi M. Delphinidin-3-glucoside protects human umbilical vein endothelial cells against oxidized low-density lipoprotein-induced injury by autophagy upregulation via the AMPK/SIRT1 signaling pathway. Mol Nutr Food Res. 2014; 58:1941–1951. PMID:
25047736.

28. Salazar J, Martínez MS, Chávez-Castillo M, Núñez V, Añez R, Torres Y, Toledo A, Chacín M, Silva C, Pacheco E, Rojas J, Bermúdez V. C-reactive protein: an in-depth look into structure, function, and regulation. Int Sch Res Notices. 2014; 2014:653045. PMID:
27433484.

29. Agrawal A, Cha-Molstad H, Samols D, Kushner I. Overexpressed nuclear factor-kappaB can participate in endogenous C-reactive protein induction, and enhances the effects of C/EBPbeta and signal transducer and activator of transcription-3. Immunology. 2003; 108:539–547. PMID:
12667216.

30. Watanabe H, Fan B, Ikuyama S. Pycnogenol, a French maritime pine extract, suppresses RANKL-stimulated osteoclast differentiation of RAW264.7 cells. In : The Endocrine Society's 93rd Annual Meeting & Expo; 2011 Jun 4–7; Boston, MA. Washington, D.C.: Endocrine Society;2011. p. P1–P191.
31. Rohdewald PJ. Pycnogenol®, French maritime pine bark extract. In : Coates PM, editor. Encyclopedia of dietary supplements. 2nd ed. New York (NY): Marcel Dekker;2005. p. 545–553.
32. Fan B, Dun SH, Gu JQ, Guo Y, Ikuyama S. Pycnogenol attenuates the release of proinflammatory cytokines and expression of perilipin 2 in lipopolysaccharide-stimulated microglia in part via inhibition of NF-κB and AP-1 activation. PLoS One. 2015; 10:e0137837. PMID:
26367267.

33. Gulati OP. Pycnogenol(R) in chronic venous insufficiency and related venous disorders. Phytother Res. 2014; 28:348–362. PMID:
23775628.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download