Abstract
Teratomas of the central nervous system are rare and are frequently found in children and young adults. Cystic teratomas found in infancy is a well-recognized but infrequent entity. Intracranial teratomas,like teratomas in general, tend to arise from midline structures such as the pineal gland, but has rarely been found in the third ventricle. We report a rare case of a 6-month-old infant with a mature cystic teratoma of the third ventricle with a review of literatures
Intracranial teratomas account for 0.5% of all intracranial tumors and 2-4% of intracranial tumors in children12.Teratomas have a character of arising from deep midline structures, particularly the pineal and suprasellar regions, but has rarely been found in the third ventricle. They are one of the radioresistant tumors and total removal of the tumors is the treatment of choice3. We present a rare case of 6-month-old infant with a mature cystic teratoma in the third ventricle with review of literatures.
A 6-month-old infant was admitted with a 4-day history of vomiting. On admission, he was lethargic and showed a mild degree of motor weakness of both upper extremities.Head circumference of the patient was 46 cm (97th percentile).Brain ultrasonography showed a lobulating echogenic lesion in the third ventricle with cystic extension to the right lateral ventricle with considerable enlargement of the lateral and third ventricles. Further evaluation was made with computed tomography and magnetic resonance (MR) imaging, which showed a huge mass of approximately 2.1×2.3×2.9 cm in the third ventricle consisting of multicystic components and the portions of fat tissue and calcification, suggesting a teratomaFig 1.. The results of hormonal tumor marker studies, including human chorionic gonadotropin, α-fetoprotein, and carcinoembryonic antigen, were within normal ranges.On the 3rd hospital day, the patient showed unresponsiveness,extremity rigidity and tonicclonic movement. The anterior fontanel was quite tense and bulging. He seldom moved spontaneously. The pupils were 6 mm sized and showed no light reflex. This sudden change of condition was in accord with a hydrocephalic fit. The patient underwent emergency surgery for tumor removal. Because of the location of the tumor, the patient underwent a right frontal craniotomy and interhemispheric transcallosal approach followed by a ventriculostomy for the resection of the tumor and improvement of the hydrocephalus. The tumor was noted to have both cystic and solid components. Fluid aspirated from the cystic component was yellow and clear, with the appearance of oil or cholesterol. There were several firm calcified deposits of tumor that were tightly adhered to the wall of the third ventricle.Gross examination of the specimen demonstrated a grayish white soft tissue, measuring 2.8×2.5×2 cm in dimensions. The cut surface was homogenous tan colored and showed focal calcificationFig 2.. Histopathological examination showed a mixture of epidermis, cartilage, bone, muscle and adipose tissue. Immature tissue components were not identifiedFig 3. The lesion was diagnosed as a mature cystic teratoma
Postoperative period was uneventful except for the occurrence of several intermittent generalized seizures which were successfully managed with antiepileptic drugs. Neurological status improved gradually. On the postoperative 2 weeks, the patient underwent a subduro-peritoneal shunt (S-P shunt) to reduce the subdural hygroma of the bilateral cerebral convexities. Brain MR images, obtained 4 months after surgery, demonstrated a small residual mass in the left foramen Monro and reduced ventriculomegaly. At present, he lives well with mild global developmental delay
Teratomas of the central nervous system are rare and are frequently found in children and young adults45. Teratomas are types of germ cell tumors composed of tissue elements derived from all three primitive germ cell layers of the embryo: endoderm, mesoderm, and ectoderm6. Histopathologically, teratomas have been further classified into mature, immature, and teratoma with malignant transformation, and comprising only mature elements is uncommon 367. Although teratomas may originate anywhere along the midline, they are most commonly become implanted in the sacrococcygeal, gonadal, mediastinal, retroperitoneal, or intracranial regions18. Intracranial teratomas, like teratomas in general, tend to arise from midline structures with the pineal gland being the most common. Their occurrence in the third ventricle has been reported less frequently9.
Recently, Goyal et al.10reviewed 12 cases of pediatric intracranial teratomas that were treated at a single institute. Six cases of teratoma were located in the third ventricle. Histologically, eight cases were classified as mature teratomas and four as immature teratomas. All four cases of immature teratoma were located in the posterior third ventricle.
Zhou et al.3 reported 1 case of a mature cystic teratoma attached to the wall of the third ventricle. Cystic teratomas arising from the wall of the third ventricle are unusual and most of them are malignant. Furthermore, teratomas comprising of only mature elements is extremely rare, as in this case. Thus, we found a teratoma arising from the wall of the third ventricle of a 6-month-old infant to be extremely rare.
The radiological appearance of a teratoma varies with its contents. They are lobulated lesions with variable contrast enhancement and features such as intratumoral calcifications, cystic regions, and low-signal attenuated areas characteristic of fat11. However, it is impossible to make the diagnosis of a teratoma on the basis of radiology imaging alone. The definitive diagnosis of intracranial teratomas is on the basis of histopathology. Mature teratomas consist of fully differentiated ectodermal, mesodermal, and endodermal components. Ectodermal derivatives such as skin, skin adnexae, and neural tissues may be present. The mesoderm is frequently represented by cartilage, bone, fat, fibrous tissue, and smooth muscle. Endodermal components may consist of respiratory and gastrointestinal tract epithelial cells12. In our case, the teratoma showed mature tissue components such as bronchial cartilage, respiratory epithelium, bone, muscle, adipose tissue, and squamous cell lining epithelium.
Radical resection of the tumor is the treatment of choice for intracranial teratomas, which was successfully done in this case. The approach depends on the location of the tumor. Some patients might require a cerebrospinal fluid (CSF) diversion procedure in addition to tumor excision for treatment of an associated hydrocephalus with third ventricular lesions and prevention of CSF leak in cases associated with encephaloceles 10. In our case, the patient underwent a S-P shunt procedure to reduce the subdural hygroma after a transcallosal approach. Radiotherapy or chemotherapy should be reserved for immature teratomas or teratomas with malignant transformation. Mature teratomas are benign and have been reported to have 10-year-survival rates of up to 93%13.
In conclusion, the clinical presentation of a mature cystic teratoma in the third ventricle is rare but should be considered when the mass shows variable contrast enhancement and features such as calcification or cystic regions. Due to its location, immediate treatment such as surgical resection or CSF diversion procedures are necessary to prevent catastrophic events such as a hydrocephalic fit.
Figures and Tables
Fig. 1
A: Preoperative CT scan reveals a mass in the third ventricle with extension to the right lateral ventricle, consisting of fat components (white arrow). B and C: Preoperative magnetic resonance (MR) axial image shows a heterogenous mass with portions of calcification (white arrow) and fat tissue (black arrow), suggesting a teratoma. D: Preoperative MR coronal image shows a multi-cystic mass (asterisks) involving the third and right lateral ventricles.
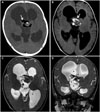
Fig. 2
A: Gross view of the tumor specimen shows a grayish white soft tissue, measuring 2.8×2.5×2 cm in dimensions. B: Gross view of the cut surface of the tumor specimen shows homogenous tan color and focal calcification.

Fig. 3
A: Low power photographic veiw of the tumor showing a mixture of bronchial lining epithelium, bronchial glands, muscle, adipose tissue and squamous cell lining cyst filled with keratin materials (hematoxylin-eosin, ×20). B: Representitive view of the tumor components consist of bronchial repiratory epithelium and bronchial cartilage (hematoxylin-eosin, ×100)

References
1. Goyal N, Singh PK, Kakkar A, Sharma MC, Mahapatra AK. Mature teratoma in association with neural tube defect (occipital encephalocele): series of four cases and review of the literature. Pediatr Neurosurg. 2012; 48:67–72.

2. Sinha VD, Dharker SR, Pandey CL. Congenital intracranial teratoma of the lateral ventricle. Neurol India. 2001; 49:170–173.
3. Zhou P, Li Y, Yang Z, Shu J. Mature cystic teratoma of skull base and attached to the wall of third ventricle. Turk Neurosurg. 2014; 24:292–293.
4. Echevarria ME, Fangusaro J, Goldman S. Pediatric central nervous system germ cell tumors: a review. Oncologist. 2008; 13:690–699.

5. Jubran RF, Finlay J. Central nervous system germ cell tumors: controversies in diagnosis and treatment. Oncology (Williston Park). 2005; 19:705–711. discussion 711-2, 715-7, 721.
6. Agrawal M, Uppin MS, Patibandla MR, et al. Teratomas in central nervous system: a clinico-morphological study with review of literature. Neurol India. 2010; 58:841–846.

7. Verma R, Malone S, Canil C, Jansen G, Lesiuk H. Primary skull-based yolk-sac tumour: case report and review of central nervous system germ cell tumours. J Neurooncol. 2011; 101:129–134.

8. Sandow BA, Dory CE, Aguiar MA, Abuhamad AZ. Best cases from the AFIP: congenital intracranial teratoma. Radiographics. 2004; 24:1165–1170.
9. Kim JM, Cheong JH, Yi HJ, Bak KH, Kim CH, Oh SJ. Metachronous germinoma after total removal of mature teratoma in the third ventricle: a case report. J Korean Med Sci. 2002; 17:287–291.

10. Goyal N, Kakkar A, Singh PK, et al. Intracranial teratomas in children: a clinicopathological study. Childs Nerv Syst. 2013; 29:2035–2042.

11. Rosenblum MK, Nakazato Y, Matsutani M. CNS germ cell tumours. In : Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, editors. WHO classification of tumours of the central nervous system. 4th ed. IARC: Lyon;2007. p. 197–204.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download